NEW
YORK, July 5, 2024 /PRNewswire/ -- The
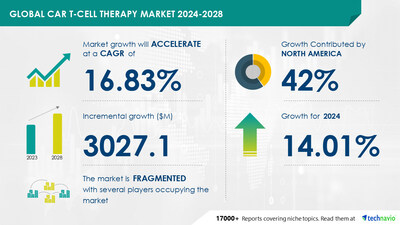
global car T-cell therapy market size is estimated to
grow by USD 3.02 billion from
2024-2028, according to Technavio. The market is estimated to grow
at a CAGR of 16.83% during the forecast
period. Growing awareness regarding car t-cell
therapy is driving market growth, with a trend
towards rising number of product approvals and clinical trials
related to car T- cell therapy. However, high cost associated
with car t-cell therapy poses a challenge. Key market players
include ACROBIOSYSTEMS INC., Allogene Therapeutics Inc., Autolus
Therapeutics plc, Bristol Myers Squibb Co., Celyad Oncology SA, Eli
Lilly and Co., Fate Therapeutics Inc., Fortress Biotech Inc.,
Gilead Sciences Inc., GlaxoSmithKline Plc, Johnson and Johnson
Services Inc., Les Laboratoires Servier, Merck KGaA, Miltenyi
Biotec B.V. And Co. KG, Noile Immune Biotech Inc., Novartis AG,
Pfizer Inc., Sangamo Therapeutics Inc, Sorrento Therapeutics Inc.,
and TCR2 Therapeutics Inc..
Get a detailed analysis on regions, market
segments, customer landscape, and companies - Click for the
snapshot of this report
|
Forecast
period
|
2024-2028
|
|
Base Year
|
2023
|
|
Historic
Data
|
2018 - 2022
|
|
Segment
Covered
|
End-user (Hospitals and
Cancer treatment centers), Type (CD19, CD22, BCMA, and Others), and
Geography (North America, Europe, Asia, and Rest of World
(ROW))
|
|
Region
Covered
|
North America, Europe,
Asia, and Rest of World (ROW)
|
|
Key companies
profiled
|
ACROBIOSYSTEMS INC.,
Allogene Therapeutics Inc., Autolus Therapeutics plc, Bristol Myers
Squibb Co., Celyad Oncology SA, Eli Lilly and Co., Fate
Therapeutics Inc., Fortress Biotech Inc., Gilead Sciences Inc.,
GlaxoSmithKline Plc, Johnson and Johnson Services Inc., Les
Laboratoires Servier, Merck KGaA, Miltenyi Biotec B.V. And Co. KG,
Noile Immune Biotech Inc., Novartis AG, Pfizer Inc., Sangamo
Therapeutics Inc, Sorrento Therapeutics Inc., and TCR2 Therapeutics
Inc.
|
Key Market Trends Fueling Growth
The global CAR T-cell therapy market is experiencing significant
growth due to an increasing number of product approvals and
clinical trials. For instance, Yescarta, the first CAR T-cell
therapy to receive FDA approval for relapsed or refractory large
B-cell lymphoma, was granted approval in February 2022. Similarly, ciltacabtagene
autoleucel (Carvykti) for multiple refractory myeloma received FDA
approval in the same month. The number of clinical trials for CAR-T
cell therapies has surged, with 415 trials registered on
Clinicaltrials.gov as of November 23,
2022. These trials cover various stages of development and
focus on different types of cancer and patient groups. Supportive
government policies, rising research and development expenditure,
and the potential to treat various types of cancer are driving the
growth of the global CAR T-cell therapy market.
The Car T-cell therapy market is experiencing significant
financial backing with companies like Juno Therapeutics and Tessa
Therapeutics leading the charge in clinical trials for blood cancer
treatments. Juno's T-cell therapy, Tisagenlecleucel, has shown
promising results in treating Non-Hodgkin lymphoma and leukemia.
Tessa Therapeutics' TT-02 is also making strides in solid tumors,
including ovarian cancer. Adaptimmune, Celgene Corporation, and
others are focusing on scalability and cost-effectiveness while
maintaining product consistency. CAR T-cell therapies are
revolutionizing cancer treatment, with precision medicine and
tailoring treatments to individual patients becoming increasingly
important. Hematologic malignancies, such as leukemia and lymphoma,
are the primary indications. Despite the high cost and systemic
toxicity, investments continue to pour in due to the potential for
a cure. Reimbursement policies, public awareness, patient
population, and healthcare infrastructure are key factors
influencing market growth. Innovation in cell-based therapies,
including genetic modification using CRISPR/Cas9, is driving
progress in cancer treatments. The prevalence of cancer and
incidence of specific diseases, such as leukemia and lymphoma,
continue to fuel demand. Quality control measures are essential to
ensure safety and efficacy, making patient-centric approaches
crucial for success. With the growing patient population and
disposable income, the market for Car T-cell therapies is poised
for continued growth.
Discover 360° analysis of this market. For
complete information, schedule your consultation- Book
Here!
Market Challenges
- The global CAR T-cell therapy market faces a significant
challenge due to the high cost of this innovative treatment. The
cost is influenced by the complex manufacturing process,
personalized nature, and specialized healthcare infrastructure
requirements. CAR T-cell therapies involve collecting a patient's
T-cells through leukapheresis, modifying them in the lab, expanding
their quantity, and reinfusing them back. This process necessitates
specialized facilities, equipment, and expertise, contributing to
the overall cost. Moreover, each patient's therapy is personalized,
adding complexity and cost. Extensive clinical trials and research
further increase the cost burden. CAR T-cell acquisition costs
range from USD373,000 to USD475,000 per infusion, not including additional
procedures or facility fees. The inpatient setting for therapy
administration adds an additional cost of USD79,466 to USD85,267. Consequently, the high costs
associated with CAR T-cell therapies pose a significant challenge
to the market's growth during the forecast period.
- The Car T-cell therapy market is experiencing significant
growth due to the potential of these treatments for blood cancer,
specifically lymphoma and leukemia. Companies like Juno
Therapeutics and Tessa Therapeutics are leading the charge with
their CAR T-cell therapies, TT-02 for solid tumors, and Tessa's
TCR-based therapy for ovarian cancer. However, challenges remain,
including securing financial backing for clinical trials, ensuring
scalability, cost-effectiveness, and product consistency. The
market is also facing hurdles in treating solid tumors and
addressing systemic toxicity. Collaborations with companies like
Celgene Corporation and innovation through precision medicine are
key to overcoming these challenges. Reimbursement policies, public
awareness, investments, and patient population size are also
crucial factors. Genetic modification through CRISPR/Cas9 holds
promise for improving CAR T-cell therapies. Despite these
challenges, the high prevalence of cancer and the potential for a
cure make this an exciting area for innovation and patient-centric
approaches.
For more insights on driver and
challenges - Download a Sample Report
Segment Overview
This car t-cell therapy market report extensively covers market
segmentation by
- End-user
- 1.1 Hospitals
- 1.2 Cancer treatment centers
- Type
- 2.1 CD19
- 2.2 CD22
- 2.3 BCMA
- 2.4 Others
- Geography
- 3.1 North America
- 3.2 Europe
- 3.3 Asia
- 3.4 Rest of World (ROW)
1.1 Hospitals- The hospital segment dominates the
global CAR T-cell therapy market due to hospitals being the primary
administrators of these treatments. Hospitals provide essential
infrastructure, medical staff, and resources for CAR T-cell
therapy. Oncologists, hematologists, and nurses manage the
treatment process, ensuring patient safety and monitoring response.
Hospitals identify eligible patients, assessing medical history,
performing diagnostics, and evaluating suitability. Pre-treatment
evaluations, including imaging and lab tests, establish baselines
and monitor progress. Hospitals closely monitor patients during
therapy for adverse reactions and provide post-treatment follow-up
care. The growing number of cancer treatments in hospitals is
expected to fuel market growth.
For more information on market segmentation with
geographical analysis including forecast (2024-2028) and historic
data (2018 - 2022) - Download a Sample Report
Research Analysis
Car T-cell therapy is a revolutionary cancer treatment that
utilizes a patient's own immune system to fight cancer cells. This
innovative approach involves engineering T-cells to recognize and
attack specific cancer antigens, making it a promising solution for
various types of cancers, particularly leukemia and lymphoma. The
global market for Car T-cell therapy is expected to grow
significantly due to the increasing incidence of cancer, prevalence
of blood cancer, and advancements in healthcare infrastructure.
Reimbursement policies and public awareness are key factors
influencing the market's growth. The patient population for cancer
treatments is vast, and the precision medicine approach of Car
T-cell therapies offered by companies like Celgene Corporation and
Juno Therapeutics is tailoring treatments to individual patients,
further boosting its adoption. The future of Car T-cell therapy
holds great promise in transforming the landscape of cancer
treatment.
Market Research Overview
Car T-cell therapy is a revolutionary cancer treatment that
involves genetically modifying a patient's T-cells to recognize and
attack cancer cells. With the increasing incidence of cancer
worldwide, the demand for effective and innovative treatments is on
the rise. Reimbursement policies and public awareness are key
factors influencing the market growth. Significant investments are
being made in this field, driven by the potential for high
disposable income and a large patient population. Prevalence of
cancer, particularly leukemia and lymphoma, is a significant driver
for the market. Innovation in the field is ongoing, with the use of
CRISPR/Cas9 technology and patient-centric approaches gaining
attention. Financial backing from major corporations such as
Celgene Corporation and clinical trials for solid tumors, including
ovarian cancer, are also contributing to the market's growth.
Scalability, cost-effectiveness, and product consistency are
crucial challenges for the industry. Tailoring treatments to
individual patients through precision medicine and hematologic
malignancies are key indications for CAR T-cell therapies.
Immunotherapy, genetic modification, and systemic toxicity are
important considerations in the development of these therapies.
Quality control measures are essential to ensure safety and
efficacy. Companies like Juno Therapeutics, Tessa Therapeutics, and
Adaptimmune are leading the way in this field, with TT-02 and other
CAR T-cell therapies showing promise for the treatment of various
cancers.
Table of Contents:
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation
- End-user
-
- Hospitals
- Cancer Treatment Centers
- Type
-
- Geography
-
- North America
- Europe
- Asia
- Rest Of World (ROW)
7 Customer Landscape
8 Geographic Landscape
9 Drivers, Challenges, and Trends
10 Company Landscape
11 Company Analysis
12 Appendix
About Technavio
Technavio is a leading global technology research and advisory
company. Their research and analysis focuses on emerging market
trends and provides actionable insights to help businesses identify
market opportunities and develop effective strategies to optimize
their market positions.
With over 500 specialized analysts, Technavio's report library
consists of more than 17,000 reports and counting, covering 800
technologies, spanning across 50 countries. Their client base
consists of enterprises of all sizes, including more than 100
Fortune 500 companies. This growing client base relies on
Technavio's comprehensive coverage, extensive research, and
actionable market insights to identify opportunities in existing
and potential markets and assess their competitive positions within
changing market scenarios.
Contacts
Technavio Research
Jesse Maida
Media & Marketing Executive
US: +1 844 364 1100
UK: +44 203 893 3200
Email: media@technavio.com
Website: www.technavio.com/
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/car-t-cell-therapy-market-size-is-set-to-grow-by-usd-3-02-billion-from-2024-2028--growing-awareness-regarding-car-t-cell-therapy-to-boost-the-market-growth-technavio-302189578.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/car-t-cell-therapy-market-size-is-set-to-grow-by-usd-3-02-billion-from-2024-2028--growing-awareness-regarding-car-t-cell-therapy-to-boost-the-market-growth-technavio-302189578.html
SOURCE Technavio