
We could not find any results for:
Make sure your spelling is correct or try broadening your search.
| Share Name | Share Symbol | Market | Type |
|---|---|---|---|
| Procter and Gamble Co | NYSE:PG | NYSE | Common Stock |
| Price Change | % Change | Share Price | High Price | Low Price | Open Price | Shares Traded | Last Trade | |
|---|---|---|---|---|---|---|---|---|
| -0.9698 | -0.56% | 171.3102 | 172.21 | 168.46 | 170.30 | 8,372,538 | 00:42:37 |
CAN A DISINFECTANT BE POWERFUL AND STILL BE GENTLE AND SAFE?
June 29, 2020 -- InvestorsHub NewsWire -- via BioResearchAlert
Summary
The COVID-19 pandemic has caused demand for disinfection products to skyrocket to the point where suppliers are struggling to meet demand. The problem is compounded by the fact that bleach disinfectants are the most commonly used products in spite of CDC warnings that even "low level exposure to chlorine (which is the active ingredient in bleach) can result in nose, throat, and eye irritation. At higher levels, breathing chlorine gas may result in changes in breathing rate and coughing, and damage to the lungs. Additional symptoms of exposure to chlorine can be severe. Workers may be harmed from exposure to chlorine. The level of exposure depends upon the dose, duration, and work being done."
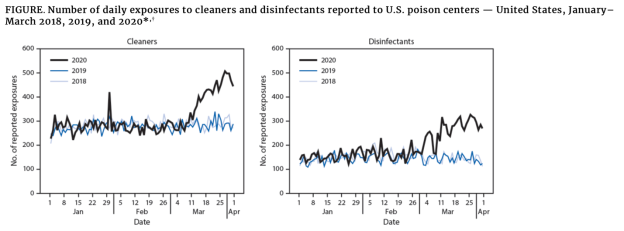
The CDC reports calls to poison centers due to cleaning and disinfectant exposures increased by 20% in early 2020. In line with toxicity claims, the CDC reports that during January - March 2020, poison centers received 45,550 exposure calls related to cleaners (28,158) and disinfectants (17,392), representing overall increases of 20.4% and 16.4% from January - March 2019 (37,822).
The FDA recently advised consumers to avoid 9 different brands of hand sanitizers because of their potential harmful effects from methanol that is wood alcohol.

"Businesses across the U.S. have begun intensive Covid-19 disinfection regimens, exposing returning workers and consumers to some chemicals that are largely untested for human health, a development that's alarming health and environmental safety experts.
The rush to disinfect is well-intentioned. Executives want to protect employees while abiding by U.S. Centers for Disease Control & Prevention guidelines (and to avoid liability). Pre-pandemic, corporate cleaning staffs typically "freshened" lobbies every three hours, sanitized restrooms every four hours and cleaned other areas at night, said Rich Feczko, national director of systems, standards and innovation at Crothall Healthcare, which cleans hundreds of hospitals.
That pace has now accelerated. "Our frequencies have ramped up in public places like lobbies and elevators to 6-8 times per day," said Feczko. Restrooms are cleaned every two hours. "Before the pandemic, clients were happy if their trash was emptied and vacuum marks were in the plush carpet," said Jill Frey, owner of Ohio-based Cummins Facility Services. Now, customers ask for sanitization (reducing pathogens on a surface) and disinfection (killing all pathogens).
"This is a hazardous proposition," said Dr. Claudia Miller, an immunologist, allergist and co-author of Chemical Exposures: Low Levels and High Stakes. "Cleaners tend to go in with hugely toxic chemicals. We're creating another problem for a whole group of people, and I'm not sure we're actually controlling infections.
Cleaning companies are selecting disinfectants from hundreds on List N, the month-old compendium of products approved by the Environmental Protection Agency to kill the novel coronavirus. Those chemicals have passed tests to show they're effective against the pathogen, but "this doesn't mean that they have been approved because they're considered safe with regard to human health," said exposure scientist Lesliam Quirós-Alcalá, an assistant professor at Johns Hopkins Bloomberg School of Public Health."
Key Investor Takeaway
What would it mean to the $16 billion disinfection industry if there was a powerful disinfectant that would inactivate viruses like SARS-CoV-2, and destroy harmful bacteria, fungi and biofilm, but that was truly safe and gentle and did not harm healthy tissue such as skin, nose, throat, eyes, lungs or mucous membranes? If it was competitively priced, marketed effectively, and well capitalized, it seems that in time, it could have the potential to become a new standard for disinfection.
BioLargo's subsidiaries and their effective, safe and gentle products are just being discovered
Like chlorine or bleach, iodine has long been known as a powerful disinfecting tool against viruses, bacteria and fungi, but its widespread adoption was limited because until now, traditional iodine products were often toxic and create problematic staining. A paper titled, "Iodine: A Forgotten Weapon Against Influenza Viruses" was published in Thyroid Science and describes how effective iodine has been against Influenza such as the great Spanish Flu Pandemic of 1918.


While it subsidiary is launching its FDA registered Clyraguard, BioLargo has also reported that it intends to pursue EPA registration on fast-track basis if available, or traditional filing if required for CupriDyne.
Leading Suppliers of Disinfectants
The EPA lists a large number of disinfectant products for use against SARS-CoV-2 and the following companies are leading suppliers:
Proctor & Gamble (PG: NYSE)
3 M Company (MMM: NYSE)
Kimberly Clark (KMB: NYSE)
Ecolab (ECL: NYSE)
Clorox Company (CLX: NYSE)
DOW, Inc. (DOW: NYSE)
Conclusion
BioResearchAlert believes BioLargo is worthy of a serious look by investors for several reasons.
SOURCE: BioResearchAlert
1 Year Procter and Gamble Chart |
1 Month Procter and Gamble Chart |

It looks like you are not logged in. Click the button below to log in and keep track of your recent history.
Support: +44 (0) 203 8794 460 | support@advfn.com
By accessing the services available at ADVFN you are agreeing to be bound by ADVFN's Terms & Conditions