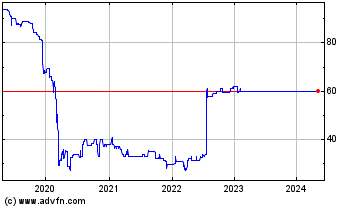
Doric Nimrod Air One (LSE:DNA)
Historical Stock Chart
From Dec 2019 to Dec 2024