
We could not find any results for:
Make sure your spelling is correct or try broadening your search.
| Share Name | Share Symbol | Market | Type |
|---|---|---|---|
| Eli Lilly and Co | NYSE:LLY | NYSE | Common Stock |
| Price Change | % Change | Share Price | High Price | Low Price | Open Price | Shares Traded | Last Trade | |
|---|---|---|---|---|---|---|---|---|
| -15.91 | -2.10% | 740.00 | 745.00 | 730.34 | 743.10 | 4,593,881 | 01:00:00 |
INDIANAPOLIS, Aug. 18, 2015 /PRNewswire/ -- Humalog® 200 units/mL KwikPen® (insulin lispro 200 units/mL; U-200), the first and only concentrated mealtime insulin analog in the U.S., is now available in pharmacies, Eli Lilly and Company (NYSE: LLY) announced today. Humalog U-200 KwikPen is a prefilled pen containing a concentrated formulation of Lilly's rapid-acting insulin Humalog® (insulin lispro 100 units/mL) to improve glycemic control in people with type 1 and type 2 diabetes. It is intended for people who are already taking mealtime insulin.
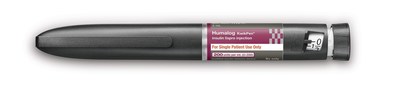
Humalog U-200 KwikPen holds twice as many units of insulin (600 units vs. 300 units) as the U-100 formulation in the same three-milliliter cartridge volume, offering patients a pen that lasts longer, which facilitates fewer pen changes every month. This may help patients who require higher daily doses of mealtime insulin fit their treatment into their lives.
"This is an important new option for many people with diabetes," said David Kendall, M.D., vice president, Medical Affairs, Lilly Diabetes. "Like everyone else, people with diabetes have busy lives. More rapid-acting insulin in a single pen may help patients worry less about running out when their plans change unexpectedly."
"For nearly 20 years, the Humalog brand has offered people who require mealtime insulin an array of options to meet their needs," Kendall said. "Lilly is pleased to offer another choice to help people with diabetes improve their glycemic control."
The most common side effect of Humalog is hypoglycemia (low blood sugar) that may be severe and cause unconsciousness, seizures and death. Humalog U-200 KwikPen delivers the same dose in half the volume of U-100 mealtime insulin pens, with no dose conversions required, and can be dialed in one-unit increments to a maximum of 60 units per injection.
As with other mealtime insulin pens, Humalog U-200 KwikPen must be used within 28 days after initial use, even if insulin remains. Unused pens should be stored in the refrigerator, but once a pen has been used for the first time, it should be kept at room temperature.
For more information on Humalog U-200 KwikPen, visit http://Humalog.com/u200.
Indication: HUMALOG is a rapid-acting human insulin analog indicated to improve glycemic control in adults and children with diabetes mellitus.
Important Safety Information for Humalog 100 units/mL and Humalog 200 units/mL KwikPen
What is the most important information I should know about Humalog?
Who should not take Humalog?
Before using Humalog, what should I tell my healthcare providers?
How should I use Humalog?
What are the possible side effects of Humalog?
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
Humalog is available by prescription only.
For additional information, talk to your healthcare providers and please click to access Full Prescribing Information and Patient Prescribing Information.
Please see Instructions for Use included with the pen.
HI U200 CON ISI 26MAY2015
About Diabetes
Approximately 29 million Americans have type 1 and type 2 diabetes.1 Type 2 diabetes is the most common type, accounting for an estimated 90 to 95 percent of all diabetes cases.1 Diabetes is a chronic disease that occurs when the body either does not properly produce or use the hormone insulin.1
About Lilly Diabetes
Lilly has been a global leader in diabetes care since 1923, when we introduced the world's first commercial insulin. Today we are building upon this heritage by working to meet the diverse needs of people with diabetes and those who care for them. Through research and collaboration, a broad and growing product portfolio and a continued determination to provide real solutions—from medicines to support programs and more—we strive to make life better for all those affected by diabetes around the world. For more information, visit www.lillydiabetes.com and follow @LillyDiabetes on Twitter.
About Eli Lilly and Company
Lilly is a global healthcare leader that unites caring with discovery to make life better for people around the world. We were founded more than a century ago by a man committed to creating high-quality medicines that meet real needs, and today we remain true to that mission in all our work. Across the globe, Lilly employees work to discover and bring life-changing medicines to those who need them, improve the understanding and management of disease, and give back to communities through philanthropy and volunteerism. To learn more about Lilly, please visit us at www.lilly.com and newsroom.lilly.com/social-channels.
P-LLY
This press release contains forward-looking statements about Humalog 200 units/mL KwikPen for use in the treatment of diabetes. It reflects Lilly's current beliefs; however, as with any such undertaking, there are substantial risks and uncertainties in the process of drug development and commercialization. There is no guarantee that future study results and patient experience will be consistent with study findings to date or that Humalog 200 units/mL KwikPen will be commercially successful. For further discussion of these and other risks and uncertainties, please see Lilly's latest Forms 10-Q and 10-K filed with the U.S. Securities and Exchange Commission. Lilly undertakes no duty to update forward-looking statements.
1 Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014.
HI97227 06/2015 ® Lilly USA, LLC 2015. All rights reserved.
Refer to: Julie Williams; williamsju@lilly.com; +1-317-627-4056


Photo - http://photos.prnewswire.com/prnh/20150817/258888
Logo - http://photos.prnewswire.com/prnh/20150817/258887LOGO
Logo - http://photos.prnewswire.com/prnh/20031219/LLYLOGO
To view the original version on PR Newswire, visit:http://www.prnewswire.com/news-releases/lillys-humalog-200-unitsml-kwikpen-now-available-in-us-pharmacies-300129849.html
SOURCE Eli Lilly and Company

Copyright 2015 PR Newswire
1 Year Eli Lilly Chart |
1 Month Eli Lilly Chart |

It looks like you are not logged in. Click the button below to log in and keep track of your recent history.
Support: +44 (0) 203 8794 460 | support@advfn.com
By accessing the services available at ADVFN you are agreeing to be bound by ADVFN's Terms & Conditions