
We could not find any results for:
Make sure your spelling is correct or try broadening your search.
| Share Name | Share Symbol | Market | Type |
|---|---|---|---|
| Ocuphire Pharma Inc | NASDAQ:OCUP | NASDAQ | Common Stock |
| Price Change | % Change | Share Price | Bid Price | Offer Price | High Price | Low Price | Open Price | Shares Traded | Last Trade | |
|---|---|---|---|---|---|---|---|---|---|---|
| 0.00 | 0.00% | 1.71 | 1.63 | 1.90 | 1.7475 | 1.64 | 1.7261 | 67,796 | 05:00:09 |
|
Annual report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
|
|
Transition report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
|
|
|
|
|
(State or other jurisdiction of
incorporation or organization)
(Address of principal executive offices) |
(I.R.S. Employer
Identification No.)
(Zip Code)
|
|
Title of each class
|
Trading Symbol
|
Name of each exchange on which registered
|
|
|
|
|
|
Large accelerated filer ☐
|
Accelerated filer ☐
|
|||
|
|
Smaller reporting company
|
|||
|
Emerging growth company
|
||||
|
PART I
|
7 | ||
|
ITEM 1.
|
7 | ||
|
ITEM 1A.
|
44 | ||
|
ITEM 1B.
|
83 | ||
|
ITEM 1C.
|
83 | ||
|
ITEM 2.
|
84
|
||
|
ITEM 3.
|
84 | ||
|
ITEM 4.
|
84 | ||
|
|
|||
|
PART II
|
|||
|
ITEM 5.
|
85 | ||
|
ITEM 6.
|
85 | ||
|
ITEM 7.
|
85 | ||
|
ITEM 7A.
|
99
|
||
|
ITEM 8.
|
100 | ||
|
ITEM 9.
|
100 | ||
|
ITEM 9A.
|
100 | ||
|
ITEM 9B.
|
101 | ||
|
ITEM 9C.
|
101 | ||
|
PART III
|
|||
|
ITEM 10.
|
102 | ||
|
ITEM 11.
|
102 | ||
|
ITEM 12.
|
102
|
||
|
ITEM 13.
|
102 | ||
|
ITEM 14.
|
102 | ||
|
|
|||
|
PART IV
|
|||
|
|
|||
|
ITEM 15.
|
103 | ||
|
ITEM 16.
|
108 | ||
|
SIGNATURES
|
|||
| • |
We depend heavily on the success of our product pipeline and we (or our current or future strategic partners). Ocuphire and/or Viatris may never complete clinical development of, receive marketing approval for,
or successfully commercialize, PS alone or as adjunctive therapy with low dose pilocarpine (LDP), APX3330, or any of our other product candidates. Moreover, if we (or our strategic partner) fail to adequately develop and commercialize APX3330
or PS, our business may be materially harmed.
|
| • |
The results of previous clinical trials may not be predictive of future results, and the results of our current and planned clinical trials may not satisfy the requirements of the FDA or non-U.S. regulatory
authorities.
|
| • |
If we experience delays or difficulties in the enrollment of patients in clinical trials, our ability to conduct and complete clinical trials, and our ability to seek and receive necessary regulatory approvals,
could be delayed or prevented.
|
| • |
Changes in regulatory requirements or FDA guidance, or unanticipated events during our clinical trials, may result in changes to clinical trial protocols or additional clinical trial requirements, which could
result in increased costs to us or delays in its development timeline.
|
| • |
We or others could discover that our product candidates lack sufficient efficacy, or sufficient efficacy compared to competitor products or that they cause undesirable side effects that were not previously
identified, which could delay or prevent regulatory approval or commercialization.
|
| • |
Even if we receive marketing approval for our product candidates in the United States, we may never receive regulatory approval to market such product candidates outside of the United States.
|
| • |
We face substantial competition and rapid technological change, which may result in others discovering, developing, or commercializing products before or more successfully than we do.
|
| • |
We do not currently have any sales or marketing infrastructure in place and, if APX3330 is approved, we may face difficulties in establishing sales and marketing capabilities or engaging third parties to sell,
market and distribute APX3330.
|
| • |
Our future commercial success depends upon attaining significant market acceptance of our product candidates, if approved, among physicians, patients, third-party payors, and others in the medical community.
|
| • |
If the FDA or a comparable foreign regulatory authority approves generic versions of our product candidates that receive marketing approval, or if such authorities do not grant our product candidates
appropriate periods of exclusivity before approving generic versions of our products, the sales of our products could be adversely affected.
|
| • |
We have not generated any revenue from sales of any products, and we expect to incur losses for the foreseeable future and may never achieve or maintain profitability.
|
| • |
Our relatively short operating history may make it difficult for investors to evaluate our business to date and to assess our future viability.
|
| • |
We will need substantial additional capital in the future. If additional capital is not available or is not available on acceptable terms (whether as a result of financial services industry changes, our
financial performance or otherwise), we will have to delay, reduce or cease operations.
|
| • |
Raising additional capital may cause dilution to our stockholders, restrict our operations, or require us to relinquish rights to our technologies or product candidates.
|
| • |
Worldwide economic and social instability or adverse global economic conditions could adversely affect our revenue, financial condition, or results of operations.
|
| • |
Our employees or our representatives may engage in misconduct or other improper activities, including violating applicable regulatory standards and requirements, which could significantly harm our business.
|
| • |
Product liability lawsuits against us, or our suppliers and manufacturers, could cause us to incur substantial liabilities and could limit commercialization of any product candidate that we may develop.
|
| • |
Federal legislation and actions by state and local governments could permit reimportation of drugs from foreign countries into the United States, which could adversely affect our operating results when the
drugs are sold at lower prices in foreign countries.
|
| • |
We rely on third parties to conduct our preclinical and clinical trials and perform other tasks for us. If these third parties do not successfully carry out their contractual duties, meet expected deadlines or
comply with regulatory requirements, we may not be able to obtain regulatory approval for or commercialize our product candidates and our business could be harmed.
|
| • |
We rely completely on third parties to supply and manufacture bulk drug substances and to formulate and package nonclinical and clinical drug supplies of our product candidates as well as to conduct analytical
testing of drug substances and products in the manufacturing processes and we intend to rely on third parties to produce and test commercial supplies of our current and any future product candidates.
|
| • |
We have entered and may enter into licensing arrangements for the development or sale of product candidates (such as the Viatris License Agreement (as defined below)) and may form or seek additional strategic
alliances or enter into licensing arrangements in the future, including for APX3330. If we are unsuccessful in forming or maintaining these alliances on favorable terms, our business could be harmed and we may have to alter development,
manufacturing and commercialization plans.
|
| • |
If we engage in acquisitions, in-licensing or strategic partnerships, this may increase our capital requirements, dilute our stockholders, cause us to incur debt or assume contingent liabilities and subject us
to other risks.
|
| • |
If we are unable to obtain and maintain sufficient patent protection for our product candidates, our competitors could develop and commercialize products or technology similar or identical to those of us, which
would adversely affect our ability to successfully commercialize any product candidates we may develop, our business, results of operations, financial condition and prospects.
|
| • |
If we do not obtain protection under the Hatch-Waxman Act and similar foreign legislation by extending the patent terms and obtaining data exclusivity for our product candidate, our business may be materially
harmed.
|
| • |
We may not be able to protect or practice our intellectual property rights throughout the world.
|
| • |
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment, and other requirements imposed by governmental agencies, and our patent
protection could be reduced or eliminated for noncompliance with these requirements.
|
| • |
We are dependent on our key personnel, and if we are not successful in attracting and retaining highly qualified personnel, we may not be able to successfully implement our business strategy.
|
| • |
A variety of risks associated with operating internationally for us and our collaborators could adversely affect our business.
|
| • |
Our business and operations would suffer in the event of system failures or unplanned events, including cyber incidents, network security breaches, service interruptions, or data corruption.
|
| • |
We currently have a substantial number of shares subject to potential issuance associated with our Equity Line of Credit arrangement. The issuance or sale of shares under our ELOC arrangement would
substantially increase the number of shares outstanding and result in dilution to our security holders. This might substantially decrease the market price of the common stock.
|
| • |
We do not anticipate paying any cash dividends in the foreseeable future.
|
| • |
If we fail to comply with the continued listing standards of the Nasdaq Capital Market, our common stock could be delisted. If it is delisted, our common stock and the liquidity of our common stock would be
impacted.
|
| • |
The market price of our common stock may fluctuate significantly.
|
| • |
We may be subject to securities litigation, which is expensive and could divert management attention.
|
| ITEM 1. |
BUSINESS
|
| • |
Advance the clinical development of our products.
|
| • |
Maintain and expand its intellectual property portfolio. Ocuphire owns an exclusive worldwide sublicense for the Ref-1 Inhibitor program, including its product candidate
APX3330, for all its ophthalmic and diabetic indications, and compositions and methods of use for Ref-1 pipeline candidates, including APX2009 and APX2014.
|
| • |
Maximize the value of APX3330 and PS. Ocuphire may seek one or more partners to develop and commercialize APX3330 both in and/or outside of the United States.
|
| • |
Evaluate in-licensing and acquisition opportunities. Ocuphire’s team is well qualified to identify and in-license or acquire clinical-stage assets and continually
evaluates opportunities to expand and diversify its pipeline.
|
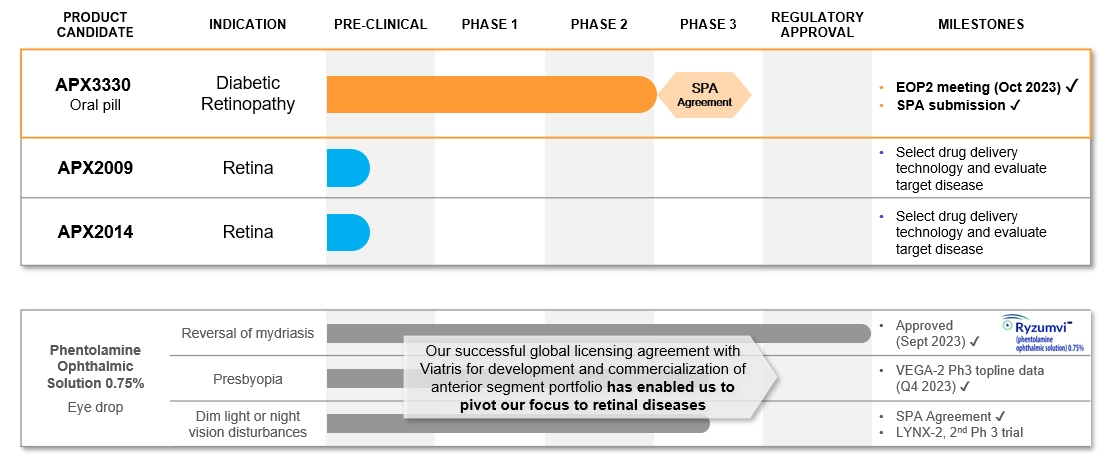
| • |
Potential to be the first oral therapy. Compared to frequent intravitreal anti-VEGF injections, associated with ocular complications, twice a day oral administration of
APX3330 could be a convenient, new preventative therapeutic option or adjunctive treatment option for large number of patients with retinal diseases, if approved.
|
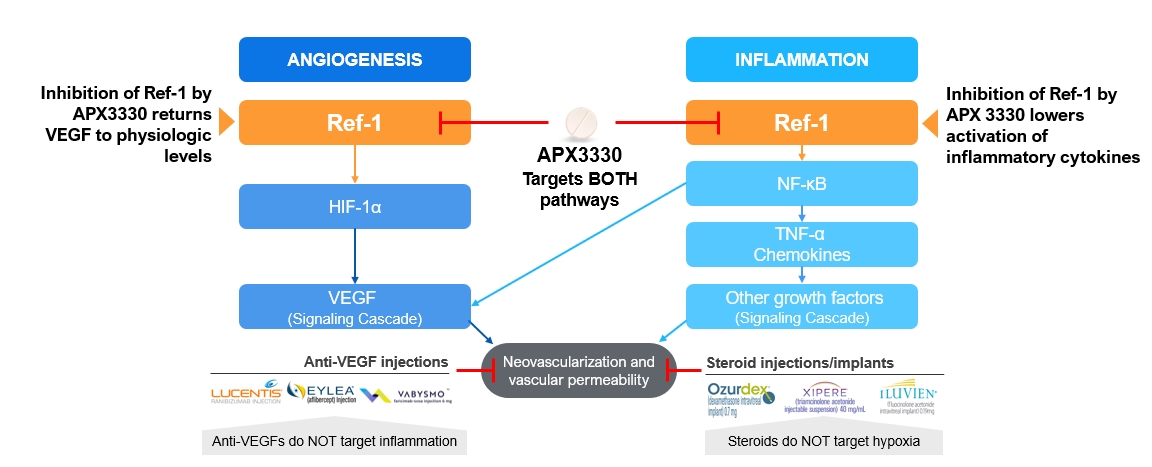
| • |
Upstream target implicated in two validated pathways. APX3330 is designed to modulate two validated cell signaling pathways (angiogenesis and inflammation) known to cause
various retinal diseases. Moreover, the APX3330 mechanism of action is distinct in working upstream of the current anti-VEGF therapies, suggesting that it could complement anti-VEGF therapies and potentially reduce frequency of doctor visits
and intravitreal injections.
|
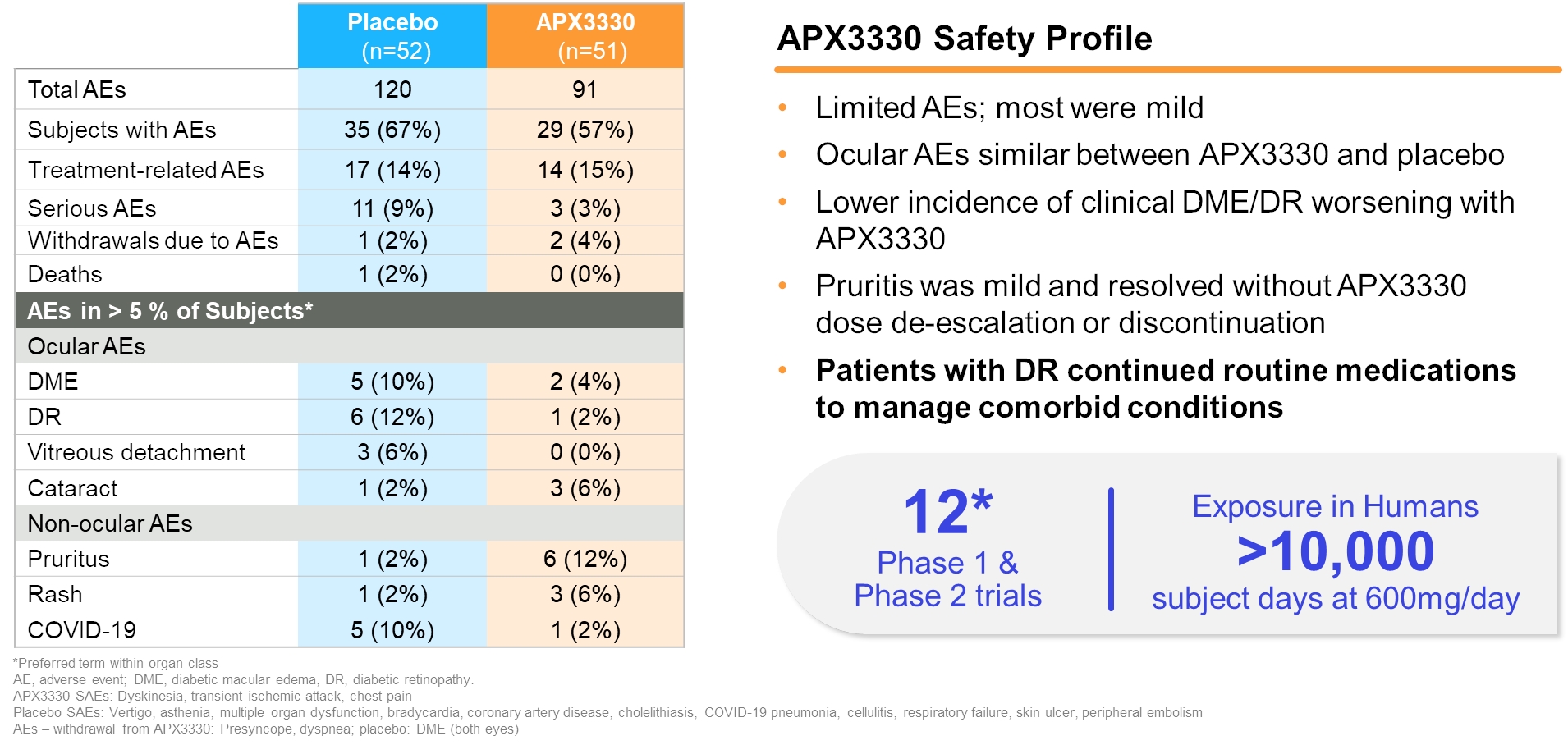
| • |
Favorable tolerability profile. In 12 completed Phase 1 and Phase 2 clinical trials, APX3330 was observed to be well-tolerated. The adverse events (“AEs”) were mostly
infrequent and mild with transient pruritis being the most common.
|
| • |
Potential benefit of systemic administration. As a systemic agent, APX3330 can be expected to treat bilateral binocular (both eyes) retinal vascular disease.
|
| • |
Oral tablet with scalable manufacturing process. APX3330 is formulated as an oral tablet with favorable stability characteristics, and its active pharmaceutical
ingredient is a small molecule with the advantages of standardized, scalable, and lower-cost manufacturing processes.
|
| • |
DR, the leading cause of vision loss in adults aged 20–74 years, which results from chronic elevations of glucose in the blood that leads to cell damage in the retina.
Retinal key opinion leaders’ feedback suggests that slowing of DR progression with an oral agent would be a useful treatment in patients with background DR and good visual function.
|
| • |
DME, one of the most common complications of DR, in which vascular leakage causes swelling of the retinal macula and a loss of visual acuity.
|
| • |
wAMD, a chronic eye disorder that causes visual distortions in the central part of one’s vision, in which abnormal blood vessels leak fluid or blood into the macula, the
part of the eye that is critical for central and color vision.
|
| • |
GA, an advanced form of age-related macular degeneration (AMD) that leads to progressive and irreversible vision loss.
|
| • |
Presbyopia, a condition in which the eye’s lens loses elasticity, affecting its ability to focus on near objects. Presbyopia typically occurs after age 40 and most patients use reading glasses in order to read
or see objects close to them. VuityTM approved in October 2021, and QLOSI™ approved in October 2023 are the only two approved eye drops for the treatment of
presbyopia.
|
| • |
Decreased vision under dim (mesopic or low) light conditions after keratorefractive surgery, a condition in which peripheral imperfections (aberrations) of the cornea scatter light when the pupil opens wide in
dim light. Patients experience glare, halos, starbursts, and decreased contrast sensitivity. decreased visual acuity under dim (mesopic) light conditions is a new indication with no approved therapies.
|
| • |
Non-proliferative DR, or NPDR. NPDR is an earlier stage of DR and can progress into more severe forms of DR
over time if untreated and if exposure to elevated blood sugar levels persists.
|
| • |
Proliferative DR, or PDR. PDR is a more advanced stage of DR than NPDR. It is characterized by retinal
neovascularization and, if left untreated, leads to permanent damage and blindness.
|
| • |
completion of preclinical laboratory tests, animal studies and formulation studies in compliance, as applicable, with the Animal Welfare Act and FDA’s good laboratory practice, or GLP, regulations;
|
| • |
submission to the FDA of an IND, which must take effect before human clinical trials may begin;
|
| • |
approval by an independent institutional review board, or IRB, representing each clinical site before each clinical trial may be initiated;
|
| • |
performance of adequate and well-controlled human clinical trials in accordance with good clinical practices, or GCP, and other applicable regulations to establish the safety and efficacy of the proposed drug
product for each proposed indication;
|
| • |
manufacturing, packaging, labelling, and distribution of drug substances and drug products consistent with the FDA’s Good Manufacturing Practice (GMP) regulations which are utilized in the GLP non-clinical and
GCP clinical studies to investigate the drug candidate;
|
| • |
development of product label, package inserts, and prescriber information that is intended to be used and included with the commercial product;
|
| • |
preparation and submission to the FDA of an NDA;
|
| • |
review of the product by an FDA advisory committee, where appropriate or if applicable;
|
| • |
satisfactory completion of one or more FDA inspections of the manufacturing facility or facilities at which the product, or components thereof, are produced to assess compliance with current Good Manufacturing
Practices, or cGMP, requirements and to assure that the facilities, methods and controls are adequate to preserve the product’s identity, strength, quality and purity;
|
| • |
satisfactory completion of FDA audits of clinical trial sites to assure compliance with GCPs and the integrity of the clinical data;
|
| • |
payment of user fees, if appropriate, and securing FDA approval of the NDA; and
|
| • |
compliance with any post-approval requirements, including Risk Evaluation and Mitigation Strategies, or REMS, and post-approval studies required by the FDA.
|
| • |
Phase 1. The drug is initially introduced into healthy human subjects or, in certain indications such as cancer, patients with the target disease or condition and tested
for safety, dosage tolerance, absorption, metabolism, distribution, excretion and, if possible, to gain an early indication of its effectiveness and to determine optimal dosage.
|
| • |
Phase 2. The drug is administered to a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the
product for specific targeted diseases and to determine dosage tolerance and optimal dosage.
|
| • |
Phase 3. The drug is administered to an expanded patient population, generally at geographically dispersed clinical trial sites, in well-controlled clinical trials to
generate enough data to statistically evaluate the efficacy and safety of the product for approval, to establish the overall risk-benefit profile of the product and to provide adequate information for the labeling of the product.
|
| • |
restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls;
|
| • |
fines, warning letters or holds on post-approval clinical trials;
|
| • |
refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product license approvals;
|
| • |
product seizure or detention, or refusal to permit the import or export of products; or
|
| • |
injunctions or the imposition of civil or criminal penalties.
|
| • |
A special, nondeductible fee on any entity that manufactures or imports specified branded prescription drugs and biologic agents, apportioned among these entities according to their market share in certain
government healthcare programs, although this fee would not apply to sales of certain products approved exclusively for orphan indications;
|
| • |
Expansion of eligibility criteria for Medicaid programs by, among other things, allowing states to offer Medicaid coverage to certain individuals with income at or below 133% of the federal poverty level,
thereby potentially increasing a manufacturer’s Medicaid rebate liability;
|
| • |
Expanded manufacturers’ rebate liability under the Medicaid Drug Rebate Program (“MDRP”) by increasing the minimum rebate for both branded and generic drugs and revising the definition of “average manufacturer
price,” or AMP, for calculating and reporting Medicaid drug rebates on outpatient prescription drug prices and extending rebate liability to prescriptions for individuals enrolled in Medicare Advantage plans; addressed a new methodology by
which rebates owed by manufacturers under the MDRP are calculated for drugs that are inhaled, infused, instilled, implanted or injected;
|
| • |
Expanded the types of entities eligible for the 340B drug discount program;
|
| • |
A new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; and
|
| • |
Established the Center for Medicare and Medicaid Innovation within the Centers of Medicare and Medicaid Services (“CMS”) to test innovative payment and service delivery models to lower Medicare and Medicaid
spending, potentially including prescription drug spending.
|
| • |
The federal Anti-Kickback Statute, which is a criminal law that prohibits, among other things, persons and entities from knowingly and willfully soliciting, offering, receiving or providing remuneration,
directly or indirectly, in cash or in kind, to induce or reward either the referral of an individual for, or the purchase, order or recommendation of, any good or service, for which payment may be made, in whole or in part, under a federal
healthcare program such as Medicare and Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. Violations of the federal Anti-Kickback
Statute can result in significant civil monetary penalties and criminal fines, as well as imprisonment and exclusion from participation in government health care programs;
|
| • |
The federal civil False Claims Act, which may be enforced through civil whistleblower or qui tam actions and imposes significant civil penalties, treble damages and potential exclusion from government health
care programs against individuals or entities for, among other things, knowingly presenting, or causing to be presented, to the federal government, claims for payment that are false or fraudulent or for making a false record or statement
material to an obligation to pay the federal government or for knowingly and improperly avoiding, decreasing or concealing an obligation to pay money to the federal government. Further, a violation of the federal Anti-Kickback Statute can
serve as a basis for liability under the federal civil False Claims Act. There is also the federal Criminal False Claims Act, which is similar to the federal Civil False Claims Act and imposes criminal liability on those that make or present
a false, fictitious or fraudulent claim to the federal government;
|
| • |
The federal Civil Monetary Penalties Law, which authorizes the imposition of substantial civil monetary penalties against an entity that engages in activities including, among others (1) knowingly presenting,
or causing to be presented, a claim for services not provided as claimed or that is otherwise false or fraudulent in any way; (2) arranging for or contracting with an individual or entity that is excluded from participation in federal health
care programs to provide items or services reimbursable by a federal health care program; (3) violations of the federal Anti-Kickback Statute; or (4) failing to report and return a known overpayment;
|
| • |
The federal payment transparency tracking and reporting requirements known as the federal Physician Payments Sunshine Act, implemented as the Open Payments Program, which requires certain manufacturers of
drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid, or the Children’s Health Insurance Program, among others, to track and report annually to CMS, within the United States Department of
Health and Human Services (“HHS”), information related to payments and other transfers of value made by that entity to US-licensed physicians (defined to include doctors, dentists, optometrists, podiatrists and chiropractors), physician
assistants, nurse practitioners, clinical nurse specialists, anesthesiologist assistants, certified registered nurse anesthetists, certified nurse midwives, and teaching hospitals, as well as ownership and investment interests held by
physicians and their immediate family members. Failure to timely, accurately, and completely submit the required information for all payments, transfers of value and ownership or investment interests may result in civil monetary penalties;
|
| • |
The federal Health Insurance Portability and Accountability Act of 1996 (“HIPAA”) which imposes criminal and civil liability for, among other things, executing or attempting to execute a scheme to defraud any
healthcare benefit program, including any third-party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing a criminal investigation of a healthcare offense, and knowingly and
willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statements or representations, or making false statements relating to healthcare benefits, items or services. Similar to
the federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it to have committed a violation
|
| • |
HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, which mandates, among other things, the adoption of uniform standards for the electronic exchange of
information in common health care transactions as well as standards relating to the privacy and security of individually identifiable health information. These standards require the adoption of administrative, physical and technical
safeguards to protect such information. In addition, many states have enacted comparable laws addressing the privacy and security of health information, some of which are more stringent than HIPAA. Failure to comply with these laws can result
in the imposition of significant civil and criminal penalties;
|
| • |
State laws that require the reporting of certain pricing information, including information pertaining to and justifying price increases, prohibit prescription drug price gouging; or impose payment caps on
certain pharmaceutical products deemed by the state to be “high cost”; and
|
| • |
Analogous state and foreign laws and regulations, such as state anti-kickback and false claims laws, may apply to sales or marketing arrangements and claims involving healthcare items or services reimbursed by
non-governmental third-party payors, including private insurers, and some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and the relevant compliance guidance
promulgated by the federal government, in addition to requiring drug manufacturers to report information related to payments to physicians and other healthcare providers or marketing expenditures.
|
| • |
Additionally, we expect our products, if and when approved, may be eligible for coverage under Medicare, the federal health care program that provides health care benefits to the aged and disabled.
Specifically, we expect our products would be primarily reimbursed under Medicare Part D, which provides an outpatient prescription drug benefit for Medicare beneficiaries. Medicare Part D is implemented through private insurance plans under
contractual arrangements between the plans and the federal government. Similar to pharmaceutical coverage through private health insurance, Part D plans develop formularies, impose utilization controls (such as prior authorization, step
therapy, and quantity limits), and negotiate discounts from drug manufacturers. Because of this, the list of prescription drugs covered by Part D plans varies by plan. However, with limited exceptions, individual plans are required by statute
to cover certain therapeutic categories and classes of drugs or biologics and to have at least two drugs in each unique therapeutic category or class. Our products may also be covered and reimbursed under other government programs, including
those discussed below:
|
| • |
The MDRP requires pharmaceutical manufacturers to enter into and have in effect a national rebate agreement with the Secretary of the Department of Health and Human Services as a condition for states to receive
federal matching funds for the manufacturer’s outpatient drugs furnished to Medicaid patients. Under the MDRP, manufacturers must pay a rebate to each state Medicaid program for quantities of products utilized on an outpatient basis (with
some exceptions) that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program. MDRP rebates are calculated using a statutory formula, state-reported utilization data, and pricing data that are calculated and reported
by manufacturers on a monthly and quarterly basis to CMS. These data include the AMP and, in the case of single source and innovator multiple source products, the best price for each drug.
|
| • |
The 340B drug pricing program requires participating manufacturers to agree to charge statutorily-defined covered entities no more than the 340B “ceiling price” for the manufacturer’s covered outpatient drugs.
These 340B covered entities include health care organizations that have certain federal designations or receive funding from specific federal programs, including Federally Qualified Health Centers, Ryan White HIV/AIDS Program grantees, and
certain types of hospitals and specialized clinics, as well as certain hospitals that serve a disproportionate share of low-income patients. The ACA expanded the 340B program to also include certain children’s hospitals, certain free-standing
cancer hospitals, critical access hospitals, certain rural referral centers and certain sole community hospitals, each as defined by ACA. The 340B ceiling price is calculated using a statutory formula, which is based on the AMP and rebate
amount for the covered outpatient drug as calculated under the MDRP, and in general, products subject to the MDRP are also subject to the 340B ceiling price calculation and discount requirement. Any changes to the definition of Medicaid AMP
and the Medicaid rebate amount also could affect our 340B ceiling price calculation for our products and could negatively impact our results of operations.
|
| • |
Federal law further requires that for a company to be eligible to have its products paid for with federal funds under the MDRP and Medicare Part B programs, as well as to be purchased by certain federal
agencies and grantees, it also must participate in the Department of Veterans Affairs (“VA”) Federal Supply Schedule (“FSS”) pricing program. To participate, manufacturers are required to enter into an FSS contract and other agreements with
the VA for any covered drugs. Under these agreements, manufacturers must make such products available to the “Big Four” federal agencies—the VA, the Department of Defense (“DoD”), the Public Health Service (including the Indian Health
Service), and the Coast Guard—at pricing that is capped pursuant to a statutory federal ceiling price (“FCP”), formula set forth in Section 603 of the Veterans Health Care Act of 1992 (“VHCA”). The FCP is based on a weighted average
non-federal average manufacturer price (“Non-FAMP”), which manufacturers are required to report on a quarterly and annual basis to the VA.
|
| • |
Any failure to comply with price reporting and rebate payment obligations under federal healthcare programs could negatively impact our financial results. Civil monetary penalties can be applied if we are found
to have knowingly submitted any false price information to the government, if we are found to have made a misrepresentation in the reporting of our average sales price, or if we fail to submit the required price data on a timely basis. Such
conduct also could provide a basis for other potential liability under other federal laws such as the False Claims Act.
|
| ITEM 1A. |
RISK FACTORS
|
| • |
delays in the launch or difficulties in the widespread commercialization of RYZUMVI (currently the launch is anticipated in the first half of 2024);
|
| • |
delays in, termination, or numerous unforeseen events during, or as a result of, manufacturing or clinical trials;
|
| • |
obtaining unfavorable results from nonclinical and clinical studies for our product candidates;
|
| • |
the cost of clinical trials being greater than anticipated;
|
| • |
the willingness of patients or medical investigators to follow our clinical trial protocols and the number of patients willing to participate;
|
| • |
delays in applying for and receiving marketing and NDA approvals from applicable regulatory authorities for our product candidates;
|
| • |
other government or regulatory delays and changes in regulatory requirements, policy and guidelines may require us to perform additional clinical trials or use substantial additional resources to obtain
regulatory approval;
|
| • |
issues with making arrangements with third-party manufacturers for commercial quantities of RYZUMVI and our product candidates and receiving regulatory approval of our manufacturing processes and our
third-party manufacturers’ facilities from applicable regulatory authorities;
|
| • |
establishing sales, marketing, and distribution capabilities and launching commercial sales of RYZUMVI and our product candidates, if and when approved, whether alone or in collaboration with others;
|
| • |
acceptance of RYZUMVI and our product candidates by patients, the medical community, and third-party payors;
|
| • |
effectively competing with other therapies, including the existing standard-of-care;
|
| • |
maintaining a continued acceptable safety profile of RYZUMVI and our product candidates following approval;
|
| • |
obtaining and maintaining coverage and adequate reimbursement from third-party payors;
|
| • |
obtaining and maintaining patent and trade secret protection and regulatory exclusivity;
|
| • |
protecting our rights in our intellectual property portfolio related to RYZUMVI and our product candidates; and
|
| • |
our ability to fulfill requests for additional data regarding our product candidates.
|
| • |
In addition, under the Apexian License Agreement, Ocuphire has rights to certain compounds for use in ophthalmic and diabetic diseases. Ocuphire does not control the development of these compounds in other
non-ophthalmic indications.
|
| • |
Viatris may not be able to manufacture our products in a timely or cost-effective manner;
|
| • |
Viatris may not timely perform its obligations under the Viatris License Agreement;
|
| • |
Viatris may fail to effectively commercialize our products;
|
| • |
Viatris may not be able to sublicense RYZUMVI or PS to one or more suitable parties outside the United States; or
|
| • |
Contractual disputes or other disagreements between us and Viatris, including those regarding the development, manufacture, sub licensure and commercialization of our products, interpretation of the License
Agreement, and ownership of proprietary rights. Viatris may select a new development partner for RYZUMVI and PS in the U.S. upon 90 days’ notice to Ocuphire.
|
| • |
severity of the disease under investigation;
|
| • |
availability and efficacy of medications already approved for the disease under investigation;
|
| • |
eligibility criteria and visit schedule for the trial in question;
|
| • |
competition for eligible patients with other companies conducting clinical trials for product candidates seeking to treat the same indication or patient population;
|
| • |
our payments for conducting clinical trials;
|
| • |
perceived risks and benefits of the product candidate under study;
|
| • |
efforts to facilitate timely enrollment in clinical trials;
|
| • |
patient referral practices of physicians;
|
| • |
the ability to monitor patients adequately during and after treatment;
|
| • |
proximity and availability of clinical trial sites for prospective patients; and
|
| • |
the ability of patients to participate in clinical trials during any public health emergencies.
|
| • |
discover that they are less effective, or identify undesirable side effects caused by our product candidates:
|
| • |
regulatory authorities may withdraw their approval of the product;
|
| • |
we may be required to recall the product, change the way this product is administered, conduct additional clinical trials, or change the labeling or distribution of the product (including REMS);
|
| • |
additional restrictions may be imposed on the marketing of, or the manufacturing processes for, the product;
|
| • |
we may be subject to fines, injunctions, or the imposition of civil or criminal penalties;
|
| • |
we could be sued and held liable for harm caused to patients;
|
| • |
the product may be rendered less competitive and sales may decrease; or
|
| • |
our reputation may suffer generally among both clinicians and patients.
|
| • |
the successful launch and widespread commercialization of RYZUMVI;
|
| • |
obtain favorable results from and complete the nonclinical and clinical development of our product candidates for their planned indications, including successful completion of additional clinical trials for
these indications;
|
| • |
submit applications to regulatory authorities for both product candidates and receive timely marketing approvals in the United States and foreign countries;
|
| • |
establish and maintain commercially viable supply and manufacturing relationships with third parties that can provide adequate, in both amount and quality, products and services to support clinical development
and meet the market demand for RYZUMVI and our product candidates that we develop, if approved;
|
| • |
establish sales and marketing capabilities to effectively market and sell our product candidates in the United States or other markets, either alone or with a pharmaceutical partner;
|
| • |
address any competing products and technological and market developments;
|
| • |
obtain coverage and adequate reimbursement for customers and patients from government and third-party payors for RYZUMVI and our product candidates that we develop; and
|
| • |
achieve market acceptance of RYZUMVI and our product candidates.
|
| • |
Delayed access to deposits or other financial assets or the uninsured loss of deposits or other financial assets;
|
| • |
Loss of access to revolving existing credit facilities or other working capital sources and/or the inability to refund, roll over or extend the maturity of, or enter into new credit facilities or other working
capital resources;
|
| • |
Potential or actual breach of contractual obligations that require us to maintain letters or credit or other credit support arrangements; or
|
| • |
Termination of cash management arrangements and/or delays in accessing or actual loss of funds subject to cash management arrangements.
|
| • |
the scope, size, rate of progress, results, and costs of researching and developing our product candidates, and initiating and completing our nonclinical studies and clinical trials;
|
| • |
the cost, timing and outcome of our efforts to obtain further marketing approval for our product candidates in the United States and other countries, including to fund the preparation and filing of NDAs with
the FDA for our product candidates and to satisfy related FDA requirements and regulatory requirements in other countries;
|
| • |
the number and characteristics of any additional product candidates we develop or acquire, if any;
|
| • |
our ability to establish and maintain collaborations on favorable terms, if at all;
|
| • |
the amount of revenue, if any, from commercial sales, should our product candidates receive marketing approval;
|
| • |
the costs associated with commercializing our product candidates, if we receive marketing approval, including the cost and timing of developing sales and marketing capabilities or entering into strategic
collaborations to market and sell our product candidates;
|
| • |
the cost of manufacturing our product candidates or products we successfully commercialize; and
|
| • |
the costs associated with general corporate activities, such as the cost of filing, prosecuting and enforcing patent claims and making regulatory filings.
|
| • |
litigation involving patients taking our drugs;
|
| • |
restrictions on such drugs, manufacturers, or manufacturing processes;
|
| • |
restrictions on the labeling or marketing of a drug;
|
| • |
restrictions on drug distribution or use;
|
| • |
requirements to conduct post-marketing studies or clinical trials;
|
| • |
warning letters or untitled letters;
|
| • |
withdrawal of the drugs from the market;
|
| • |
refusal to approve pending applications or supplements to approved applications that we submit;
|
| • |
product recall or public notification or medical product safety alerts to healthcare professionals;
|
| • |
fines, restitution, or disgorgement of profits or revenues;
|
| • |
suspension or withdrawal of marketing approvals;
|
| • |
damage to relationships with any potential collaborators;
|
| • |
unfavorable press coverage and damage to our reputation;
|
| • |
refusal to permit the import or export of drugs;
|
| • |
product seizure; or
|
| • |
injunctions or the imposition of civil or criminal penalties.
|
| • |
comply with the regulations of the FDA and applicable non-U.S. regulators;
|
| • |
provide accurate information to the FDA and applicable non-U.S. regulators;
|
| • |
comply with healthcare fraud and abuse laws and regulations in the United States and abroad;
|
| • |
report financial information or data accurately; or
|
| • |
disclose unauthorized activities to us.
|
| • |
the inability to recruit and retain adequate numbers of effective sales and marketing personnel or enter into distribution agreements with third parties;
|
| • |
the inability of sales personnel to obtain access to physicians or educate an adequate number of physicians as to the benefits of our products;
|
| • |
the lack of complementary products to be offered by sales personnel, which may put us at a competitive disadvantage relative to companies with more extensive product lines;
|
| • |
unforeseen costs and expenses associated with creating an independent sales and marketing organization; and
|
| • |
the inability to obtain sufficient coverage and reimbursement from third-party payors and governmental agencies.
|
| • |
If we enter into arrangements with third parties to perform sales, marketing, and distribution services, our product revenues or the profitability of these product revenues to us are likely
to be lower than if we were to market and sell a product that we developed ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell and market any product candidate or may be unable to do so on
terms that are favorable to us. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market a drug effectively. If we do not establish sales and
marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
|
| • |
efficacy and potential advantages compared to alternative treatments;
|
| • |
the ability to offer our product for sale at competitive prices;
|
| • |
the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies;
|
| • |
any restrictions on the use of our product together with other medications;
|
| • |
interactions of our product with other medicines patients are taking;
|
| • |
inability of certain types of patients to take our product;
|
| • |
demonstrated ability to treat patients and, if required by any applicable regulatory authority in connection with the approval for target indications as compared with other available therapies;
|
| • |
the relative convenience and ease of administration as compared with other treatments available for approved indications;
|
| • |
the prevalence and severity of any adverse side effects;
|
| • |
limitations or warnings contained in the labeling approved by the FDA;
|
| • |
availability of alternative treatments already approved or expected to be commercially launched in the near future;
|
| • |
the effectiveness of our sales and marketing strategies;
|
| • |
our ability to increase awareness through marketing efforts;
|
| • |
guidelines and recommendations of organizations involved in research, treatment and prevention of various diseases that may advocate for alternative therapies;
|
| • |
our ability to obtain sufficient third-party coverage and adequate reimbursement;
|
| • |
the willingness of patients to pay out-of-pocket in the absence of third-party coverage; and
|
| • |
physicians or patients may be reluctant to switch from existing therapies even if potentially more effective, safe or convenient.
|
| • |
decreased demand for any product candidate that we are developing;
|
| • |
injury to our reputation and significant negative media attention;
|
| • |
withdrawal of clinical trial participants;
|
| • |
increased FDA warnings on product labels;
|
| • |
significant costs to defend the related litigation;
|
| • |
substantial monetary awards to trial participants or patients;
|
| • |
distraction of management’s attention from our primary business;
|
| • |
loss of revenue;
|
| • |
the inability to commercialize any product candidate that we may develop;
|
| • |
the initiation of investigations by regulators; and
|
| • |
the inability to take advantage of limitations on product liability lawsuits that apply to generic drug products, which could increase our exposure to liability for products deemed to be dangerous or defective.
|
| • |
fail to comply with contractual obligations;
|
| • |
experience regulatory compliance issues;
|
| • |
undergo changes in ownership or management;
|
| • |
undergo changes in priorities or become financially distressed; or
|
| • |
form relationships with other entities, some of which may be our competitors.
|
| • |
collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations;
|
| • |
collaborators may not perform their obligations as expected;
|
| • |
collaborators may not pursue development and commercialization or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator’s
strategic focus or available funding, or external factors such as an acquisition that diverts resources or creates competing priorities;
|
| • |
collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials, or require a new
formulation of a product candidate for clinical testing;
|
| • |
collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidate if the collaborators believe that competitive products are more
likely to be successfully developed or can be commercialized under terms that are more attractive than ours;
|
| • |
a collaborator with marketing and distribution rights to one or more product candidates may not commit sufficient resources to the marketing or distribution of any such product candidate;
|
| • |
collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our
proprietary information or expose us to litigation;
|
| • |
collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability;
|
| • |
disputes may arise between us and collaborators that result in the delay or termination of research, development, or commercialization of our product candidates, or in litigation or arbitration that diverts
management attention and resources;
|
| • |
we may lose certain valuable rights under circumstances identified in our collaborations, including if we undergo a change of control;
|
| • |
collaborations may be terminated and such terminations may create a need for additional capital to pursue further development or commercialization of the applicable product candidates;
|
| • |
collaborators may learn about our discoveries and use this knowledge to compete with us in the future;
|
| • |
the results of collaborators’ nonclinical or clinical studies could harm or impair other development programs;
|
| • |
there may be conflicts between different collaborators that could negatively affect those collaborations and potentially others;
|
| • |
the number and nature of our collaborations could adversely affect our attractiveness to potential future collaborators or acquirers;
|
| • |
collaboration agreements may not lead to development or commercialization of our product candidate in the most efficient manner or at all. If a present or future collaborator of us were to be involved in a
business combination, the continued pursuit and emphasis on our product development or commercialization program under such collaboration could be delayed, diminished, or terminated; and
|
| • |
collaborators may be unable to obtain the necessary marketing approvals.
|
| • |
increased operating expenses and cash requirements;
|
| • |
the assumption of indebtedness or contingent liabilities;
|
| • |
the issuance of our equity securities which would result in dilution to our stockholders;
|
| • |
assimilation of operations, intellectual property, products and product candidates of an acquired company, including difficulties associated with integrating new personnel;
|
| • |
the diversion of management’s attention from our existing product candidates and initiatives in pursuing such an acquisition or strategic partnership;
|
| • |
retention of key employees, the loss of key personnel, and uncertainties in our ability to maintain key business relationships;
|
| • |
risks and uncertainties associated with the other party to such a transaction, including the prospects of that party and their existing products or product candidates and regulatory approvals; and
|
| • |
our inability to generate revenue from acquired intellectual property, technology and/or products sufficient to meet our objectives or even to offset the associated transaction and maintenance costs.
|
| • |
any of our patents, or any of our pending patent applications, if issued, will include claims having a scope sufficient to protect our product candidates;
|
| • |
any of our pending patent applications will result in issued patents;
|
| • |
we will be able to successfully commercialize our product candidates, if approved, before our relevant patents expire;
|
| • |
we were the first to make the inventions covered by each of our patents and pending patent applications;
|
| • |
we were the first to file patent applications for these inventions;
|
| • |
others will not develop similar or alternative technologies that do not infringe our patents;
|
| • |
any of our patents will be valid and enforceable;
|
| • |
any patents issued to us will provide a basis for an exclusive market for our commercially viable products, will provide us with any competitive advantages or will not be challenged by third parties;
|
| • |
we will develop additional proprietary technologies or product candidates that are separately patentable; or
|
| • |
our commercial activities or products will not infringe upon the patents of others.
|
| • |
the scope of rights granted under the license agreement and other interpretation-related issues;
|
| • |
the extent to which our product candidates, technology and processes infringe on intellectual property of the licensor that is not subject to the licensing agreement;
|
| • |
the sublicensing of patent and other rights under our collaborative development relationships;
|
| • |
our diligence obligations under the license agreement and what activities satisfy those diligence obligations;
|
| • |
the inventorship and ownership of inventions and know-how resulting from the joint creation or use of intellectual property; and
|
| • |
the priority of invention of patented technology.
|
| • |
compliance with differing or unexpected regulatory requirements for our product candidates;
|
| • |
different medical practices and customs affecting acceptance of our product candidates, if approved, or any other approved product in the marketplace;
|
| • |
language barriers;
|
| • |
the interpretation of contractual provisions governed by foreign law in the event of a contract dispute;
|
| • |
difficulties in staffing and managing foreign operations, and an inability to control commercial or other activities where it is relying on third parties;
|
| • |
workforce uncertainty in countries where labor unrest is more common than in the United States;
|
| • |
potential liability under the Foreign Corrupt Practice Act of 1977 or comparable foreign regulations;
|
| • |
production shortages resulting from any events affecting raw material supply or manufacturing capability abroad;
|
| • |
foreign government taxes, regulations, and permit requirements;
|
| • |
U.S. and foreign government tariffs, trade restrictions, price and exchange controls, and other regulatory requirements;
|
| • |
economic weakness, including inflation, natural disasters, war, events of terrorism, or political instability in particular foreign countries;
|
| • |
fluctuations in currency exchange rates, which could result in increased operating expenses and reduced revenues;
|
| • |
compliance with tax, employment, immigration, and labor laws, regulations, and restrictions for employees living or traveling abroad;
|
| • |
changes in diplomatic and trade relationships; and
|
| • |
challenges in enforcing our contractual and intellectual property rights, especially in those foreign countries that do not respect and protect intellectual property rights to the same extent as the United States.
|
| • |
In connection with our equity line of credit, or ELOC, arrangement, we issued Lincoln Park Capital Fund, LLC 246,792 shares of our common stock.
|
| • |
Under our ELOC arrangement, we can sell up to $50,000,000 worth of our Common Stock over the thirty-six month term of the ELOC arrangement, to Lincoln Park Capital, LLC, beginning only after certain conditions
set forth in the Purchase Agreement have been satisfied.
|
| • |
the announcement of new products or product enhancements by us or our competitors;
|
| • |
changes in our relationships with our licensors or other strategic partners;
|
| • |
developments concerning intellectual property rights and regulatory approvals;
|
| • |
variations in ours and our competitors’ results of operations;
|
| • |
substantial sales of shares of our common stock due to the release of lock-up agreements;
|
| • |
the announcement of clinical trial results;
|
| • |
the announcement of potentially dilutive financings;
|
| • |
changes in earnings estimates or recommendations by securities analysts;
|
| • |
changes in the structure of healthcare payment systems;
|
| • |
developments and market conditions in the pharmaceutical and biotechnology industries, including due to the COVID-19 pandemic; and
|
| • |
the results of clinical trials of APX3330, PS, or any other product candidate that we may develop.
|
| ITEM 1B. |
UNRESOLVED STAFF COMMENTS
|
| ITEM 1C. |
CYBERSECURITY
|
| ITEM 2. |
PROPERTIES
|
| ITEM 3. |
LEGAL PROCEEDINGS
|
| ITEM 4. |
MINE SAFETY DISCLOSURES
|
| ITEM 5. |
MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES
|
| ITEM 6. |
[RESERVED]
|
| • |
continue clinical trials for APX3330, PS and for any other product candidate in our future pipeline;
|
| • |
continue nonclinical studies for APX3330, APX2009 and APX2014, PS and for any other product candidate in our future pipeline;
|
| • |
develop additional product candidates that we identify, in-license or acquire;
|
| • |
seek regulatory approvals for any product candidates that successfully complete clinical trials;
|
| • |
contract to manufacture our product candidates;
|
| • |
maintain, expand and protect our intellectual property portfolio;
|
| • |
hire additional staff, including clinical, scientific, operational and financial personnel, to execute our business plan;
|
| • |
add operational, financial and management information systems and personnel to support our product development and potential future commercialization efforts;
|
| • |
continue to operate as a public company; and
|
| • |
establish on our own or with partners, a sales, marketing and distribution infrastructure to commercialize any products for which we may obtain regulatory approval.
|
|
For the Year Ended
December 31,
|
||||||||||||
|
2023
|
2022
|
Change
|
||||||||||
|
License and collaborations revenue
|
$
|
19,049
|
$
|
39,850
|
$
|
(20,801
|
)
|
|||||
|
Operating expenses:
|
||||||||||||
|
General and administrative
|
11,959
|
7,269
|
4,690
|
|||||||||
|
Research and development
|
17,653
|
14,355
|
3,298
|
|||||||||
|
Total operating expenses
|
29,612
|
21,624
|
7,988
|
|||||||||
|
(Loss) income from operations
|
(10,563
|
)
|
18,226
|
(28,789
|
)
|
|||||||
|
Financing costs
|
(1,328
|
)
|
—
|
(1,328
|
)
|
|||||||
|
Interest expense
|
—
|
(9
|
)
|
9
|
||||||||
|
Fair value change in derivative liabilities
|
80
|
—
|
80
|
|||||||||
|
Other income (expense), net
|
1,837
|
(14
|
)
|
1,851
|
||||||||
|
(Loss) income before income taxes
|
(9,974
|
)
|
18,203
|
(28,177
|
)
|
|||||||
|
Provision for income taxes
|
(12
|
)
|
(315
|
)
|
303
|
|||||||
|
Net (loss) income
|
$
|
(9,986
|
)
|
$
|
17,888
|
$
|
(27,874
|
)
|
||||
|
For the Year Ended
|
||||||||||||
|
December 31,
|
||||||||||||
|
2023
|
2022
|
Change
|
||||||||||
|
External costs:
|
||||||||||||
|
Phentolamine Ophthalmic Solution 0.75% (“PS”)
|
$
|
9,983
|
$
|
8,962
|
$
|
1,021
|
||||||
|
APX 3330
|
4,818
|
3,047
|
1,771
|
|||||||||
|
Unallocated
|
678
|
647
|
31
|
|||||||||
|
Total external cost
|
15,479
|
12,656
|
2,823
|
|||||||||
|
Internal costs:
|
||||||||||||
|
Employee related expenses
|
2,148
|
1,637
|
511
|
|||||||||
|
Facilities, supplies and other
|
26
|
62
|
(36
|
)
|
||||||||
|
Total internal costs
|
2,174
|
1,699
|
475
|
|||||||||
|
Total research and development expenses
|
$
|
17,653
|
$
|
14,355
|
$
|
3,298
|
||||||
|
For the Year Ended
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Net cash (used in) provided by operating activities
|
$
|
(1,112
|
)
|
$
|
14,314
|
|||
|
Net cash used in investing activities
|
—
|
—
|
||||||
|
Net cash provided by financing activities
|
8,979
|
3,786
|
||||||
|
Net increase in cash and cash equivalents
|
$
|
7,867
|
$
|
18,100
|
||||
| ITEM 7A. |
QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
|
| ITEM 8. |
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
|
| ITEM 9. |
CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
|
| ITEM 9A. |
CONTROLS AND PROCEDURES
|
| ITEM 9C. |
DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
|
| ITEM 10. |
DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
|
| ITEM 11. |
EXECUTIVE COMPENSATION
|
| ITEM 12. |
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
|
| ITEM 13. |
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE
|
| ITEM 14. |
PRINCIPAL ACCOUNTANT FEES AND SERVICES
|
| ITEM 15. |
EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
|
| (a) |
Financial Statements: The financial statements filed as part of this report are listed in Part II, Item 8.
|
| (b) |
Financial Statement Schedules: The schedules are either not applicable or the required information is presented in the financial statements or notes thereto.
|
| (c) |
Exhibits: The following exhibits are incorporated by reference or filed as part of this Annual Report on Form 10-K:
|
|
EXHIBIT
NUMBER
|
DESCRIPTION OF DOCUMENT
|
|
Amended and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Appendix G to the Registrant’s Definitive Proxy Statement on Schedule 14A, filed on April 29, 2005).
|
|
|
Certificate of Amendment of Amended and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K, filed on May 5,
2017).
|
|
|
Certificate of Amendment of Amended and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K, filed on August 30,
2018).
|
|
|
Certificate of Amendment of Amended and Restated Certificate of Incorporation of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K, filed on April 12,
2019).
|
|
|
Certificate of Amendment of Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K, filed on November 6, 2020).
|
|
|
Certificate of Amendment of Amended and Restated Certificate of Incorporation (incorporated by reference to Exhibit 3.2 to the Registrant’s Current Report on Form 8-K, filed on November 6, 2020).
|
|
|
Second Amended and Restated Bylaws of the Registrant (incorporated by reference to Exhibit 3.3 to the Registrant’s Current Report on Form 8-K, filed on November 6, 2020).
|
|
|
First Amendment to Second Amended and Restated Bylaws of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed on June 10, 2022).
|
|
|
Second Amendment to Second Amended and Restated Bylaws of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed on June 17, 2022).
|
|
|
Third Amendment to Second Amended and Restated Bylaws of the Registrant (incorporated by reference to Exhibit 3.1 to the Registrant’s Current Report on Form 8-K filed on June 2, 2023).
|
|
Form of Common Stock Purchase Warrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K, filed on October 13, 2017).
|
|
|
Form of Common Stock Purchase Warrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K, filed on October 19, 2018).
|
|
|
Form of Common Stock Purchase Warrant (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K, filed on January 25, 2019).
|
|
|
Form of Series A/B Warrants (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K, filed on July 1, 2020).
|
|
|
Description of Securities (incorporated by reference to Exhibit 4.11 to the Registrant’s Annual Report on Form 10-K, filed on March 11, 2021).
|
|
|
Form of Warrant to purchase shares of common stock (incorporated by reference to Exhibit 4.1 to the Registrant’s Current Report on Form 8-K/A, filed on June 7, 2021).
|
|
|
Form of Indenture (incorporated by reference to Exhibit 4.13 to the Registrant’s Registration Statement on Form S-3, filed on January 10, 2024).
|
|
|
Form of Common Stock Warrant Agreement and Warrant Certificate (incorporated by reference to Exhibit 4.15 to the Registrant’s Registration Statement on Form S-3, filed on January 10,
2024).
|
|
|
Form of Preferred Stock Warrant Agreement and Warrant Certificate (incorporated by reference to Exhibit 4.16 to the Registrant’s Registration Statement on Form S-3, filed on January 10,
2024).
|
|
|
Form of Debt Securities Warrant Agreement and Warrant Certificate (incorporated by reference to Exhibit 4.17 to the Registrant’s Registration Statement on Form S-3, filed on January 10,
2024).
|
|
|
Amended and Restated Employment Agreement by and among the Company and Mina Sooch, effective as of November 5, 2020 (incorporated by reference to Exhibit 10.27 to the Registrant’s
Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
First Amendment to the Amended and Restated Employment Agreement by and among
the Company and Mina Sooch, effective as of March 26, 2023 (incorporated by reference to Exhibit 10.1.1 to the Registrant’s Annual Report on Form
10-K , filed on March 30, 2023).
|
|
| 10.1.2* |
Separation and Release Agreement, dated as of June 8, 2023, by and between Ocuphire Pharma, Inc. and Mina Sooch (incorporated by reference to Exhibit 10.3 to
the Registrant’s Quarterly Report on Form 10-Q, filed on August 11, 2023).
|
|
Amended and Restated Employment Agreement by and among the Company and Bernhard Hoffmann, effective as of November 5, 2020 (incorporated by reference to Exhibit 10.29 to the
Registrant’s Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
First Amendment to the Amended and Restated Employment Agreement by and among the Company and Bernhard Hoffmann, effective as of March 26,
2023 (incorporated by reference to Exhibit 10.2.1 to the Registrant’s Annual Report on Form 10-K , filed on March 30, 2023).
|
|
Form of Indemnification Agreement (incorporated by reference to Exhibit 10.30 to the Registrant’s Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
Sublicense Agreement, dated as of January 21, 2020, by and between Ocuphire Pharma, Inc. and Apexian Pharmaceuticals, Inc (incorporated by reference to Exhibit 10.31 to the Registrant’s
Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
First Amendment to Sublicense Agreement, dated as of June 4, 2020, by and between Apexian Pharmaceuticals, Inc. and Ocuphire Pharma, Inc (incorporated by reference to Exhibit 10.32 to
the Registrant’s Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
Lease Agreement, dated as of May 19, 2019, by and between Ocuphire Pharma, Inc. and Duke & Duke, LP (incorporated by reference to Exhibit 10.33 to the Registrant’s Registration
Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
First Amendment to Lease Agreement, dated as of October 29, 2019, by and between Ocuphire Pharma, Inc. and Duke & Duke, LP (incorporated by reference to Exhibit 10.34 to the
Registrant’s Registration Statement on Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
Second Lease Amendment, dated as of November 17, 2020, by and between the Company and Duke & Duke (incorporated by reference to Exhibit 10.43 to the Registrant’s Annual Report on
Form 10-K, filed on March 11, 2021).
|
|
|
Third Lease Amendment, dated as of September 9, 2021, by and between the Company and Duke & Duke (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on
Form 10-Q, filed on November 12, 2021).
|
|
|
Fourth Lease Amendment, dated as of October 17, 2022, by and between the Company and Duke & Duke (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on
Form 10-Q, filed on November 4, 2022).
|
|
|
Fifth Lease Amendment, dated as of November 29, 2023, by and between the Company and Duke & Duke
|
|
|
Ocuphire Pharma, Inc. 2018 Equity Incentive Plan, dated as of April 9, 2018 (incorporated by reference to Exhibit 10.35 to the Registrant’s Registration Statement on Form S-4 (File No.
333-239702), filed on September 30, 2020).
|
|
|
First Amendment to 2018 Equity Incentive Plan, dated as of December 23, 2019 (incorporated by reference to Exhibit 10.36 to the Registrant’s Registration Statement on Form S-4 (File No.
333-239702), filed on September 30, 2020).
|
|
|
Form of Option Agreement issuable under the Ocuphire Pharma, Inc. 2018 Equity Incentive Plan (incorporated by reference to Exhibit 10.37 to the Registrant’s Registration Statement on
Form S-4 (File No. 333-239702), filed on September 30, 2020).
|
|
|
Ocuphire Pharma, Inc. 2020 Equity Incentive Plan (incorporated by reference to Annex D to the Registrant’s Registration Statement on Form S-4 (File No.
333-239702), filed on September 30, 2020).
|
|
|
Form of Restricted Stock Unit Grant Notice issued under the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan (incorporated by reference to
Exhibit 10.7.1 to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
|
Form of Stock Option Grant Notice issued under the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan (incorporated by reference to Exhibit
10.7.2 to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
Contingent Value Rights Agreement, dated as of November 5, 2020, by and among the Company, Shareholder Representative Services LLC and the Olde Monmouth Stock Transfer Co., Inc.
(incorporated by reference to Exhibit 10.4 to the Registrant’s Current Report on Form 8-K, filed on November 6, 2020).
|
|
|
Ocuphire Pharma, Inc. 2021 Inducement Plan (incorporated by reference to Exhibit 10.41 to the Registrant’s Annual Report on Form 10-K, filed on March 11, 2021)
|
|
|
Form of Stock Option Grant Notice issued under the Ocuphire Pharma, Inc. 2021 Inducement Plan (incorporated by reference to Exhibit 10.9.1
to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
|
Employment Agreement dated November 11, 2020, by and between the Company and Amy Rabourn (incorporated by reference to Exhibit 10.42 to the Registrant’s Annual Report on Form 10-K,
filed on March 11, 2021)
|
|
|
First Amendment to the Employment Agreement by and among the Company and Amy Rabourn, effective as of March 26, 2023 (incorporated by
reference to Exhibit 10.10.1 to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
|
Capital on Demand™ Sales Agreement, dated March 11, 2021 between the Company and JonesTrading Institutional Services LLC (incorporated by reference to Exhibit 1.1 to the Registrant’s
Current Report on Form 8-K, filed on March 11, 2021).
|
|
|
Form of Purchase Agreement, dated as of June 4, 2021, by and among Ocuphire Pharma, Inc. and the purchasers identified on the signature pages thereto (incorporated by reference to
Exhibit 10.1 to the Registrant’s Current Report on Form 8-K/A, filed on June 7, 2021).
|
|
|
Processa License Agreement dated June 16, 2021 (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on June 23, 2021).
|
|
|
Viatris (f/k/a Famy Life Sciences) License and Collaboration Agreement dated November 6, 2022 (incorporated by reference to Exhibit 10.1 to
the Registrant’s Current Report on Form 8-K, filed on November 7, 2022).
|
|
|
Consulting Agreement dated April 8, 2022, by and between the Company and Jay Pepose (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q, filed
on May 13, 2022).
|
|
|
First Amendment to the Consulting Agreement dated September 19, 2022, by and between the Company and Jay Pepose (incorporated by reference to
Exhibit 10.15.1 to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
|
Amendment No. 2 to the Consulting Agreement dated December 1, 2022, by and between the Company and Jay Pepose (incorporated by reference to
Exhibit 10.15.2 to the Registrant’s Annual Report on Form 10-K (File No. 001-34079), filed on March 30, 2023).
|
|
|
Amended and Restated Non-Employee Director Compensation Policy dated July 1, 2022 (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly Report on Form 10-Q, filed on
August 12, 2022).
|
|
|
Interim President and CEO Consulting Letter Agreement by and between Ocuphire Pharma, Inc. and Richard Rodgers, dated April 20, 2023 (incorporated by reference to Exhibit 10.1 to the Registrant’s Quarterly
Report on Form 10-Q filed on May 15, 2023).
|
|
|
Non-Employee Director Compensation Policy (incorporated by reference to Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q filed on May 15, 2023).
|
|
Second Amended and Restated Non-Employee Director Compensation Policy (incorporated by reference to Exhibit 10.2 to the Registrant’s Quarterly Report on Form 10-Q filed on May 15, 2023).
|
|
|
Purchase Agreement, dated as of August 10, 2023, by and between Ocuphire Pharma, Inc. and Lincoln Park Capital Fund, LLC (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form
8-K, filed on August 11, 2023).
|
|
|
Registration Rights Agreement, dated as of August 10, 2023, by and between Ocuphire Pharma, Inc. and Lincoln Park Capital Fund, LLC (incorporated by reference to Exhibit 10.2 to the Registrant’s Current
Report on Form 8-K, filed on August 11, 2023).
|
|
|
Employment Agreement entered into on October 31, 2023 by and between Ocuphire Pharma, Inc. and George Magrath (incorporated by reference to Exhibit 10.1 to the Registrant’s Current
Report on Form 8-K, filed on November 1, 2023).
|
|
|
Form of Restricted Stock Unit Award and Form of Award Agreement (incorporated by reference to Exhibit 10.2 to the Registrant’s
Current Report on Form 8-K, filed on November 1, 2023).
|
|
|
First Amendment to 2021 Inducement Plan (incorporated by reference to Exhibit 10.3 to the Registrant’s Current Report on Form 8-K, filed on November 1, 2023).
|
|
|
Amended and Restated Employment Agreement, dated as of April 24, 2023, by and between Ocuphire Pharma, Inc. and Ronil Patel (incorporated by reference to Exhibit 10.1 to the
Registrant’s Current Report on Form 8-K, filed on December 6, 2023).
|
|
|
First Amendment to Amended and Restated Employment Agreement, dated as of December 1, 2023, by and between Ocuphire Pharma, Inc. and Ronil Patel (incorporated by reference to Exhibit
10.2 to the Registrant’s Current Report on Form 8-K, filed on December 6, 2023).
|
|
|
Employment Agreement, dated February 13, 2024, by and between the Company and Nirav Jhaveri (incorporated by reference to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K,
filed on February 16, 2024).
|
|
|
First Amendment to Employment Agreement, entered into on February 16, 2024, by and between the Company and Nirav Jhaveri (incorporated by reference to Exhibit 10.2 to the Registrant’s
Current Report on Form 8-K, filed on February 16, 2024).
|
|
|
Offer Letter entered into on November 20, 2023 by and between Ocuphire Pharma, Inc. and Joseph Schachle (incorporated by reference
to Exhibit 10.1 to the Registrant’s Current Report on Form 8-K, filed on November 27, 2023).
|
|
|
Subsidiaries of the Registrant.
|
|
|
Consent of Ernst & Young, LLP.
|
|
|
Certification of Principal Executive Officer pursuant to Rules 13a-14(a) and 15d-14(a) promulgated under the Securities Exchange Act of 1934, as adopted pursuant to Section 302 of the
Sarbanes-Oxley Act of 2002.
|
|
|
Certification of Principal Financial Officer pursuant to Rules 13a-14(a) and 15d-14(a) promulgated under the Securities Exchange Act of 1934, as adopted pursuant to section 302 of the
Sarbanes-Oxley Act of 2002.
|
|
|
Certification of Principal Executive Officer and Principal Financial Officer pursuant to Rules 13a-14(b) and 15d-14(b) promulgated under the Securities Exchange Act of 1934 and 18
U.S.C. Section 1350, as adopted pursuant to section 906 of The Sarbanes-Oxley Act of 2002.
|
|
Compensation Recovery Policy.
|
|
|
101.INS
|
Inline XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document).
|
|
101.SCH
|
Inline XBRL Taxonomy Extension Schema Document.
|
|
101.CAL
|
Inline XBRL Taxonomy Extension Calculation Linkbase Document
|
|
101.DEF
|
Inline XBRL Taxonomy Extension Definition Linkbase Document
|
|
101.LAB
|
Inline XBRL Taxonomy Extension Label Linkbase Document
|
|
101.PRE
|
Inline XBRL Taxonomy Extension Presentation Linkbase Document
|
|
104
|
Cover Page Interactive Data File (formatted as inline XBRL and contained in Exhibit 101)
|
| * |
Indicates management contract or compensatory plan.
|
| ** |
Indicates exhibits that are being filed herewith.
|
| + |
Certain schedules and exhibits have been omitted pursuant to Item 601(b)(2) of Regulation S-K. A copy of any omitted schedule and/or exhibit will be furnished to the SEC upon request.
|
|
++
|
Portions of this exhibit have been omitted in compliance with Item 601 of Regulation S-K.
|
| ITEM 16. |
FORM 10-K SUMMARY
|
| ITEM 8. |
FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
|
| 110 |
|
| 111 |
|
| 112 |
|
| 113 |
|
| 114 |
|
| 115-135 |
|
As of December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Assets
|
||||||||
|
Current assets:
|
||||||||
|
Cash and cash equivalents
|
$
|
|
$
|
|
||||
|
Accounts receivable
|
||||||||
|
Contract assets and unbilled receivables (Note
9)
|
||||||||
|
Prepaids and other current assets
|
|
|
||||||
|
Short-term investments
|
|
|
||||||
|
Total current assets
|
|
|
||||||
|
Property and equipment, net
|
|
|
||||||
|
Total assets
|
$
|
|
$
|
|
||||
|
Liabilities and stockholders’ equity
|
||||||||
|
Current liabilities:
|
||||||||
|
Accounts payable
|
$
|
|
$
|
|
||||
|
Accrued expenses
|
|
|
||||||
| Derivative liability | ||||||||
|
Total current liabilities
|
|
|
||||||
|
Total liabilities
|
|
|
||||||
|
Commitments and contingencies (Note 3 and Note 7)
|
||||||||
|
Stockholders’ equity:
|
||||||||
|
Preferred stock, par value $
|
|
|
||||||
|
Common stock, par value $
|
|
|
||||||
|
Additional paid-in capital
|
|
|
||||||
|
Accumulated deficit
|
(
|
)
|
(
|
)
|
||||
|
Total stockholders’ equity
|
|
|
||||||
|
Total liabilities and stockholders’ equity
|
$
|
|
$
|
|
||||
|
For the Year Ended
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
| License and collaborations revenue |
$ | $ | ||||||
| Operating expenses: | ||||||||
|
General and administrative
|
|
|
||||||
|
Research and development
|
|
|
||||||
|
Total operating expenses
|
|
|
||||||
|
(Loss) income from operations
|
(
|
)
|
|
|||||
| Financing costs (Note 8) |
( |
) | ||||||
|
Interest expense (Note 4)
|
|
(
|
)
|
|||||
|
Fair value change in derivative liabilities
|
|
|
||||||
|
Other income (expense), net
|
|
(
|
)
|
|||||
|
(Loss) income before income taxes
|
(
|
)
|
|
|||||
|
Provision for income taxes
|
(
|
)
|
(
|
)
|
||||
|
Net (loss) income
|
(
|
)
|
|
|||||
|
Other comprehensive (loss) income, net of tax
|
|
|
||||||
|
Comprehensive (loss) income
|
$
|
(
|
)
|
$
|
|
|||
|
Net (loss) income per share (Note 10):
|
||||||||
|
Basic
|
$
|
(
|
)
|
$
|
|
|||
| Diluted |
$ | ( |
) | $ | ||||
|
Number of shares used in per share calculations:
|
||||||||
|
Basic
|
|
|
||||||
| Diluted |
||||||||
|
Common Stock
|
Additional
Paid–In
|
Accumulated
|
Total
Stockholders’
|
|||||||||||||||||
|
Shares
|
Amount
|
Capital
|
Deficit
|
Equity
|
||||||||||||||||
|
Balance at December 31, 2021
|
|
$
|
|
$
|
|
$
|
(
|
)
|
$
|
|
||||||||||
|
Issuance of common stock in connection with the at-the-market program
|
||||||||||||||||||||
| Issuance costs |
— | ( |
) | ( |
) | |||||||||||||||
| Exercise of Series B warrants |
||||||||||||||||||||
| Stock-based compensation |
||||||||||||||||||||
| Exercise of stock options |
||||||||||||||||||||
| Net and comprehensive income |
— | |||||||||||||||||||
| Balance at December 31, 2022 | ( |
) | ||||||||||||||||||
|
Issuance of common stock in connection with the at-the-market program and purchase agreement
|
||||||||||||||||||||
|
Issuance costs
|
— | ( |
) | ( |
) | |||||||||||||||
|
Exercise of Series B warrants
|
||||||||||||||||||||
|
Stock-based compensation
|
||||||||||||||||||||
|
Exercise of stock options
|
||||||||||||||||||||
|
Net and comprehensive loss
|
— | ( |
) | ( |
) | |||||||||||||||
|
Balance at December 31, 2023
|
$ | $ | $ | ( |
) | $ | ||||||||||||||
|
For the Year Ended
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Operating activities
|
||||||||
|
Net (loss) income
|
$
|
(
|
)
|
$
|
|
|||
|
Adjustments to reconcile net (loss) income to net cash (used in) provided by operating activities:
|
||||||||
|
Stock-based compensation
|
|
|
||||||
|
Depreciation
|
|
|
||||||
|
Fair value change in derivative liabilities
|
(
|
)
|
|
|||||
| Financing costs |
||||||||
|
Unrealized loss from short-term investments
|
||||||||
|
Change in assets and liabilities:
|
||||||||
|
Accounts receivable
|
( |
) | ||||||
| Contract assets and unbilled receivables |
( |
) | ||||||
|
Prepaid expenses and other assets
|
|
(
|
)
|
|||||
|
Accounts payable
|
|
(
|
)
|
|||||
|
Accrued expenses
|
|
(
|
)
|
|||||
|
Net cash (used in) provided by operating activities
|
(
|
)
|
|
|||||
|
Investing activities
|
||||||||
|
Transaction costs in connection with asset acquisition
|
|
|
||||||
|
Net cash used in investing activities
|
|
|
||||||
|
Financing activities
|
||||||||
| Proceeds from issuance of common stock |
||||||||
|
Issuance costs attributed to common stock
|
(
|
)
|
(
|
)
|
||||
| Payments made on short-term loan principal |
( |
) | ||||||
|
Exercise of stock options and Series B warrants
|
|
|
||||||
|
Net cash provided by financing activities
|
|
|
||||||
|
Net increase in cash and cash equivalents
|
|
|
||||||
|
Cash and cash equivalents at beginning of period
|
|
|
||||||
|
Cash and cash equivalents at end of period
|
$
|
|
$
|
|
||||
|
Supplemental disclosure of cash flow information:
|
||||||||
|
Cash paid for income taxes
|
$
|
|
$
|
|
||||
|
Cash paid for interest
|
$
|
|
$
|
|
||||
|
Supplemental non-cash financing transactions:
|
||||||||
| Non-cash issuance of common stock in connection with equity purchase agreement |
$
|
$
|
||||||
| Value of derivative established in connection with the equity purchase agreement |
$
|
$
|
||||||
|
Unpaid issuance costs
|
$
|
|
$
|
|
||||
| 1. |
Company Description and Summary of Significant Accounting Policies
|
| • |
Level 1 inputs: Unadjusted quoted prices for identical assets or liabilities in active markets;
|
| • |
Level 2 inputs: Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, whether directly or indirectly, for substantially the full term of the
asset or liability; and
|
| • |
Level 3 inputs: Unobservable inputs in which there is little or no market data available, which requires management to develop its own assumptions in pricing the asset or liability.
|
|
As of December 31, 2023
|
||||||||||||||||
|
Description
|
Total
|
Level 1
|
Level 2
|
Level 3
|
||||||||||||
|
Assets:
|
||||||||||||||||
|
Short-term investments
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Total assets at fair value
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Liabilities:
|
||||||||||||||||
|
Derivative liability
|
$ |
$ |
$ |
$ | ||||||||||||
|
Total liabilities at fair value
|
$ |
$ | $ |
$ | ||||||||||||
|
As of December 31, 2022
|
||||||||||||||||
|
Description
|
Total
|
Level 1
|
Level 2
|
Level 3
|
||||||||||||
|
Assets:
|
||||||||||||||||
|
Short-term investments
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
Total assets at fair value
|
$
|
|
$
|
|
$
|
|
$
|
|
||||||||
|
|
2023
|
2022
|
||||||
|
Short-term investments
|
||||||||
|
Balance as of beginning of period
|
$
|
|
$
|
|
||||
|
Unrealized loss
|
(
|
)
|
(
|
)
|
||||
|
Balance as of end of period
|
$
|
|
$
|
|
||||
|
|
2023
|
2022
|
||||||
|
Derivative liabilities
|
||||||||
|
Balance as of beginning of period
|
$
|
|
$
|
|
||||
|
Purchase agreement execution
|
|
|
||||||
|
Unrealized gain
|
(
|
)
|
|
|||||
|
Balance as of end of period
|
$
|
|
$
|
|
||||
| 2. |
Merger
|
| • |
|
| • |
|
| • |
|
| 3. |
Commitments and Contingencies
|
| 4. |
Supplemental Balance Sheet Information
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Prepaids
|
$
|
|
$
|
|
||||
|
Other
|
|
|
||||||
|
Total prepaids and other current assets
|
$
|
|
$
|
|
||||
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
Equipment
|
$
|
|
$
|
|
||||
|
Furniture
|
|
|
||||||
|
Total property and equipment
|
|
|
||||||
|
Less accumulated depreciation
|
(
|
)
|
(
|
)
|
||||
|
Property and equipment, net
|
$
|
|
$
|
|
||||
|
Accrued expenses consist of the following (in thousands):
|
December 31,
|
|||||||
|
2023
|
2022
|
|||||||
|
Income taxes
|
$ | $ | ||||||
|
Payroll
|
|
|
||||||
|
Professional services
|
|
|
||||||
|
R&D services and supplies
|
|
|
||||||
|
Other
|
|
|
||||||
|
Total
|
$
|
|
$
|
|
||||
| 5. |
Related Party Transactions
|
| 6. |
Stock-based Compensation
|
|
December 31,
|
||||||||
|
2023
|
2022
|
|||||||
|
General and administrative
|
$
|
|
$
|
|
||||
|
Research and development
|
|
|
||||||
|
Total stock-based compensation
|
$
|
|
$
|
|
||||
|
Number of
Options
|
Weighted
Average
Exercise
Price
|
Weighted Average
Remaining
Contractual
Term (years)
|
Aggregate
Intrinsic
Value(1)
(in thousands)
|
|||||||||||||
|
Outstanding at December 31, 2021
|
|
$
|
|
|
$
|
|
||||||||||
|
Granted
|
|
$
|
|
|
|
|||||||||||
|
Exercised
|
(
|
)
|
$
|
|
|
|
||||||||||
|
Forfeited/Cancelled
|
(
|
)
|
$
|
|
|
|
||||||||||
|
Outstanding at December 31, 2022
|
|
$
|
|
|
$
|
|
||||||||||
|
Granted
|
|
$
|
|
|||||||||||||
|
Exercised
|
(
|
)
|
$
|
|
||||||||||||
|
Forfeited/Cancelled
|
(
|
)
|
$
|
|
||||||||||||
|
Outstanding at December 31, 2023
|
|
$
|
|
|
$
|
|
||||||||||
| Vested and expected to vest at December 31, 2023 | $ | $ | ||||||||||||||
|
Vested and exercisable at December 31, 2023
|
|
$
|
|
|
$
|
|
||||||||||
| (1) |
|
|
2023
|
2022
|
|||||||
|
Expected stock price volatility
|
|
%
|
|
%
|
||||
|
Expected life of options (years)
|
|
|
||||||
|
Expected dividend yield
|
|
%
|
|
%
|
||||
|
Risk free interest rate
|
|
%
|
|
%
|
||||
|
Number of
Shares
|
||||
|
Non-vested at December 31, 2022
|
|
|||
|
Granted
|
|
|||
| Forfeited |
( |
) | ||
|
Vested
|
(
|
)
|
||
|
Non-vested at December 31, 2023
|
|
|||
| 7. |
Apexian Sublicense Agreement
|
| 8. |
Stockholders' Equity
|
| • |
the lowest sale price for the Company’s common stock on Nasdaq on the
purchase date of such shares; and
|
| • |
the average of the three (3) lowest closing sale prices for the
Company’s common stock on Nasdaq during the ten (
|
| • |
three
(
|
| • |
|
| • |
the closing sale price of the Company’s common stock on Nasdaq on the applicable
Accelerated Purchase date; and
|
| • |
the volume-weighted average price of the Company’s common stock on Nasdaq during the
applicable Accelerated Purchase Measurement Period on the applicable Accelerated Purchase date.
|
| 2023 | 2022
|
|||||||
| Contract Assets and Unbilled Receivables | ||||||||
|
Balance as of beginning of period
|
$
|
|
$ | |||||
| License transfer |
( |
) | ||||||
|
Revenue recognized
|
||||||||
|
Reclassification to accounts receivable related to costs billed under the Viatris License Agreement
|
( |
) | ( |
) | ||||
| Balance as of end of period | $ | $ | ||||||
| 10. |
Net (loss) income per share
|
| 2023 | 2022 |
|||||||
|
Denominator (weighted average shares)
|
|
|
||||||
|
Basic common shares outstanding
|
|
|
|
|
||||
|
Dilutive stock options
|
|
|
||||||
|
Dilutive warrants
|
|
|
||||||
|
Diluted common shares outstanding
|
|
|
||||||
|
2023
|
2022
|
|||||||
|
Series A, Series B and RDO warrants
|
|
|||||||
|
Stock options
|
|
|
||||||
|
RSUs
|
|
|
||||||
|
Former Rexahn warrants
|
|
|||||||
| 11. |
Income Taxes
|
|
2023
|
2022
|
|||||||
| Income tax (benefit) provision at federal statutory rate | ( |
)% | % | |||||
|
Valuation allowance
|
( |
) | ||||||
|
State income tax, net of federal benefit
|
( |
) | ||||||
|
Financing contracts
|
||||||||
|
Stock options
|
||||||||
|
Research and development
|
( |
) | ( |
) | ||||
|
Other
|
( |
) | ||||||
|
Effective tax rate
|
% | % | ||||||
| 2023 |
2022 |
|||||||
|
(Loss) income before income taxes:
|
$ | ( |
) | $ | ||||
|
Current:
|
||||||||
|
Federal
|
$ | $ | ||||||
|
State
|
||||||||
|
Total current tax provision (benefit)
|
$ | |||||||
|
Deferred:
|
||||||||
|
Federal
|
||||||||
|
State
|
||||||||
|
Total tax provision (benefit)
|
$ | $ | ||||||
|
2023
|
2022
|
|||||||
|
Deferred tax assets:
|
||||||||
| Federal and state operating loss carryforwards | $ | $ | ||||||
|
Acquired intangibles
|
|
|||||||
| Deferral of research and development costs |
||||||||
|
Organizational costs
|
|
|||||||
| Other | ||||||||
|
Stock-based compensation
|
|
|||||||
|
Research and development credit carryforward
|
|
|||||||
|
Subtotal
|
|
|||||||
|
Valuation allowance
|
(
|
)
|
(
|
)
|
||||
|
Total deferred tax assets, net of valuation allowance
|
|
|
||||||
|
Deferred tax liabilities:
|
||||||||
| Deferred revenue |
( |
) | ||||||
|
Total deferred tax liabilities
|
|
(
|
)
|
|||||
|
Net deferred tax assets
|
$
|
|
$
|
|
||||
| 12. |
Deferred Compensation Plan
|
| 13. |
Subsequent Events
|
|
OCUPHIRE PHARMA, INC.
|
||
|
Dated: March 8, 2024
|
By:
|
/s/ George Magrath
|
|
George Magrath
|
||
|
Chief Executive Officer and Director
|
||
|
By
|
/s/ George Magrath
|
Date: March 8, 2024
|
|
|
George Magrath
|
|||
|
Chief Executive Officer and Director (Principal Executive Officer)
|
|||
|
By
|
/s/ Nirav Jhaveri
|
Date: March 8, 2024
|
|
|
Nirav Jhaveri
|
|||
|
Chief Financial Officer (Principal Financial Officer)
|
|||
|
By
|
/s/ Amy Rabourn
|
Date: March 8, 2024
|
|
|
Amy Rabourn
|
|||
|
Senior Vice President of Finance (Principal Accounting Officer)
|
|||
|
By
|
/s/ Sean Ainsworth
|
Date: March 8, 2024
|
|
|
Sean Ainsworth
|
|||
|
Director
|
|||
|
By
|
/s/ James S. Manuso
|
Date: March 8, 2024
|
|
|
James S. Manuso
|
|||
|
Director
|
|||
|
By
|
/s/ Cam Gallagher
|
Date: March 8, 2024
|
|
|
Cam Gallagher
|
|||
|
Director
|
|||
|
By
|
/s/ Jay Pepose
|
Date: March 8, 2024
|
|
|
Jay Pepose
|
|||
|
Director
|
|||
|
By
|
/s/ Richard J. Rodgers
|
Date: March 8, 2024
|
|
|
Richard J. Rodgers
|
|||
|
Director
|
|||
|
By
|
/s/ Susan K. Benton
|
Date: March 8, 2024
|
|
|
Susan K. Benton
|
|||
|
Director
|
|
Subsidiaries
|
Jurisdiction of Incorporation
|
|
None
|
| (1) |
Registration Statement (Form S-3 No. 333-276462) pertaining to the registration of Ocuphire Pharma, Inc. debt and equity securities;
|
| (2) |
Registration Statement (Form S-8 No. 333-276471) pertaining to the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan;
|
| (3) |
Registration Statement (Form S-8 No. 333-275673) pertaining to the Ocuphire Pharma, Inc. 2021 Inducement Plan;
|
| (4) |
Registration Statement (Form S-8 No. 333-271150) pertaining to the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan;
|
| (5) |
Registration Statement (Form S-8 No. 333-264139) pertaining to the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan;
|
| (6) |
Registration Statement (Form S-8 No. 333-254923) pertaining to the Ocuphire Pharma, Inc. 2021 Inducement Plan and Ocuphire Pharma, Inc. 2020 Equity Incentive Plan;
|
| (7) |
Registration Statement (Form S-8 No. 333-249978) pertaining to the Ocuphire Pharma, Inc. 2020 Equity Incentive Plan and Ocuphire Pharma, Inc. 2018 Equity Incentive Plan;
|
| (8) |
Registration Statement (Form S-8 No. 333-217627) pertaining to the Rexahn Pharmaceuticals, Inc. 2013 Stock Option Plan;
|
| (9) |
Registration Statement (Form S-8 No. 333-189240) pertaining to the Rexahn Pharmaceuticals, Inc. 2013 Stock Option Plan; and
|
| (10) |
Registration Statement (Form S-8 No. 333-129294) pertaining to the Rexahn Pharmaceuticals, Inc. 2005 Stock Option Plan
|
| 1. |
I have reviewed this Annual Report on Form 10-K of Ocuphire Pharma, Inc. (the “Company”);
|
| 2. |
Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not
misleading with respect to the period covered by this report;
|
| 3. |
Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and
for, the periods presented in this report;
|
| 4. |
The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting
(as defined in Exchange Act Rules 13a-15(f) and 15d-15(f) for the registrant and have:
|
| (a) |
Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant is made known to us by others within
those entities, particularly during the period in which this report is being prepared;
|
| (b) |
Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and
the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
|
| (c) |
Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this
report based on such evaluation; and
|
| (d) |
Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report)
that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
|
| 5. |
The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of
directors (or persons performing the equivalent functions):
|
| (a) |
all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and report
financial information; and
|
| (b) |
any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
|
|
Date: March 8, 2024
|
/s/ George Magrath
|
|
|
Name:
|
George Magrath
|
|
|
Title:
|
Chief Executive Officer
(Principal Executive Officer)
|
|
| 1. |
I have reviewed this Annual Report on Form 10-K of Ocuphire Pharma, Inc. (the “Company”);
|
| 2. |
Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not
misleading with respect to the period covered by this report;
|
| 3. |
Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and
for, the periods presented in this report;
|
| 4. |
The registrant’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial
reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f) for the registrant and have:
|
| (a) |
Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant is made known to us by others within
those entities, particularly during the period in which this report is being prepared;
|
| (b) |
Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and
the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
|
| (c) |
Evaluated the effectiveness of the registrant’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by
this report based on such evaluation; and
|
| (d) |
Disclosed in this report any change in the registrant’s internal control over financial reporting that occurred during the registrant’s most recent fiscal quarter (the registrant’s fourth fiscal quarter in the case of an annual report)
that has materially affected, or is reasonably likely to materially affect, the registrant’s internal control over financial reporting; and
|
| 5. |
The registrant’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant’s auditors and the audit committee of the registrant’s board of
directors (or persons performing the equivalent functions):
|
| (a) |
all significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant’s ability to record, process, summarize and
report financial information; and
|
| (b) |
any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant’s internal control over financial reporting.
|
|
Date: March 8, 2024
|
/s/ Nirav Jhaveri
|
|
|
Name:
|
Nirav Jhaveri
|
|
|
Title:
|
Chief Financial Officer
|
|
|
(Principal Financial Officer)
|
||
|
Date: March 8, 2024
|
|
|
/s/ George Magrath
|
|
|
George Magrath
|
|
|
Chief Executive Officer
|
|
|
(Principal Executive Officer)
|
|
|
/s/ Nirav Jhaveri
|
|
|
Nirav Jhaveri
|
|
|
Chief Financial Officer
|
|
|
(Principal Financial Officer)
|
|
|
1.
|
That the rent for the Suite known as Suite 120 will be as follows:
|
|
| 01/01/2024 – 12/31/2024 | $22.25 per rentable square foot | ||
| |
|
||
| 2. | Expiration Date: | December 31, 2024 | |
| 3. | Tenant Share: | 2.13% | |
| 4. | Base Tax: | $1.39 per sq. ft. |
|
TENANT:
|
LANDLORD:
|
|
Ocuphire Pharma, Inc.
|
DUKE & DUKE,
|
|
a Limited Partnership
|
|
| /s/ Amy Rabourn | /s/ Trevor Duke |
|
Name: Amy Rabourn
|
Name: Trevor Duke
|
|
Its: SVP of Finance
|
Its:Property Manager
|
|
Date:11/29/2023
|
Date:11/29/2023
|

| Signature | ||
| Print Name | ||
| Date | ||
|
Ocuphire Pharma, Inc.
Compensation Recovery Policy
|
Page 7
|
Balance Sheets (Parenthetical) - $ / shares |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Stockholders' equity: | ||
| Preferred stock, par value (in dollars per share) | $ 0.0001 | $ 0.0001 |
| Preferred stock, shares authorized (in shares) | 10,000,000 | 10,000,000 |
| Preferred stock, shares issued (in shares) | 0 | 0 |
| Preferred stock, shares, outstanding (in shares) | 0 | 0 |
| Common stock, par value (in dollars per share) | $ 0.0001 | $ 0.0001 |
| Common stock, shares authorized (in shares) | 75,000,000 | 75,000,000 |
| Common stock, shares issued (in shares) | 23,977,491 | 20,861,315 |
| Common stock, shares, outstanding (in shares) | 23,977,491 | 20,861,315 |
Company Description and Summary of Significant Accounting Policies |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company Description and Summary of Significant Accounting Policies [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company Description and Summary of Significant Accounting Policies |
Nature of Business
Ocuphire Pharma, Inc. (the “Company” or “Ocuphire”) is a
clinical-stage biopharmaceutical company focused on developing novel therapies for the treatment of unmet needs of patients with retinal and refractive eye disorders. The Company’s headquarters is located in Farmington Hills,
Michigan.
The Company’s lead retinal product candidate, APX3330, is a small-molecule inhibitor of Ref-1 (reduction oxidation effector factor-1 protein).
Ref-1 is a regulator of transcription factors such as HIF-1α and NF-kB. Inhibiting Ref-1 reduces levels of vascular endothelial growth factor (“VEGF”) and inflammatory cytokines which are known to play key roles in ocular angiogenesis and
inflammation. APX3330 is an oral tablet administered once or twice per day in development for the treatment of diabetic retinopathy (“DR”). A
Phase 2 study in subjects with DR or diabetic macular edema was completed and results were reported in January 2023. An End-of-Phase 2 (“EOP2”) meeting with the U.S. Food and Drug Administration (the “FDA”) was held in October 2023 at
which the Company obtained agreement on the registration endpoint supporting the advancement of APX3330 into future clinical trials. Ocuphire submitted a Special Protocol
Assessment (“SPA”) to the FDA in February 2024 to seek agreement on the clinical trial protocol and statistical analysis plan.
The Company has also in-licensed APX2009 and APX2014, which are second-generation analogs of APX3330. The unique mechanism of action of this family of Ref-1 inhibitors of reducing
angiogenesis and inflammation could potentially be beneficial in treating other retinal diseases such as age-related macular degeneration, geographic atrophy, and non-ophthalmic diseases.
In November 2022, the Company entered into a license and collaboration agreement (the “Viatris License Agreement”) with FamyGen Life Sciences, Inc. (“Famy”) (acquired by Viatris, Inc.
(“Viatris”) in January 2023) pursuant to which it granted Viatris an exclusive license to develop, manufacture, import, export and commercialize its refractive product candidate Phentolamine Ophthalmic Solution 0.75%, formerly known as Nyxol
(“PS”). PS is a once-daily eye drop formulation of phentolamine mesylate designed to reduce pupil diameter and improve visual acuity. PS was approved by the FDA for the treatment for pharmacologically-induced mydriasis under the brand name
RYZUMVI in September 2023. The VEGA-2 Phase 3 study in presbyopia achieved its primary endpoint. PS is currently in Phase 3 clinical trials for presbyopia (age-related blurry near vision). On December 5, 2023, the Company received FDA
Agreement Under Special Protocol Assessment for LYNX-2, a Phase 3 Trial of PS for the treatment of decreased Visual Acuity under dim (mesopic) light conditions following keratorefractive surgery.
Reverse Merger with Rexahn
On June 17, 2020, Ocuphire, Rexahn
Pharmaceuticals, Inc. (“Rexahn”) and Razor Merger Sub, Inc., a Delaware corporation and wholly-owned subsidiary of Rexahn (“Merger Sub”), entered into an Agreement and Plan of Merger and Reorganization, as amended on June 29, 2020 (as
amended, the “Merger Agreement”), pursuant to which, among other things, and subject to the satisfaction or waiver of certain conditions set forth in the Merger Agreement, Merger Sub merged with and into Ocuphire, with Ocuphire continuing
as a wholly-owned subsidiary of Rexahn and the surviving corporation of the merger (the “Merger”). The Merger closed on November 5, 2020. Upon completion of the Merger, Rexahn changed its name to Ocuphire Pharma, Inc. and changed its ticker
symbol on the Nasdaq Capital Market (“Nasdaq”) to “OCUP”.
Basis of Presentation
The accompanying financial statements have been prepared in accordance with accounting standards generally accepted in the United States of America (“GAAP”).
The Company does not have any subsidiaries or other entities that require consolidation for financial statement reporting purposes.
Liquidity
The accompanying financial statements have been prepared
on the basis that the Company will continue as a going concern. From its inception, the Company has devoted substantially all of its efforts to drug development and conducting clinical trials.
As of
December 31, 2023, the Company had $50.5 million in cash and cash equivalents. The Company
believes its current available cash and cash equivalents will be sufficient to fund the Company’s planned expenditures and meet its obligations for at least twelve months from the date of issuance of these financial statements.
In the future, the Company may need to raise additional funds until it is able to generate sufficient revenues to fund its development activities. The Company’s future
operating activities, coupled with its plans to raise capital or issue debt financing, may provide additional liquidity in the future, however these actions are not solely within the control of the Company and the Company is unable to
predict the outcome of these actions to generate the liquidity ultimately required.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the
financial statements and accompanying notes. Actual results could differ from those estimates.
Segment Information
Operating segments are components of an enterprise for which separate financial information is available and is evaluated regularly by the Company’s chief operating
decision maker in deciding how to allocate resources and assessing performance. The Company’s chief operating decision maker is its Chief Executive Officer or such person functioning in such role. The Company’s Chief Executive Officer views the
Company’s operations and manages its business in one operating segment, which is the business of development of products related to
vision performance and health. Accordingly, the Company has a single reporting segment.
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of 90 days or less at the time of deposit to be cash equivalents.
Concentration of Credit Risk
Financial
instruments that potentially subject the Company to a concentration of credit risk consist of cash and cash equivalents. Management follows approved policies established by its Board of Directors to reduce credit risk associated with the
Company’s cash deposit and investment accounts. Pursuant to these policies, the Company limits its exposure through the kind, quality and concentration of its investments. The Company’s cash and cash equivalents are held or managed by two
financial institutions in the United States. As of December 31, 2023, the Company had cash equivalents of $50.2 million that
were not eligible for coverage by Federal Deposit Insurance Corporation (“FDIC”). These balances are invested in funds whose assets consist almost entirely of securities issued by the U.S. Treasury or guaranteed by the U.S. government.
Short-term Investments
The Company determines the appropriate classification of its investments in debt and equity securities at the time of purchase and records them on a settlement
date basis. The Company’s short-term investments are comprised of equity securities, which in accordance with the fair value hierarchy described below are recorded at fair value using Level l inputs on the balance sheets. Subsequent changes
in fair values are recorded in other income (expense), net on the statements of comprehensive (loss) income. The Company classifies investments available to fund current operations as current assets on its balance sheets. The Company did not recognize any impairments on its investments to date through December 31, 2023.
Revenue Recognition
The Company follows the provisions of Accounting Standards Codification (“ASC”) 606, Revenue from Contracts with Customers. The guidance provides a five-step model to determine
how revenue is recognized. The Company has entered into license agreements which have revenue recognition implications (See Note 9 – License and Collaboration Agreements).
In determining the appropriate amount of revenue to be recognized, the Company performs the following steps: (i) identification of the contracts with a
customer; (ii) determination of the performance obligations in the contract; (iii) measurement of the transaction price, including potential constraints on variable consideration; (iv) allocation of the transaction price to the performance
obligations based on estimated stand-alone selling prices; and (v) recognition of revenue when (or as) the Company satisfies a performance obligation.
A performance obligation is a promise in a contract to transfer a distinct good or service to
the customer and is the unit of account in ASC 606. Performance obligations may include license rights, development and other services. Significant management judgment is required to determine the level of effort required under an
arrangement and the period over which the Company expects to complete its performance obligations under the arrangement. If the Company cannot reasonably estimate when its performance obligations are either completed or become
inconsequential, then revenue recognition is deferred until the Company can reasonably make such estimates. Revenue is then recognized over the remaining estimated period of performance using the cumulative catch-up method.
As part of the accounting for these arrangements, the Company must develop assumptions that require judgment to determine the stand-alone selling price of each
performance obligation identified in the contract. The Company uses key assumptions to determine the stand-alone selling price, which may include forecasted revenues, development timelines, reimbursement rates for personnel costs, discount
rates and probabilities of technical and regulatory success. The Company allocates the total transaction price to each performance obligation based on the relative standalone selling prices of the promised goods or service underlying each
performance obligation.
Licenses of intellectual property and research and development services: If the license to the
Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company recognizes revenues from non-refundable, up-front fees allocated to the license when the license
is transferred to the customer, and the customer can use and benefit from the license. For licenses that are bundled with other obligations, such as research and development services, the Company utilizes judgment to assess the nature of
the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue
from non-refundable, up-front fees. For research and development services that are distinct from a license transfer obligation, the Company determines whether the services are satisfied over time or at a point in time and, if over time, the
appropriate method of measuring progress for purposes of recognizing revenue from such services. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue
recognition.
Milestone payments: At the inception of each arrangement
that includes milestone payments, the Company evaluates whether the milestones are considered probable of being achieved and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable
that a significant revenue reversal would not occur, the value of the associated milestone (such as a regulatory submission) is included in the transaction price. Milestone payments that are not within the control of the Company, such as
approvals from regulators, are not considered probable of being achieved until such contingency occurs (such as receipt of those approvals).
Royalties: For arrangements that include sales-based royalties, including milestone payments
based on the level of sales, and the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (a) when the related sales occur, or (b) when the performance obligation to
which some or all of the royalty has been allocated has been satisfied (or partially satisfied).
Contract Assets and Unbilled Receivables
The Company recognizes contract assets and unbilled receivables when goods or services are transferred to the customer before the customer pays or before reimbursement for payment is
billed or due, excluding any amounts presented as an account receivable. The Company recorded contract assets and unbilled receivables in connection with a license and collaboration agreement (See Note 9 – License and Collaboration Agreements).
Accounts Receivable and Allowances for Credit Losses
The Company records a provision for credit losses, when
appropriate, based on historical experience, current conditions and reasonable supportable forecasts. In estimating the allowance for credit losses, the Company considers, among other factors, the estimate of credit losses over
the remaining expected life of the asset, primarily using historical experience and current economic conditions that could affect the collectability of the balances in the future. Account balances are charged off against the
allowance when the Company believes that it is probable that the receivable will not be recovered. Actual write-offs may be in excess of the Company’s estimated allowance. The Company has not incurred any bad debt expense to date and no allowance for credit losses has been recorded during the periods presented.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel-related costs, including salaries, benefits and stock-based compensation costs, for personnel in
functions not directly associated with research and development activities. Other significant costs include insurance coverage for directors and officers and other property and liability exposures, legal fees relating to intellectual property and
corporate matters, professional fees for accounting and tax services, other services provided by business consultants and legal settlements.
Research and Development
Research and development expenses consist of costs
incurred in performing research and development activities, including compensation, benefits and stock-based compensation costs for research and development employees and costs for consultants, costs associated with nonclinical studies and
clinical trials, regulatory activities, manufacturing activities to support clinical activities, license fees, nonlegal patent costs, fees paid to external service providers that conduct certain research and development, and an allocation of
overhead expenses. Research and development expenses include costs that are reimbursed under the Viatris License Agreement (See Note 9 – License and
Collaboration Agreements).
Financing costs
Financing costs consist of issuance costs
attributed to an equity line financing facility with Lincoln Park (See Note 8 – Stockholders’ Equity).
Interest Expense
Interest expenses were attributed to interest on principal related to a short-term loan during the period it was outstanding. The short-term loan was fully repaid in May 2022. Other Income (Expense), net
Other income (expense), net includes interest earned from cash and cash equivalent investments, realized and unrealized gains (losses) from equity investments and
reimbursements in connection with grants and other sources when they occur. In addition, this line item includes payments made by the Company in connection with the Contingent Value Rights Agreement discussed further below with former Rexahn
shareholders.
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with the provisions of the Financial Accounting Standards Board (“FASB”) ASC 718, Compensation — Stock Compensation. Accordingly, compensation costs related to equity instruments granted are recognized at the grant date fair value. The Company records forfeitures when they occur.
Stock-based compensation arrangements to non-employees are accounted for in accordance with the applicable provisions of ASC 718.
Derivative Liability
The Company evaluates all
features contained in financing agreements to determine if there are any embedded derivatives that require separation from the underlying agreement under ASC 815 – Derivatives and Hedging. An embedded
derivative that requires separation is accounted for as a separate liability from the host agreement. The separated embedded derivative is accounted for separately on a fair market value basis. The Company records the fair value changes of a
separated embedded derivative at each reporting period in the statements of comprehensive (loss) income under the fair value change in derivative liability line item. The Company determined that certain features under an equity line financing
(See Note 8 — Stockholders’ Equity) collectively qualified as an embedded derivative. The derivative was accounted for separately from the underlying equity line financing agreement.
Fair Value Measurements
The Company follows accounting guidance that emphasizes that fair value is a market-based measurement, not an entity-specific measurement. Fair value is defined as
“the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.” Fair value measurements are defined on a three-level hierarchy:
As of December 31, 2023 and
2022, the fair values of cash and cash equivalents, accounts receivable, contract assets and unbilled receivables, prepaid and other current assets, accounts payable, and accrued expenses approximated their carrying values because of the
short-term nature of these assets or liabilities. The fair value of the short-term investments, while outstanding, were based on observable Level 1 inputs in the form of quoted market prices from a major stock exchange. The fair value of the
derivative liability associated with the equity line financing facility (See Note 8 – Stockholders’ Equity) was based on cash flow models discounted at current implied market rates representing expected returns by market participants for
similar instruments and are based on Level 3 inputs as well the Company’s underlying stock price and associated volatility, expected term of the financing and market interest rates. The fair value of the warrant liabilities, while outstanding, were based on a Black-Scholes option model using Level 3 inputs. There were no transfers between fair value hierarchy levels during the years ended December 31, 2023 and 2022.
The fair value of financial instruments measured on a recurring basis is as follows (in thousands):
The following table provides a roll-forward of short-term investments and derivative liabilities measured at fair value on a recurring basis using observable Level
1 and Level 3 inputs, as applicable, for the years ended December 31, 2023 and 2022 (in thousands):
Rexahn Warrants
The fair value of the warrant
liabilities associated with the Rexahn warrants was de minimis during the periods presented. The last of the Rexahn warrants classified as liabilities expired in April 2023 unexercised. See Note 2 – Merger for additional background.
There were no financial instruments measured on a
non-recurring basis for any of the periods presented.
Recent Accounting Pronouncements
In June 2016, the FASB issued Accounting Standards Update (“ASU”)
2016-13, Financial Instruments – Credit Losses. The ASU sets forth a current expected credit loss (CECL) model which requires the Company to measure all expected credit losses for financial instruments
held at the reporting date based on historical experience, current conditions, and reasonable supportable forecasts. This replaces the existing incurred loss model and is applicable to the measurement of credit losses on financial assets
measured at amortized cost and applies to some off-balance sheet credit exposures. The Company adopted this ASU on January 1, 2023 and it did not have a significant impact on its financial statements.
In August 2020, FASB issued ASU 2020-06,
Debt—Debt
with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, which, among other things, provides guidance on how to account for contracts on an entity’s own equity. This ASU eliminates the
beneficial conversion and cash conversion accounting models for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted for as derivatives because of specific
settlement provisions. In addition, this ASU modifies how particular convertible instruments and certain contracts that may be settled in cash or shares impact the diluted earnings per share computation. The amendments in this ASU are
effective for public business entities that meet the definition of an SEC filer, excluding entities eligible to be smaller reporting companies as defined by the SEC, for fiscal years beginning after December 15, 2021, including interim
periods within those fiscal years. For all other entities, the amendments are effective for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. The Company adopted this ASU on January 1, 2023
and the adoption did not have a material impact on its financial statements.
In November 2023, the FASB issued ASU 2023-07 - Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which enhances reportable segment disclosure requirements, primarily through disclosures of significant segment expenses. This ASU is effective for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years, with early adoption permitted. The guidance must be applied retrospectively to all prior periods presented. The Company is currently evaluating the impact of adoption of this guidance on its financial statements. In December 2023, the FASB issued ASU 2023-09 Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which enhances income tax disclosures primarily related to the rate reconciliation and income taxes paid information. This guidance also includes certain other amendments to improve the effectiveness of income tax disclosures. This ASU is effective for fiscal years beginning after December 15, 2024, including interim periods within those fiscal years and should be applied on a prospective basis, with retrospective application permitted. The Company is currently evaluating the impact of adoption of this guidance on its financial statements. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Merger |
12 Months Ended | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||
| Merger [Abstract] | ||||||||||||
| Merger |
On November 5, 2020, the Company completed the Merger transaction with Rexahn. In connection with the Merger, the Company, Shareholder Representatives Services
LLC, as representative of the Rexahn stockholders prior to the Merger, and Olde Monmouth Stock Transfer Co., Inc., as the rights agent, entered into a Contingent Value Rights Agreement (the “CVR Agreement”).
Pursuant to the terms of the Merger and the CVR Agreement, Rexahn stockholders of record as of immediately prior to the effective time of the Merger received one contingent value right (“CVR”) for each share of Rexahn common stock held.
Each CVR entitles such holders to receive, for each calendar quarter (each, a “CVR Payment Period”) during the 15-year period after the closing (the “CVR Term”), an amount equal to the following:
The CVRs are not transferable, except in certain limited circumstances, will not be certificated or evidenced by any instrument, will not accrue interest and will not be registered with the SEC or listed for trading on any exchange.
The CVR Agreement will continue in effect until the later of the end of the CVR Term and the payment of all amounts payable thereunder. As of December 31, 2023, no payments subject to the CVR had been received beyond those previously reported in the second and third quarters of calendar year 2021. In addition, no milestones had been accrued as there were no
potential milestones yet considered probable beyond those previously reported.
Former Rexahn Warrants
Following the closing of the Merger, 231,433
outstanding, unexercised Rexahn warrants to purchase common stock remained outstanding, the majority of which were subsequently repurchased according to the terms of the original warrant agreements. As of December 31, 2023, 58,597 of the Rexahn warrants remained outstanding with an exercise price of $38.40 per share with an average remaining contractual life of 0.1 years
and were accounted for and classified as equity.
|
Commitments and Contingencies |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2023 | |||
| Commitments and Contingencies [Abstract] | |||
| Commitments and Contingencies |
Apexian Sublicense Agreement
On January 21, 2020, the Company entered into a sublicense agreement with Apexian Pharmaceuticals, Inc., pursuant to which it obtained exclusive worldwide patent and
other intellectual property rights. In exchange for the patent and other intellectual rights, the Company agreed to certain milestone payments and royalty payments on future sales (See Note 7 — Apexian Sublicense Agreement). As of December 31,
2023, there was sufficient uncertainty with regard to any future cash milestone payments under the sublicense agreement that no liabilities were recorded related to the sublicense agreement.
Facility Leases
The Company has a short-term, non-cancellable facility lease (the “HQ Lease”) for its headquarters. The HQ Lease qualified for the short-term lease exception under
ASC 842, Leases. The monthly base rent for the HQ Lease is approximately $3,000. The rent
expense associated with the HQ Lease amounted to $36,000 and $39,000 during the years ended December 31, 2023 and 2022, respectively. The total remaining expected rental payments under the HQ Lease amount to $36,000 through its current expiration date of December 31, 2024.
Other
In the ordinary course of business, from time to time, the Company may be subject to a broad range of claims and legal proceedings that relate to contractual
allegations, patent infringement and other claims. In addition, the Company from time to time may be potentially committed to reimburse third parties for costs incurred associated with business development related transactions upon the
achievement of certain milestones. The Company establishes accruals when applicable for matters and commitments which it believes losses are probable and can be reasonably estimated. To date, no loss contingency for such matters and potential
commitments have been recorded. Although it is not possible to predict with certainty the outcome of these matters or potential commitments, the Company is of the opinion that the ultimate resolution of these matters and potential commitments
will not have a material adverse effect on its results of operations or financial position.
|
Supplemental Balance Sheet Information |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supplemental Balance Sheet Information [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supplemental Balance Sheet Information |
Prepaid and Other Current Assets
Prepaid and other current assets consist of the following (in thousands):
Property and Equipment, net
Property and equipment held for use by category are presented in the following table (in thousands):
Depreciation expense was $6,000 and 4,000 during the years ended December 31, 2023 and 2022, respectively.
Accrued Expenses
Short-Term Loan
The Company entered into an unsecured short-term loan (the “Loan”) agreement in the amount of $0.6 million in November 2021 related to financing an insurance policy. The Loan was payable in six monthly installments of $108,000 beginning in December
2021. The Loan had an annual interest rate of 5.5% per annum. Interest expense in the amount of $9,000 was recognized in connection with the Loan during the year ended December 31, 2022. The final payment on the Loan was made in May 2022.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Related Party Transactions |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2023 | |||
| Related Party Transactions [Abstract] | |||
| Related Party Transactions |
On April 8, 2022, Ocuphire entered into a consulting agreement for medical advisory services with Jay Pepose, a director of the Company. The consulting agreement
provided for $10,000 a month in cash payments, effective as of April 1, 2022. Additionally, on April 8, 2022, in connection with the
consulting arrangement, Dr. Pepose received a stock option grant for 50,000 options, of which 25% vested on March 31, 2023, with the remainder vesting in equal monthly installments over 36 months. The consulting agreement was amended on September 19, 2022 to provide for vesting acceleration for stock-based awards in the event of a change in control. The consulting
agreement was also amended effective December 1, 2022 to increase the cash payment to $25,000 per month.
The Company incurred related consulting expenses of $300,000
and $105,000 during the years ended December 31, 2023 and 2022, respectively. As of December 31, 2023 and 2022, $25,000 and $25,000 of the related
consulting expenses were unpaid, respectively.
On April 19, 2023, Ocuphire
appointed Richard Rodgers, a director of the Company, as interim President and Chief Executive Officer. In connection with his appointment, Ocuphire and Mr. Rodgers entered into a letter agreement concerning Mr. Rodgers’s services (the “Letter
Agreement”). The Letter Agreement provided that Mr. Rodgers (i) was to receive a $40,000 monthly salary, and (ii) is eligible for a
potential prorated bonus at the discretion of Ocuphire’s Board of Directors, at the end of his term as interim President and Chief Executive Officer. Mr. Rodgers also received 50,000 restricted stock units under the Company’s 2020 Equity Incentive Plan which will vest 12 months following the grant date. The Company incurred related expenses of $255,000 during the
year ended December 31, 2023. As of December 31, 2023, $100,000 of the related expenses were unpaid related to a prorated bonus.
|
Stock-based Compensation |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-based Compensation [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-based Compensation |
Stock-based compensation expense was included in general and administrative and research and development costs as follows in the accompanying statements of
comprehensive (loss) income for the periods indicated below (in thousands):
Inducement Plan
On February 22,
2021, the Company adopted the Ocuphire Pharma, Inc. 2021 Inducement Plan (the “Inducement Plan”) which was amended on November 1, 2023, pursuant to which the Company reserved 2,325,258 shares of its common stock to be used exclusively for grants of awards to individuals who were not previously employees or directors of the Company, as an inducement material
to the individual’s entry into employment with the Company within the meaning of Rule 5635(c)(4) of the Nasdaq Listing Rules.
2020 Equity Incentive Plan
In November 2020, the stockholders of the Company approved the 2020 Equity Incentive Plan (the “2020 Plan”) for stock-based awards. Under the 2020 Plan, (i) 1,000,000 new shares of common stock were reserved for issuance and (ii) up to 70,325 additional shares of common stock may be issued, consisting of (A) shares that remain available for the issuance of awards under prior equity plans and (B) shares of
common stock subject to outstanding stock options or other awards covered by prior equity plans that have been cancelled or expire on or after the date that the 2020 Plan became effective. Under the 2020 Plan, the shares reserved automatically
increase on January 1 of each year, for a period of not more than ten years from the date the 2020 Plan is approved by the
stockholders of the Company, commencing on January 1, 2021 and ending on (and including) January 1, 2030, by an amount equal to 5% of
the shares of common stock outstanding as of December 31st of the preceding calendar year. The 2020 Plan permits the grant of incentive and nonstatutory stock options, appreciation rights, restricted stock, restricted stock units, performance stock and cash awards, and other
stock-based awards.
2018 Equity Incentive Plan
Prior to the 2020 Plan, the Company had adopted a 2018 Equity Incentive Plan (the “2018 Plan”) in April 2018 under which 1,175,000 shares of the Company’s common stock were reserved for issuance to employees, directors and consultants. Upon the effective date of the 2020 Plan, no additional shares were available for issuance under the 2018 Plan.
Stock Options
During the years ended December 31, 2023 and 2022, 1,768,116
and 893,305 stock options were granted to officers, directors, employees and consultants, respectively, generally vesting over a
five (5) to forty-eight (48)
month period. The Company recognized $2.5 million and $1.7 million in stock-based compensation expense related to stock options during the years ended December 31, 2023 and 2022, respectively. Stock-based compensation expense during the year ended December 31, 2023 included a one-time charge of $0.4 million attributed to the modification of the Company’s former Chief Executive Officer’s stock options with respect to their exercisability
provisions.
During the years ended December 31, 2023 and 2022, 27,469
and 24,309 stock options were exercised, respectively, with an intrinsic value of $70,000 and $59,000, respectively. The following table
summarizes the Company’s stock option plan activity:
The weighted average
fair value per share of options granted during the years ended December 31, 2023 and 2022 was $2.53 and $2.06, respectively. The Company measures the fair value of stock options with service-based vesting criteria to employees, directors and consultants
on the date of grant using the Black-Scholes option pricing model. The Company does not have adequate history to support an internal calculation of volatility and expected term. As such, the Company has used a weighted average volatility
considering the volatilities of several guideline companies.
For purposes of
identifying similar entities (guideline companies), the Company considered characteristics such as industry, length of trading history, and stage of life cycle. The average expected life of the options was based on the contractual term for
agreements that allow for exercise of vested options through the end of the contractual term upon termination of continuous service, and for all other agreements, was based on the mid‑point between the vesting date and the end of the
contractual term according to the “simplified method” as described in Staff Accounting Bulletin 110. The risk‑free interest rate is determined by reference to implied yields available from U.S. Treasury securities with a remaining term equal to
the expected life assumed at the date of grant. The Company records forfeitures when they occur. The assumed dividend yield was based on the Company’s expectation of not paying dividends in the foreseeable future.
The weighted average assumptions used in the Black-Scholes option pricing model are as follows during the years ended December 31, 2023 and 2022:
During the years ended December 31, 2023 and 2022, 834,818
and 488,621 stock options vested, respectively. The weighted average fair value per share of options vesting during the years ended
December 31, 2023 and 2022 was $2.41 and $3.29,
respectively. During the years ended December 31, 2023 and 2022, 266,433 and 29,788 stock options were forfeited, respectively.
Restricted Stock Units
During the year ended December 31, 2023, the Company granted an aggregate of 936,156 restricted stock units
(“RSUs”), respectively, to certain officers and employees under the 2020 Plan. The weighted average grant date per unit fair value of the RSUs granted during the year ended December 31, 2023 was $3.37. The vesting period of the RSUs range from a six month
to four year period with vesting tranches on a quarterly, semi-annual and annual basis, subject to the recipient’s continued service
on such dates. There were no RSUs granted during the year ended December 31, 2022.
During the year ended December 31, 2023, 33,614 RSUs vested and 100,842 RSUs were forfeited, attributed solely to the departure of the Company’s former Chief Executive Officer. The total expense for the year ended December 31, 2023 related to the RSUs was $0.7 million. A summary of RSU activity is as follows for the year ended December 31, 2023:
Common Stock Issued
for Services
The Company granted common stock for services in the amount
of 72,986 and 74,396
shares of common stock during the years ended December 31, 2023 and 2022, respectively, with a weighted grant date fair value of $3.77
and $2.04 per share, respectively, to board members during those periods, respectively, who elected to receive their board
retainers in the form of stock for services. The stock-based compensation related to these services amounted to $275,000 and $154,000 during the years ended December 31, 2023 and 2022, respectively.
General
Unrecognized
stock-based compensation cost was $6.4 million as of December 31, 2023. The unrecognized stock-based compensation cost is
expected to be recognized over a weighted average period of 1.8 years. As of December 31, 2023, 1,528,003 shares in the aggregate were available for future issuance under the 2020 Plan and Inducement Plan.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Apexian Sublicense Agreement |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2023 | |||
| Apexian Sublicense Agreement [Abstract] | |||
| Apexian Sublicense Agreement |
On January 21, 2020, the Company entered into a sublicense agreement (as amended on June 4, 2020,
the “Apexian Sublicense Agreement”) with Apexian, pursuant to which it obtained exclusive worldwide patent and other intellectual property rights that constitute a Ref-1 Inhibitor program relating to therapeutic applications to treat disorders
related to ophthalmic and diabetes mellitus conditions. The lead compound in the Ref-1 Inhibitor program is APX3330, which the Company intends to develop as an oral tablet therapeutic to treat diabetic retinopathy initially, and potentially
later to treat diabetic macular edema, geographic atrophy and age-related macular degeneration. In connection with the Apexian Sublicense Agreement, the Company issued a total of 891,422 shares of its common stock to Apexian and to certain affiliates of Apexian in calendar year 2020. As a result of the common stock issued pursuant to the Apexian
Sublicense Agreement, Apexian is considered by Ocuphire to be a related party.
The Company also agreed to make one-time milestone payments
under the Apexian Sublicense Agreement for each of the first ophthalmic indication and the first diabetes mellitus indication for the development and regulatory milestones, and once for each of several sales milestones. These milestone payments
include (i) payments for specified developmental and regulatory milestones (including completion of the first Phase 2 trial and the first Phase 3 pivotal trial in the United States, and filing and achieving regulatory approval from the FDA for
the first New Drug Application for a compound) totaling up to $11 million in the aggregate and (ii) payments for specified sales
milestones of up to $20 million in the aggregate, which net sales milestone payments are payable once, upon the first achievement of
such milestone. Lastly, the Company also agreed to make a royalty payment equal to a single-digit percentage of its net sales of products associated with the covered patents under the Apexian Sublicense Agreement. If it is not terminated
pursuant to its terms, the Apexian Sublicense Agreement shall remain in effect until expiration of the last to expire of the covered patents.
None of the milestone
or royalty payments, were triggered or deemed probable as of December 31, 2023.
|
Stockholders' Equity |
12 Months Ended | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||
| Stockholders' Equity [Abstract] | |||||||||||||||||||||
| Stockholders' Equity |
Lincoln Park Purchase
Agreement
On August 10, 2023, the Company entered into a common stock purchase agreement with Lincoln Park Capital Fund, LLC (“Lincoln Park”) for an equity line financing (the “Purchase Agreement”). The Purchase Agreement provides that, subject to the terms and conditions set forth therein, the Company has the sole right, but not the obligation, to direct Lincoln Park to purchase up to $50 million of shares of the Company’s common stock from time to time over the 30-month term of the Purchase Agreement. Concurrently with entering into the Purchase Agreement, the Company also entered into a registration rights agreement with Lincoln Park (the “Registration Rights Agreement”), pursuant to which the Company agreed to register the resale of the shares of the Company’s common stock that have been and may be issued to Lincoln Park under the Purchase Agreement pursuant to a registration statement. Upon the execution of the Purchase Agreement, the Company issued 246,792 shares of the Company’s common stock to Lincoln Park with a fair value of $1.0 million as consideration for its commitment to purchase shares of the Company’s common stock under the Purchase Agreement which was recorded as a component of financing costs in the accompanying statements of comprehensive (loss) income during the year ended December 31, 2023. Lincoln Park has agreed not to cause or engage in any manner whatsoever in any direct or indirect short selling or hedging of the Company’s common stock. In addition to the commitment shares referenced above, a total of 1,300,000 shares of the Company’s common stock were sold under the Purchase Agreement for net proceeds through December 31, 2023 in the amount of $4.5 million. Lastly, the Company incurred issuance costs of $152,000, consisting of investor expense reimbursement and legal costs, during the year ended December 31, 2023 which were recorded as a component of financing costs in the accompanying statements of comprehensive (loss) income during the year ended December 31, 2023. No shares of the Company’s common stock were sold under the Purchase Agreement prior to the third quarter of 2023. Under the Purchase Agreement on any business day selected by the Company, the Company may direct Lincoln Park to purchase up to 50,000 shares of its common stock on such business day (or the purchase date) (a “Regular Purchase”), provided that the closing sale price of the Company’s common stock on Nasdaq on the applicable purchase date is not below $0.25 and subject to other adjustments. A Regular Purchase may be increased to up to (i) 60,000 shares if the closing sale price of the Company’s common stock on Nasdaq is not below $5.00 on the applicable purchase date and (ii) 70,000 shares if the closing sale price of the Company’s common stock on Nasdaq is not below $7.50 on the applicable purchase date. The Company may direct Lincoln Park to purchase shares in Regular Purchases as often as every business day. The purchase price per share for each such Regular Purchase will be equal to the lesser of:
In addition, the Company may also direct Lincoln Park, on any business day on which the Company has submitted a Regular Purchase notice for the maximum amount allowed for such Regular Purchase, to purchase an additional amount of the Company’s common stock (an “Accelerated Purchase”) of up to the lesser of:
The purchase price per share for each such Accelerated Purchase will be equal to 96.5% of the lower of:
The Company may also direct Lincoln Park, on any business day on which an Accelerated Purchase has been completed and all of the shares to be purchased thereunder have been delivered to Lincoln Park in accordance with the Purchase Agreement, to purchase an additional amount of the Company’s common stock (an “Additional Accelerated Purchase”) as described in the Purchase Agreement. The pricing and settlement provisions in the Purchase Agreement result in the recognition of a derivative liability accounted for on a fair value basis under the provisions of ASC 815 - Derivatives and Hedging. A Monte Carlo simulation model was used to estimate future stock pricing and purchase activity to determine the fair value of the derivative liability as of the August 10, 2023 commencement date and again as of December 31, 2023. As of August 10, 2023 and December 31, 2023, the inputs used to determine fair value of the derivative liability included the Company’s Nasdaq closing stock price of $4.14 and $3.01 per share, respectively, a stock volatility rate of 82.5% and 77.5%, respectively, an expected term of 2.5 years and 2.1 years, respectively, and a risk-free interest rate of 4.6% and 4.2%, respectively. Lastly, the fair value of the derivative liability took into account future purchase decisions based on economic considerations and relevant stock issuance rules/limitations. The fair value change in the derivative liability was recorded in the fair value change in derivative liabilities line item in the accompanying statements of comprehensive (loss) income during the year ended December 31, 2023. At-The-Market
Program
On February 4, 2021, Ocuphire filed a Form S-3 shelf registration under the Securities Act of 1933 which was declared effective by the SEC on February 12, 2021 (the “2021 Shelf”) under which the
Company may offer and sell, from time to time in its sole discretion, securities having an aggregate offering price of up to $125
million. In connection with the 2021 Shelf, on March 11, 2021, Ocuphire entered into a sales agreement with JonesTrading Institutional Services LLC (“JonesTrading”) under which the Company may offer and sell, from time to time at its sole
discretion, to or through JonesTrading, acting as agent and/or principal, shares of its common stock having an aggregate offering price of up to $40
million (the “2021 ATM”). During the years ended December 31, 2023 and 2022, 1,417,446 and 1,848,980 shares of common stock were sold under the ATM for aggregate gross proceeds in the amount of $4.7 million and $4.4 million, respectively, before
deducting issuance expenses, including the placement agent’s fees, legal and accounting expenses, in the amount of $136,000 and $133,000, respectively. See Note 13 – Subsequent Events.
Registered Direct Offering
On June 4, 2021, the Company entered into a placement agency agreement for a registered direct offering (“RDO”) with A.G.P./Alliance Global Partners (“AGP”). Pursuant to the terms of the
placement agency agreement, AGP on June 8, 2021 sold an aggregate of 3,076,923 shares of the Company’s common stock and warrants to purchase 1,538,461 shares of the Company’s common stock (the “RDO Warrants”) at an offering price of $4.875 per one share and 0.50 RDO Warrants, for gross proceeds of approximately $15.0 million, before AGP’s fees and related offering expenses in the amount of approximately $1.1 million. The proceeds were allocated between the relative fair values of common stock and warrants at the sale date. The purchase agreement contains customary representations, warranties and agreements by the
Company, customary conditions to closing, indemnification obligations of the Company, other obligations of the parties and termination provisions. The RDO was made pursuant to the Company’s 2021 shelf registration.
The RDO Warrants have an exercise price of $6.09 per share, are exercisable from the
initial issuance date of June 8, 2021, and will expire five years following the initial issuance date. As of December 31, 2023, 1,538,461 RDO Warrants were outstanding.
Subject to limited
exceptions, a holder of a RDO Warrant will not have the right to exercise any portion of its RDO Warrants if the holder, together with its affiliates, would beneficially own in excess of 4.99% (or, at the election of a holder prior to the date of issuance, 9.99%)
of the number of shares of the Company’s common stock outstanding immediately after giving effect to such exercise; provided, however, that upon prior notice to the Company, the holder may increase or decrease the beneficial ownership
limitation, provided further that in no event shall the beneficial ownership limitation exceed 9.99%.
Pre-Merger Financing
On June 17, 2020, Ocuphire, Rexahn and certain investors entered into a Securities Purchase Agreement, which was amended and restated in its entirety on June 29, 2020 (as amended and restated, the “Securities Purchase Agreement”). Pursuant to the Securities Purchase Agreement, the investors invested a total of $21.15 million in cash, including $300,000 invested by five directors of Ocuphire Pharma, Inc., prior to the Merger and one director of Rexahn upon closing of the Merger (the “Pre-Merger Financing”). The Pre-Merger Financing also included the issuance of Series A Warrants and Series B Warrants discussed further below. Series A Warrants The Series A Warrants were issued on November 19, 2020 at an initial exercise price of $4.4795 per share, were immediately exercisable upon issuance and have a term of five years from the date of issuance. The Series A Warrants are exercisable for 5,665,838 shares of common stock in the aggregate (without giving effect to any limitation on exercise contained therein) and were outstanding as of December 31, 2023. The Series A Warrants were accounted for and classified as equity on the accompanying balance sheets. Series B Warrants The Series B Warrants had an exercise price of $0.0001, were exercisable upon issuance and would have expired on the day following the later to occur of (i) the Reservation Date (as defined therein) or (ii) the date on which the investor’s Series B Warrants would have been exercised in full (without giving effect to any limitation on exercise contained therein). None of the Series B Warrants were outstanding as of December 31, 2023. During the year ended December 31, 2023 and 2022, 17,869 and 60,832 warrants were exercised for shares of common stock, respectively. The Series B Warrants were accounted for and classified as equity on the accompanying balance sheets while outstanding. |
License and Collaboration Agreements |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| License and Collaboration Agreements [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| License and Collaboration Agreements |
9. License and Collaboration Agreements
Viatris License Agreement
On November 6, 2022, the
Company entered into the Viatris License Agreement, pursuant to which it granted Viatris (as successor to Famy) an exclusive, perpetual, sub-licensable license to develop, manufacture, import, export and commercialize (i) PS, for treating (a)
reversal of mydriasis, (b) night vision disturbances or dim light vision, and (c) presbyopia, and (ii) PS and low dose pilocarpine for treating presbyopia (together, the “PS Products”) worldwide except for certain countries and jurisdictions in
Asia (the “Viatris Territory”). The Company retains the exclusive right to develop, manufacture, have manufactured, import, export and commercialize the PS Products outside of the Viatris Territory.
Under the terms of the Viatris License Agreement, the Company
in partnership with Viatris, will develop the PS Products in the United States. Viatris will reimburse the Company for budgeted costs related to the development of the PS Products through FDA approval. Viatris will be responsible for developing the
PS Products in countries and jurisdictions in the Viatris Territory outside of the United States. The parties established a joint steering committee, which oversees and makes decisions regarding the development of the PS Products. The committee is
composed of an equal number of representatives of Viatris and Ocuphire. Viatris will commercialize the PS Products in the Viatris Territory for each indication that receives regulatory approval.
Pursuant to the Viatris License Agreement, the Company received a one-time
non-refundable cash payment of $35 million in November 2022 for the exclusive, perpetual, sub-licensable license to develop, manufacture,
import, export and commercialize the PS Products in the Viatris Territory. In addition, with respect to the PS Products, the Company will be eligible to receive potential additional payments of up to $130 million in the aggregate upon achieving certain specified regulatory or net sales milestones, with the first milestone payment of $10 million to be made following approval by the FDA of PS, for reversal of mydriasis which occurred during the third quarter of 2023. The Company will also receive tiered
royalties, starting at low double-digit royalties up to low 20% royalties, based on the aggregate annual net sales of all PS Products in
the United States, and will receive low double-digit royalties based on all annual net sales in the Viatris Territory outside of the United States. The royalty payments will continue on a country-by-country basis from the date of the first
commercial sale of the first PS Product in a country of the Viatris Territory until December 31, 2040.
Either party may terminate the Viatris License Agreement upon written notice in the case of the other party’s material breach (subject to applicable
cure periods) or if the other party becomes subject to an insolvency event. In addition, the Company may terminate the agreement in its entirety if Viatris or its affiliates commences an action challenging the validity, enforceability or scope of
any of Ocuphire’s patents that are exclusively licensed under the Viatris License Agreement. Additionally, if Viatris determines not to pursue development or commercialization of a PS Product in a country or jurisdiction in the Viatris Territory,
Viatris may terminate the license with respect to such PS Product in such country or jurisdiction.
Both Ocuphire and Viatris have agreed to indemnify the other party against certain losses and expenses relating to any breach of the indemnifying
party’s obligations, representations, warranties or covenants under the Viatris License Agreement.
The Viatris License Agreement was accounted for under the provisions of ASC 606. In accordance with the provisions under ASC 606, the Company identified two distinct performance obligations at the effective date: (1) the license to its intellectual property (“license transfer”) and
(2) research and development services.
The aggregate transaction price associated with the Viatris License Agreement, as adjusted for variable consideration subsequent to December 31, 2022, was $40.0 million which comprised the initial license transfer fee of $35.0 million and the $5.0 million payment anticipated under the research and development services that
were not subject to cancellation. The transaction price was allocated between performance obligations based on their relative standalone selling price (“SSP”). The performance obligations for research and development services through the
non-cancellation period were fully met by the Company as of the first quarter of 2023.
The SSP for the license transfer and for the research and development services was determined to be $ 287.8 million and $5.0 million, respectively. The SSP for the
license transfer was determined based on a
discounted royalty cash flow approach, taking into consideration assumptions, including projected worldwide net profit for each of the respective programs based on probability assessments,
projections based on internal forecasts, industry data, and information from other guideline companies within the same industry and other relevant factors. The SSP for the research and development services was determined using a cost-plus margin
approach, based on anticipated expenditure outlays within the first 120-day non-cancellation window. On a relative SSP basis, $39.3 million and $0.7 million of the
transaction price was allocated to the license transfer and to the research and development services obligations, respectively.
The Company determined that the licenses transferred represented functional intellectual property. As such, the revenue related to the licenses was recognized at the point in time in
which the license/know-how was delivered to Viatris which occurred during the fourth quarter of 2022. The Company determined that revenue related to the research and development services constrained to the 120-day non-cancellation period was to be recognized over time as the services are rendered based on an estimated percentage of completion input model.
Recognition of Revenue
On September 25, 2023, the Company met the $10 million milestone payment
requirements attributed to the FDA’s approval of PS, for reversal of mydriasis and included the milestone in the revenue recognized during the year ended December 31, 2023. The $10 million milestone payment was previously constrained by the Company with regard to its inclusion in the initial aggregate transaction price associated with the Viatris
License Agreement. During the year ended December 31, 2022, the licenses transferred to Viatris represented functional intellectual property. As such, the revenue related to the licenses was recognized at the point in time in which the
license/know-how was delivered to Famy which occurred during the fourth quarter of 2022. The Company determined that revenue related to the research and development services was to be recognized over time as the services are rendered based on an
estimated percentage of completion input model.
Revenue recognized under the Viatris License Agreement during the years ended December 31, 2023 and 2022 was $19.0 million and $ million, respectively.
Regulatory Milestones under the Viatris
License Agreement
The Company has evaluated the regulatory milestones that may be received in connection with the Viatris License Agreement. There is uncertainty that
the events to obtain the remaining regulatory milestones (aside from the approval by the FDA of PS, for reversal of mydriasis) will be achieved given the nature of clinical development and the stage of the development of the PS Products. These
remaining regulatory milestones will be constrained until it is probable that a significant revenue reversal will not occur.
Sales Milestone and Royalty Payments
Sales milestones and royalties relate predominantly to a license of intellectual property granted to Viatris and are determined by sales or
usage-based thresholds. The sales milestones and royalties are accounted for under the royalty recognition constraint and will be accounted for as constrained variable consideration. The Company applies the royalty recognition constraint for each
commercial milestone and will not recognize revenue for each until the subsequent sale of a licensed product (achievement of each) occurs.
Each of the remaining regulatory and sales milestone performance obligations (aside from the $10 million milestone payment related to the FDA’s approval of PS, for reversal of mydriasis) and the royalty payments were fully constrained as of December 31, 2023 and no revenue was recognized.
A reconciliation of the closing balance of the contract assets and unbilled receivables associated with the Viatris License Agreement is as follows as of December 31,
2023 and 2022 (in thousands):
The remaining amounts in contract assets and unbilled receivables as of December 31, 2023 attributed to the research and development services are expected to be settled
during the first quarter of 2024.
BioSense License and Assignment Agreement
On March 10, 2020, prior to the Merger, Rexahn entered into an amendment to its collaboration and license agreement, (as amended, the “BioSense
License and Assignment Agreement”) with BioSense to advance the development and commercialization of RX-3117 for all human uses in the Republic of Singapore, China, Hong Kong, Macau, and Taiwan (the “BioSense Territory”). Under the terms of the
BioSense License and Assignment Agreement, the Company (i) granted BioSense an exclusive license to develop and commercialize pharmaceutical products containing RX-3117 as a single agent for all human uses in the BioSense Territory and (ii)
assigned and transferred all of the former Rexahn patents and patent applications related to RX-3117 in the BioSense Territory. The upfront payment consisted of an aggregate of $1,650,000, of which $1,550,000 was paid to Rexahn prior to the
Merger and the remaining $100,000 during calendar year 2021.
Under the BioSense
License and Assignment Agreement, the Company is eligible to receive additional milestone payments in an aggregate of up to $84,500,000
upon the achievement of development, regulatory and commercial goals and will also be eligible to receive tiered royalties at low double-digit rates on annual net sales in the BioSense Territory. The Company determined that none of the milestone
payments under the BioSense License and Assignment Agreement were probable of payment as of December 31, 2023, and as a result, no
revenue related to the milestones was recognized as the achievement of events entitling the Company to any milestone payments were highly susceptible to factors outside of the Company’s control. Future sales-based royalties related to the
exclusive license to develop RX-3117 will be recognized in the period the underlying sales transaction occurs.
Payments received
under the BioSense License and Assignment Agreement are subject to the CVR Agreement described in Note 2 – Merger.
Processa License Agreement
On June 16, 2021, the
Company entered into a license agreement (the “Processa License Agreement”) with Processa Pharmaceuticals, Inc. (“Processa”), pursuant to which the Company has agreed to grant Processa an exclusive license to develop, manufacture and
commercialize RX-3117 globally, excluding the BioSense Territory.
Processa will make future payments to the Company upon the achievement of certain development and regulatory milestones, which primarily consist
of dosing a patient in pivotal trials or having a drug indication approved by a regulatory authority in the United States or another country. In addition, Processa will pay the Company mid-single-digit percentage royalties based on annual sales
under the license and will make one-time sales milestone payments based on the achievement during a calendar year of certain
thresholds for annual sales. Processa is also required to give the Company 32% of any milestone payments received based on any
sub-license agreement Processa may enter into with respect to the Processa License Agreement.The Company determined that none of the milestone payments under the Processa License Agreement were probable of payment as of December 31, 2023, and as
a result, no revenue related to the milestones was recognized, as the achievement of events entitling the Company to any milestone
payments were highly susceptible to factors outside of the Company’s control.
Processa is required to use commercially reasonable efforts, at its sole cost and expense, to conduct development activities in one or more countries, including meeting specific diligence milestones that consist of: (i) first patient administered drug in a clinical trial of a
licensed product prior to the three (3) year anniversary of the effective date; and (ii) first patient administered drug in a pivotal
clinical trial of a licensed product or first patient administered drug in a clinical trial for a second indication of a licensed product prior to the five (5) year anniversary of the effective date. Either party may terminate the agreement in the event of a material breach of the agreement that has not been cured following written notice and a 120-day opportunity to cure such breach, and Processa may terminate the agreement for any reason upon 120 days prior written notice to Ocuphire.
Future payments
received under the Processa License Agreement will be subject to the CVR Agreement described in Note 2 – Merger.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Net (loss) income per share |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net (loss) income per share [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net (loss) income per share |
Basic (loss) income per share of common stock is computed by dividing net (loss) income by the weighted average number of shares of common stock outstanding during
the period. Diluted earnings or loss per share of common stock is computed similarly to basic loss or earnings per share except the weighted average shares outstanding are increased to include additional shares from the assumed exercise of any
common stock equivalents, if dilutive. The Company’s warrants, stock options and RSUs, while outstanding, are considered common stock equivalents for this purpose. Diluted earnings is computed utilizing the treasury method for the warrants,
stock options and RSUs. Incremental common stock equivalents that were antidilutive were excluded in calculating diluted income per share. For the year ended December 31, 2023, no common stock equivalents were included in the diluted loss per
share because such inclusion would be anti-dilutive given the net loss reported for the prior year period.
The following table presents the computation of weighted average common shares considered in the computation of
diluted net (loss) income per share:
The following potential common shares were not
considered in the computation of diluted net (loss) income per share as their effect would have been anti-dilutive
for the year end periods presented below:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Income Taxes |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Taxes [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Taxes |
The effective tax rate for the years ended December 31, 2023 and 2022 was 0.1
percent and 1.7 percent , respectively.
A reconciliation of income tax computed at the statutory federal income tax rate to the provision (benefit) for income taxes included in the accompanying statements of
comprehensive (loss) income is as follows for the years ended December 31, 2023 and 2022:
The components of income tax provision (benefit) consisted of the following for the years ended December 31, 2023 and 2022 (in thousands):
Significant components of the Company’s deferred tax assets and liabilities are summarized in the tables below as of December 31, 2023 and 2022 (in thousands):
As of December 31, 2023 and 2022, the Company had gross deferred tax assets of approximately $20.1 million and $18.4 million, respectively. Realization of the deferred tax
assets is primarily dependent upon future taxable income, if any, the amount and timing of which are uncertain. The Company has cumulative pre‑tax losses and faces significant challenges to becoming profitable in the future. Accordingly, the net
deferred tax assets have been fully offset by a valuation allowance of $20.1 million and $17.8 million as of December 31, 2023 and 2022, respectively. U.S. net deferred tax assets will continue to require a valuation allowance until the Company can demonstrate their
realizability through sustained profitability or another source of income.
As of December 31, 2023 and 2022, the tax effect of the Company’s federal net operating loss carryforwards was approximately $ 10.6 million and $10.9 million,
respectively. The Company had federal research credit carryforwards as of December 31, 2023 and 2022 of approximately $ 1.1 million and
$0.7 million, respectively. The federal net operating loss carryforwards will not expire and the tax credit carryforwards will begin to
expire in 2041 if not utilized. As of December 31, 2023 and 2022, the Company had state net operating loss carryforwards with a tax effect of approximately $ 2.1 million and $2.2 million, respectively. The Company did not have any state research credit carryforwards as of December 31, 2023 and 2022. The state net operating loss carryforwards will begin to expire in 2028.
During
the year ended December 31, 2023, the Company utilized federal and state net operating tax carryforwards with a tax effect in the amount of $0.2
million and $0.1 million, respectively, to offset taxable income. In addition, the Company also utilized its federal research credit
carryforwards in the amount of $26,000 to partially offset its tax liability for the year ended December 31, 2023.
During the year ended December 31, 2022, the Company utilized federal and state net operating tax carryforwards with a tax effect in the amount of $4.8 million and $1.4 million, respectively, to offset taxable
income. In addition, the Company also utilized its federal research credit carryforwards in the amount of $0.9 million to partially
offset its tax liability for the year ended December 31, 2022.
Utilization of the net operating loss carryforwards and credits may be subject to a substantial annual limitation due to the ownership change limitations provided by
Section 382 and Section 383 of the Internal Revenue Code of 1986, as amended, and similar state provisions. Generally, in addition to certain entity reorganizations, the limitation applies when one or more “5-percent shareholders” increase their
ownership, in the aggregate, by more than 50 percentage points over a 3 year testing period, or beginning the day after the most recent ownership change, if shorter. The annual limitation may result in the expiration of net operating losses and
credits before utilization. As a result of the Merger, the Company recorded deferred tax assets of $10.3 million relating to net
operating loss carryforwards which were fully offset by a valuation allowance. The $10.3 million net deferred tax assets recorded in
relation to the Merger did not include federal and state net operating loss carryforwards that were estimated to expire under Internal Revenue Code Sections 382 as a result of the Merger. The Company has not yet evaluated the impact of Section 382
and Section 383 on its remaining tax attributes that were generated by Ocuphire since the formation of the Company in 2018.
The Company recognizes interest and/or penalties related to uncertain tax positions in income tax expense. In accordance with ASC 740, Income Taxes, specifically related to uncertain tax positions, a Company is required to use a recognition threshold and a measurement attribute for the financial statement recognition and measurement of tax positions taken or expected to be taken
in a tax return. For those benefits to be recognized, a tax position must be more likely than not to be sustained upon examination by taxing authorities. The
Company believes its income tax filing positions and deductions will be sustained upon examination, and accordingly, no reserves or
related accruals for interest and penalties have been recorded at December 31, 2023 and 2022.
The Company’s corporate returns are subject to examination beginning with the 2019 tax year for federal income tax purposes and 2018 for state income tax purposes.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Deferred Compensation Plan |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2023 | |||
| Deferred Compensation Plan [Abstract] | |||
| Deferred Compensation Plan |
Effective October 1, 2021, the Company began offering a 401(k) plan (“401K Plan”) to its
employees. All employees are eligible to participate in the 401K Plan. The Company makes matching contributions equal to 100% on the
first 3% of compensation that is deferred as an elective deferral and an additional 50% on the next 2% of compensation. The Company’s matching
contributions are made on a monthly basis. During the years ended December 31, 2023 and 2022, the Company contributed $99,000 and $76,000 to the 401K Plan, respectively.
|
Subsequent Events |
12 Months Ended | ||
|---|---|---|---|
Dec. 31, 2023 | |||
| Subsequent Events [Abstract] | |||
| Subsequent Events |
Under the 2020 Plan, the shares reserved automatically increase on January 1 of each year, for a period of not more than ten years from the date the 2020 Plan is approved by the stockholders of the Company, commencing on January 1, 2021 and ending on (and including)
January 1, 2030, by an amount equal to 5% of the shares of common stock outstanding as of December 31 of the preceding calendar year.
Notwithstanding the foregoing, the Board of Directors may act prior to January 1 of a given year to provide that there will be no January 1 increase in the share reserve for such year or that the increase in the share reserve for such year will
be a lesser number of shares of common stock than would otherwise occur pursuant to the preceding sentence. On January 1, 2024, 1,198,875
shares were added to the 2020 Plan as a result of the evergreen provision.
On February 12, 2024,
the Company issued a total of 435,000 stock options and 215,000 restricted stock units under the Inducement Plan to its newly appointed Chief Financial and Chief Scientific and Development Officers. The option awards have an exercise price
of $2.66 per share. The options vest over a period of four years, with 25% vesting one year after the date of grant and the
remaining 75% vesting in equal quarterly installments thereafter, and the RSUs vest in four equal installments on the first, second, third and fourth anniversary of the grant date of February 12, 2024
On January 10, 2024, the Company filed a Form S-3 shelf registration under
the Securities Act which was declared effective by the SEC on January 23, 2024 under which the Company may offer and sell, from time to time in its sole discretion, securities having an aggregate offering price up to $175 million.
|
Insider Trading Arrangements |
3 Months Ended |
|---|---|
Dec. 31, 2023 | |
| Insider Trading Arrangements [Line Items] | |
| Rule 10b5-1 Arrangement Adopted | false |
| Non-Rule 10b5-1 Arrangement Adopted | false |
| Rule 10b5-1 Arrangement Terminated | false |
| Non-Rule 10b5-1 Arrangement Terminated | false |
Company Description and Summary of Significant Accounting Policies (Policies) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company Description and Summary of Significant Accounting Policies [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nature of Business |
Nature of Business
Ocuphire Pharma, Inc. (the “Company” or “Ocuphire”) is a
clinical-stage biopharmaceutical company focused on developing novel therapies for the treatment of unmet needs of patients with retinal and refractive eye disorders. The Company’s headquarters is located in Farmington Hills,
Michigan.
The Company’s lead retinal product candidate, APX3330, is a small-molecule inhibitor of Ref-1 (reduction oxidation effector factor-1 protein).
Ref-1 is a regulator of transcription factors such as HIF-1α and NF-kB. Inhibiting Ref-1 reduces levels of vascular endothelial growth factor (“VEGF”) and inflammatory cytokines which are known to play key roles in ocular angiogenesis and
inflammation. APX3330 is an oral tablet administered once or twice per day in development for the treatment of diabetic retinopathy (“DR”). A
Phase 2 study in subjects with DR or diabetic macular edema was completed and results were reported in January 2023. An End-of-Phase 2 (“EOP2”) meeting with the U.S. Food and Drug Administration (the “FDA”) was held in October 2023 at
which the Company obtained agreement on the registration endpoint supporting the advancement of APX3330 into future clinical trials. Ocuphire submitted a Special Protocol
Assessment (“SPA”) to the FDA in February 2024 to seek agreement on the clinical trial protocol and statistical analysis plan.
The Company has also in-licensed APX2009 and APX2014, which are second-generation analogs of APX3330. The unique mechanism of action of this family of Ref-1 inhibitors of reducing
angiogenesis and inflammation could potentially be beneficial in treating other retinal diseases such as age-related macular degeneration, geographic atrophy, and non-ophthalmic diseases.
In November 2022, the Company entered into a license and collaboration agreement (the “Viatris License Agreement”) with FamyGen Life Sciences, Inc. (“Famy”) (acquired by Viatris, Inc.
(“Viatris”) in January 2023) pursuant to which it granted Viatris an exclusive license to develop, manufacture, import, export and commercialize its refractive product candidate Phentolamine Ophthalmic Solution 0.75%, formerly known as Nyxol
(“PS”). PS is a once-daily eye drop formulation of phentolamine mesylate designed to reduce pupil diameter and improve visual acuity. PS was approved by the FDA for the treatment for pharmacologically-induced mydriasis under the brand name
RYZUMVI in September 2023. The VEGA-2 Phase 3 study in presbyopia achieved its primary endpoint. PS is currently in Phase 3 clinical trials for presbyopia (age-related blurry near vision). On December 5, 2023, the Company received FDA
Agreement Under Special Protocol Assessment for LYNX-2, a Phase 3 Trial of PS for the treatment of decreased Visual Acuity under dim (mesopic) light conditions following keratorefractive surgery.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reverse Merger with Rexahn |
Reverse Merger with Rexahn
On June 17, 2020, Ocuphire, Rexahn
Pharmaceuticals, Inc. (“Rexahn”) and Razor Merger Sub, Inc., a Delaware corporation and wholly-owned subsidiary of Rexahn (“Merger Sub”), entered into an Agreement and Plan of Merger and Reorganization, as amended on June 29, 2020 (as
amended, the “Merger Agreement”), pursuant to which, among other things, and subject to the satisfaction or waiver of certain conditions set forth in the Merger Agreement, Merger Sub merged with and into Ocuphire, with Ocuphire continuing
as a wholly-owned subsidiary of Rexahn and the surviving corporation of the merger (the “Merger”). The Merger closed on November 5, 2020. Upon completion of the Merger, Rexahn changed its name to Ocuphire Pharma, Inc. and changed its ticker
symbol on the Nasdaq Capital Market (“Nasdaq”) to “OCUP”.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Basis of Presentation |
Basis of Presentation
The accompanying financial statements have been prepared in accordance with accounting standards generally accepted in the United States of America (“GAAP”).
The Company does not have any subsidiaries or other entities that require consolidation for financial statement reporting purposes.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liquidity |
Liquidity
The accompanying financial statements have been prepared
on the basis that the Company will continue as a going concern. From its inception, the Company has devoted substantially all of its efforts to drug development and conducting clinical trials.
As of
December 31, 2023, the Company had $50.5 million in cash and cash equivalents. The Company
believes its current available cash and cash equivalents will be sufficient to fund the Company’s planned expenditures and meet its obligations for at least twelve months from the date of issuance of these financial statements.
In the future, the Company may need to raise additional funds until it is able to generate sufficient revenues to fund its development activities. The Company’s future
operating activities, coupled with its plans to raise capital or issue debt financing, may provide additional liquidity in the future, however these actions are not solely within the control of the Company and the Company is unable to
predict the outcome of these actions to generate the liquidity ultimately required.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Use of Estimates |
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the amounts reported in the
financial statements and accompanying notes. Actual results could differ from those estimates.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Segment Information |
Segment Information
Operating segments are components of an enterprise for which separate financial information is available and is evaluated regularly by the Company’s chief operating
decision maker in deciding how to allocate resources and assessing performance. The Company’s chief operating decision maker is its Chief Executive Officer or such person functioning in such role. The Company’s Chief Executive Officer views the
Company’s operations and manages its business in one operating segment, which is the business of development of products related to
vision performance and health. Accordingly, the Company has a single reporting segment.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash and Cash Equivalents |
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of 90 days or less at the time of deposit to be cash equivalents.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concentration of Credit Risk |
Concentration of Credit Risk
Financial
instruments that potentially subject the Company to a concentration of credit risk consist of cash and cash equivalents. Management follows approved policies established by its Board of Directors to reduce credit risk associated with the
Company’s cash deposit and investment accounts. Pursuant to these policies, the Company limits its exposure through the kind, quality and concentration of its investments. The Company’s cash and cash equivalents are held or managed by two
financial institutions in the United States. As of December 31, 2023, the Company had cash equivalents of $50.2 million that
were not eligible for coverage by Federal Deposit Insurance Corporation (“FDIC”). These balances are invested in funds whose assets consist almost entirely of securities issued by the U.S. Treasury or guaranteed by the U.S. government.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Short-term Investments |
Short-term Investments
The Company determines the appropriate classification of its investments in debt and equity securities at the time of purchase and records them on a settlement
date basis. The Company’s short-term investments are comprised of equity securities, which in accordance with the fair value hierarchy described below are recorded at fair value using Level l inputs on the balance sheets. Subsequent changes
in fair values are recorded in other income (expense), net on the statements of comprehensive (loss) income. The Company classifies investments available to fund current operations as current assets on its balance sheets. The Company did not recognize any impairments on its investments to date through December 31, 2023.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revenue Recognition |
Revenue Recognition
The Company follows the provisions of Accounting Standards Codification (“ASC”) 606, Revenue from Contracts with Customers. The guidance provides a five-step model to determine
how revenue is recognized. The Company has entered into license agreements which have revenue recognition implications (See Note 9 – License and Collaboration Agreements).
In determining the appropriate amount of revenue to be recognized, the Company performs the following steps: (i) identification of the contracts with a
customer; (ii) determination of the performance obligations in the contract; (iii) measurement of the transaction price, including potential constraints on variable consideration; (iv) allocation of the transaction price to the performance
obligations based on estimated stand-alone selling prices; and (v) recognition of revenue when (or as) the Company satisfies a performance obligation.
A performance obligation is a promise in a contract to transfer a distinct good or service to
the customer and is the unit of account in ASC 606. Performance obligations may include license rights, development and other services. Significant management judgment is required to determine the level of effort required under an
arrangement and the period over which the Company expects to complete its performance obligations under the arrangement. If the Company cannot reasonably estimate when its performance obligations are either completed or become
inconsequential, then revenue recognition is deferred until the Company can reasonably make such estimates. Revenue is then recognized over the remaining estimated period of performance using the cumulative catch-up method.
As part of the accounting for these arrangements, the Company must develop assumptions that require judgment to determine the stand-alone selling price of each
performance obligation identified in the contract. The Company uses key assumptions to determine the stand-alone selling price, which may include forecasted revenues, development timelines, reimbursement rates for personnel costs, discount
rates and probabilities of technical and regulatory success. The Company allocates the total transaction price to each performance obligation based on the relative standalone selling prices of the promised goods or service underlying each
performance obligation.
Licenses of intellectual property and research and development services: If the license to the
Company’s intellectual property is determined to be distinct from the other performance obligations identified in the arrangement, the Company recognizes revenues from non-refundable, up-front fees allocated to the license when the license
is transferred to the customer, and the customer can use and benefit from the license. For licenses that are bundled with other obligations, such as research and development services, the Company utilizes judgment to assess the nature of
the combined performance obligation to determine whether the combined performance obligation is satisfied over time or at a point in time and, if over time, the appropriate method of measuring progress for purposes of recognizing revenue
from non-refundable, up-front fees. For research and development services that are distinct from a license transfer obligation, the Company determines whether the services are satisfied over time or at a point in time and, if over time, the
appropriate method of measuring progress for purposes of recognizing revenue from such services. The Company evaluates the measure of progress each reporting period and, if necessary, adjusts the measure of performance and related revenue
recognition.
Milestone payments: At the inception of each arrangement
that includes milestone payments, the Company evaluates whether the milestones are considered probable of being achieved and estimates the amount to be included in the transaction price using the most likely amount method. If it is probable
that a significant revenue reversal would not occur, the value of the associated milestone (such as a regulatory submission) is included in the transaction price. Milestone payments that are not within the control of the Company, such as
approvals from regulators, are not considered probable of being achieved until such contingency occurs (such as receipt of those approvals).
Royalties: For arrangements that include sales-based royalties, including milestone payments
based on the level of sales, and the license is deemed to be the predominant item to which the royalties relate, the Company recognizes revenue at the later of (a) when the related sales occur, or (b) when the performance obligation to
which some or all of the royalty has been allocated has been satisfied (or partially satisfied).
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Contract Assets and Unbilled Receivables |
Contract Assets and Unbilled Receivables
The Company recognizes contract assets and unbilled receivables when goods or services are transferred to the customer before the customer pays or before reimbursement for payment is
billed or due, excluding any amounts presented as an account receivable. The Company recorded contract assets and unbilled receivables in connection with a license and collaboration agreement (See Note 9 – License and Collaboration Agreements).
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accounts Receivable and Allowances for Credit Losses |
Accounts Receivable and Allowances for Credit Losses
The Company records a provision for credit losses, when
appropriate, based on historical experience, current conditions and reasonable supportable forecasts. In estimating the allowance for credit losses, the Company considers, among other factors, the estimate of credit losses over
the remaining expected life of the asset, primarily using historical experience and current economic conditions that could affect the collectability of the balances in the future. Account balances are charged off against the
allowance when the Company believes that it is probable that the receivable will not be recovered. Actual write-offs may be in excess of the Company’s estimated allowance. The Company has not incurred any bad debt expense to date and no allowance for credit losses has been recorded during the periods presented.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General and Administrative Expenses |
General and Administrative Expenses
General and administrative expenses consist primarily of personnel-related costs, including salaries, benefits and stock-based compensation costs, for personnel in
functions not directly associated with research and development activities. Other significant costs include insurance coverage for directors and officers and other property and liability exposures, legal fees relating to intellectual property and
corporate matters, professional fees for accounting and tax services, other services provided by business consultants and legal settlements.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Research and Development |
Research and Development
Research and development expenses consist of costs
incurred in performing research and development activities, including compensation, benefits and stock-based compensation costs for research and development employees and costs for consultants, costs associated with nonclinical studies and
clinical trials, regulatory activities, manufacturing activities to support clinical activities, license fees, nonlegal patent costs, fees paid to external service providers that conduct certain research and development, and an allocation of
overhead expenses. Research and development expenses include costs that are reimbursed under the Viatris License Agreement (See Note 9 – License and
Collaboration Agreements).
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financing costs |
Financing costs
Financing costs consist of issuance costs
attributed to an equity line financing facility with Lincoln Park (See Note 8 – Stockholders’ Equity).
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interest Expense |
Interest Expense
Interest expenses were attributed to interest on principal related to a short-term loan during the period it was outstanding. The short-term loan was fully repaid in May 2022. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other Income (Expense), net |
Other Income (Expense), net
Other income (expense), net includes interest earned from cash and cash equivalent investments, realized and unrealized gains (losses) from equity investments and
reimbursements in connection with grants and other sources when they occur. In addition, this line item includes payments made by the Company in connection with the Contingent Value Rights Agreement discussed further below with former Rexahn
shareholders.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-Based Compensation |
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with the provisions of the Financial Accounting Standards Board (“FASB”) ASC 718, Compensation — Stock Compensation. Accordingly, compensation costs related to equity instruments granted are recognized at the grant date fair value. The Company records forfeitures when they occur.
Stock-based compensation arrangements to non-employees are accounted for in accordance with the applicable provisions of ASC 718.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Derivative Liability |
Derivative Liability
The Company evaluates all
features contained in financing agreements to determine if there are any embedded derivatives that require separation from the underlying agreement under ASC 815 – Derivatives and Hedging. An embedded
derivative that requires separation is accounted for as a separate liability from the host agreement. The separated embedded derivative is accounted for separately on a fair market value basis. The Company records the fair value changes of a
separated embedded derivative at each reporting period in the statements of comprehensive (loss) income under the fair value change in derivative liability line item. The Company determined that certain features under an equity line financing
(See Note 8 — Stockholders’ Equity) collectively qualified as an embedded derivative. The derivative was accounted for separately from the underlying equity line financing agreement.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value Measurements |
Fair Value Measurements
The Company follows accounting guidance that emphasizes that fair value is a market-based measurement, not an entity-specific measurement. Fair value is defined as
“the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.” Fair value measurements are defined on a three-level hierarchy:
As of December 31, 2023 and
2022, the fair values of cash and cash equivalents, accounts receivable, contract assets and unbilled receivables, prepaid and other current assets, accounts payable, and accrued expenses approximated their carrying values because of the
short-term nature of these assets or liabilities. The fair value of the short-term investments, while outstanding, were based on observable Level 1 inputs in the form of quoted market prices from a major stock exchange. The fair value of the
derivative liability associated with the equity line financing facility (See Note 8 – Stockholders’ Equity) was based on cash flow models discounted at current implied market rates representing expected returns by market participants for
similar instruments and are based on Level 3 inputs as well the Company’s underlying stock price and associated volatility, expected term of the financing and market interest rates. The fair value of the warrant liabilities, while outstanding, were based on a Black-Scholes option model using Level 3 inputs. There were no transfers between fair value hierarchy levels during the years ended December 31, 2023 and 2022.
The fair value of financial instruments measured on a recurring basis is as follows (in thousands):
The following table provides a roll-forward of short-term investments and derivative liabilities measured at fair value on a recurring basis using observable Level
1 and Level 3 inputs, as applicable, for the years ended December 31, 2023 and 2022 (in thousands):
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rexahn Warrants |
Rexahn Warrants
The fair value of the warrant
liabilities associated with the Rexahn warrants was de minimis during the periods presented. The last of the Rexahn warrants classified as liabilities expired in April 2023 unexercised. See Note 2 – Merger for additional background.
There were no financial instruments measured on a
non-recurring basis for any of the periods presented.
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recent Accounting Pronouncements |
Recent Accounting Pronouncements
In June 2016, the FASB issued Accounting Standards Update (“ASU”)
2016-13, Financial Instruments – Credit Losses. The ASU sets forth a current expected credit loss (CECL) model which requires the Company to measure all expected credit losses for financial instruments
held at the reporting date based on historical experience, current conditions, and reasonable supportable forecasts. This replaces the existing incurred loss model and is applicable to the measurement of credit losses on financial assets
measured at amortized cost and applies to some off-balance sheet credit exposures. The Company adopted this ASU on January 1, 2023 and it did not have a significant impact on its financial statements.
In August 2020, FASB issued ASU 2020-06,
Debt—Debt
with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, which, among other things, provides guidance on how to account for contracts on an entity’s own equity. This ASU eliminates the
beneficial conversion and cash conversion accounting models for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted for as derivatives because of specific
settlement provisions. In addition, this ASU modifies how particular convertible instruments and certain contracts that may be settled in cash or shares impact the diluted earnings per share computation. The amendments in this ASU are
effective for public business entities that meet the definition of an SEC filer, excluding entities eligible to be smaller reporting companies as defined by the SEC, for fiscal years beginning after December 15, 2021, including interim
periods within those fiscal years. For all other entities, the amendments are effective for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. The Company adopted this ASU on January 1, 2023
and the adoption did not have a material impact on its financial statements.
In November 2023, the FASB issued ASU 2023-07 - Segment Reporting (Topic 280): Improvements to Reportable Segment Disclosures, which enhances reportable segment disclosure requirements, primarily through disclosures of significant segment expenses. This ASU is effective for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years, with early adoption permitted. The guidance must be applied retrospectively to all prior periods presented. The Company is currently evaluating the impact of adoption of this guidance on its financial statements. In December 2023, the FASB issued ASU 2023-09 Income Taxes (Topic 740): Improvements to Income Tax Disclosures, which enhances income tax disclosures primarily related to the rate reconciliation and income taxes paid information. This guidance also includes certain other amendments to improve the effectiveness of income tax disclosures. This ASU is effective for fiscal years beginning after December 15, 2024, including interim periods within those fiscal years and should be applied on a prospective basis, with retrospective application permitted. The Company is currently evaluating the impact of adoption of this guidance on its financial statements. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Company Description and Summary of Significant Accounting Policies (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Company Description and Summary of Significant Accounting Policies [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value of Financial Instruments Measured on a Recurring Basis |
The fair value of financial instruments measured on a recurring basis is as follows (in thousands):
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair Value, Investments and Derivative Liabilities Measured on a Recurring Basis |
The following table provides a roll-forward of short-term investments and derivative liabilities measured at fair value on a recurring basis using observable Level
1 and Level 3 inputs, as applicable, for the years ended December 31, 2023 and 2022 (in thousands):
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Supplemental Balance Sheet Information (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supplemental Balance Sheet Information [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prepaid and Other Current Assets |
Prepaid and other current assets consist of the following (in thousands):
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Property and Equipment, Net |
Property and equipment held for use by category are presented in the following table (in thousands):
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Accrued Expenses |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stock-based Compensation (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-based Compensation [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock-Based Compensation Expense |
Stock-based compensation expense was included in general and administrative and research and development costs as follows in the accompanying statements of
comprehensive (loss) income for the periods indicated below (in thousands):
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stock Option Plan Activity | The following table
summarizes the Company’s stock option plan activity:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weighted-Average Assumptions Used in Black-Scholes Option-pricing Model |
The weighted average assumptions used in the Black-Scholes option pricing model are as follows during the years ended December 31, 2023 and 2022:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Restricted Stock Units Activity |
A summary of RSU activity is as follows for the year ended December 31, 2023:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
License and Collaboration Agreements (Tables) |
12 Months Ended | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| License and Collaboration Agreements [Abstract] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reconciliation of Contract Asset assets and Unbilled Receivables |
A reconciliation of the closing balance of the contract assets and unbilled receivables associated with the Viatris License Agreement is as follows as of December 31,
2023 and 2022 (in thousands):
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Net (loss) income per share (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net (loss) income per share [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Computation of Weighted Average Common Shares |
The following table presents the computation of weighted average common shares considered in the computation of
diluted net (loss) income per share:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anti-dilutive Securities Excluded from Computation of Net Income (Loss) per Share |
The following potential common shares were not
considered in the computation of diluted net (loss) income per share as their effect would have been anti-dilutive
for the year end periods presented below:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||
Income Taxes (Tables) |
12 Months Ended | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Dec. 31, 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income Taxes [Abstract] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reconciliation of Statutory to Effective Income Tax Rate |
A reconciliation of income tax computed at the statutory federal income tax rate to the provision (benefit) for income taxes included in the accompanying statements of
comprehensive (loss) income is as follows for the years ended December 31, 2023 and 2022:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of Income Tax Provision (Benefit) |
The components of income tax provision (benefit) consisted of the following for the years ended December 31, 2023 and 2022 (in thousands):
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deferred Tax Assets and Liabilities |
Significant components of the Company’s deferred tax assets and liabilities are summarized in the tables below as of December 31, 2023 and 2022 (in thousands):
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Company Description and Summary of Significant Accounting Policies, Liquidity (Details) - USD ($) $ in Thousands |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Liquidity [Abstract] | ||
| Cash and cash equivalents | $ 50,501 | $ 42,634 |
Company Description and Summary of Significant Accounting Policies, Segment Information (Details) |
12 Months Ended |
|---|---|
|
Dec. 31, 2023
Segment
| |
| Segment Information [Abstract] | |
| Number of operating segments | 1 |
Company Description and Summary of Significant Accounting Policies, Concentration of Credit Risk (Details) $ in Millions |
Dec. 31, 2023
USD ($)
|
|---|---|
| Concentration of Credit Risk [Abstract] | |
| Cash equivalents not eligible for coverage | $ 50.2 |
Company Description and Summary of Significant Accounting Policies, Short-term Investments (Details) $ in Thousands |
12 Months Ended |
|---|---|
|
Dec. 31, 2023
USD ($)
| |
| Other than Temporary Impairment Losses, Investments [Abstract] | |
| Impairments on investments | $ 0 |
Company Description and Summary of Significant Accounting Policies, Contract Assets and Unbilled Receivables and Accounts Receivable and Allowances for Doubtful Accounts (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Contract Assets and Unbilled Receivables and Accounts Receivable and Allowances for Doubtful Accounts [Abstract] | ||
| Allowance for credit loss | $ 0 | $ 0 |
| Bad debt expense | $ 0 |
Company Description and Summary of Significant Accounting Policies, Equity Investments Measured at Fair Value (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Equity Investments [Abstract] | ||
| Unrealized loss | $ (34) | $ (170) |
| Derivative Liability [Abstract] | ||
| Balance as of beginning of period | 0 | |
| Balance as of end of period | 74 | 0 |
| Recurring Basis [Member] | Level 1 [Member] | Short- term investments [Member] | ||
| Equity Investments [Abstract] | ||
| Balance as of beginning of period | 49 | 219 |
| Unrealized loss | (34) | (170) |
| Balance as of end of period | 15 | 49 |
| Recurring Basis [Member] | Level 3 [Member] | ||
| Derivative Liability [Abstract] | ||
| Balance as of beginning of period | 0 | 0 |
| Purchase agreement execution | 154 | 0 |
| Unrealized gain | (80) | 0 |
| Balance as of end of period | $ 74 | $ 0 |
Company Description and Summary of Significant Accounting Policies, Rexahn Warrants (Details) - USD ($) $ in Thousands |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Nonrecurring Basis [Member] | ||
| Fair Value, Liabilities Measured on Recurring Basis, Unobservable Input Reconciliation [Line Items] | ||
| Financial instruments liabilities at fair value | $ 0 | $ 0 |
Merger, Contingent Value Rights Agreement (Details) |
12 Months Ended | |||
|---|---|---|---|---|
|
Dec. 31, 2023
USD ($)
Milestone
|
Sep. 30, 2021
Milestone
|
Jun. 30, 2021
Milestone
|
Nov. 05, 2020
Right
|
|
| Contingent Value Rights Agreement [Abstract] | ||||
| Contingent value rights payment period | 15 years | |||
| Sum of cash consideration paid by a third party | 75.00% | |||
| Parent IP deal period | 10 years | |||
| Number of milestones accrued | 0 | |||
| Number of potential milestones | 0 | 0 | ||
| Payments for CVR | $ | $ 0 | |||
| Rexahn [Member] | ||||
| Contingent Value Rights Agreement [Abstract] | ||||
| Number of contingent value right received per common stock | Right | 1 | |||
| Rexahn [Member] | BioSense Global LLC [Member] | ||||
| Contingent Value Rights Agreement [Abstract] | ||||
| Percentage of payments received by Rexahn or its affiliates | 90.00% | |||
| Rexahn [Member] | Zhejiang HaiChang Biotechnology Co., Ltd [Member] | ||||
| Contingent Value Rights Agreement [Abstract] | ||||
| Percentage of payments received by Rexahn or its affiliates | 90.00% |
Merger, Former Rexahn Warrants (Details) - Rexahn [Member] - $ / shares |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Nov. 05, 2020 |
|
| Former Rexahn Warrants and Stock Options [Abstract] | ||
| Number of warrants outstanding (in shares) | 58,597 | 231,433 |
| Exercise price (in dollars per share) | $ 38.4 | |
| Average remaining contractual life | 1 month 6 days |
Commitments and Contingencies (Details) - USD ($) |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Facility Lease [Abstract] | ||
| Monthly base rent | $ 3,000 | |
| Rent expense | 36,000 | $ 39,000 |
| Expected rent payment for the year end 2024 | $ 36,000 | |
Supplemental Balance Sheet Information, Prepaid and Other Current Assets (Details) - USD ($) $ in Thousands |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Prepaid and Other Current Assets [Abstract] | ||
| Prepaids | $ 997 | $ 1,373 |
| Other | 102 | 80 |
| Total prepaids and other assets | $ 1,099 | $ 1,453 |
Supplemental Balance Sheet Information, Property and Equipment, Net (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Property and Equipment, net [Abstract] | ||
| Total property and equipment | $ 25 | $ 25 |
| Less accumulated depreciation | (25) | (19) |
| Property and equipment, net | 0 | 6 |
| Depreciation expense | 6 | 4 |
| Equipment [Member] | ||
| Property and Equipment, net [Abstract] | ||
| Total property and equipment | 20 | 20 |
| Furniture [Member] | ||
| Property and Equipment, net [Abstract] | ||
| Total property and equipment | $ 5 | $ 5 |
Supplemental Balance Sheet Information, Accrued Expenses (Details) - USD ($) $ in Thousands |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Accrued Expenses [Abstract] | ||
| Income taxes | $ 0 | $ 315 |
| Payroll | 753 | 782 |
| Professional services | 591 | 208 |
| R&D services and supplies | 400 | 212 |
| Other | 71 | 167 |
| Total | $ 1,815 | $ 1,684 |
Supplemental Balance Sheet Information, Short-Term Loan (Details) |
12 Months Ended | |||
|---|---|---|---|---|
|
Dec. 31, 2023
USD ($)
Intallment
|
Dec. 31, 2022
USD ($)
|
Dec. 31, 2021
USD ($)
|
Nov. 30, 2021
USD ($)
|
|
| Short-Term Loan [Abstract] | ||||
| Short-term loan | $ 600,000 | |||
| Number of installments | Intallment | 6 | |||
| Annual interest rate | 5.50% | |||
| Interest expense | $ 0 | $ 9,000 | ||
| Short-term Loan [Member] | ||||
| Short-Term Loan [Abstract] | ||||
| Payment on short term loan | $ 108,000 | |||
Stock-based Compensation, Stock-Based Compensation Expense (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Stock-based Compensation Expense [Abstract] | ||
| Stock-based compensation | $ 3,510 | $ 1,807 |
| General and Administrative [Member] | ||
| Stock-based Compensation Expense [Abstract] | ||
| Stock-based compensation | 2,435 | 1,060 |
| Research and Development [Member] | ||
| Stock-based Compensation Expense [Abstract] | ||
| Stock-based compensation | $ 1,075 | $ 747 |
Stock-based Compensation, Weighted Average Assumptions Used in Black-Scholes Option-pricing Model (Details) - 2018 Equity Incentive Plan [Member] - $ / shares |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Weighted-average Assumptions Used in Black-Scholes Option-pricing Model [Abstract] | ||
| Expected stock price volatility | 96.00% | 97.40% |
| Expected life of options (years) | 6 years 1 month 6 days | 5 years 9 months 18 days |
| Expected dividend yield | 0.00% | 0.00% |
| Risk free interest rate | 4.20% | 2.30% |
| Share-based Compensation Arrangement by Share-based Payment Award [Abstract] | ||
| Stock options vested (in shares) | 834,818 | 488,621 |
| Weighted average fair value per share of options vesting (in dollars per share) | $ 2.41 | $ 3.29 |
| Stock options forfeited (in shares) | 266,433 | 29,788 |
Stock-based Compensation, Restricted Stock Units (Details) - USD ($) $ / shares in Units, $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Restricted Stock Units [Abstract] | ||
| Stock-based compensation | $ 3,510 | $ 1,807 |
| Restricted Stock Units [Member] | ||
| Restricted Stock Units [Abstract] | ||
| Stock-based compensation | $ 700 | |
| Summary of RSU Activity [Abstract] | ||
| Non-vested at beginning (in shares) | 0 | |
| Granted (in shares) | 936,156 | |
| Forfeited (in shares) | (100,842) | |
| Vested (in shares) | (33,614) | |
| Non-vested at ending (in shares) | 801,700 | 0 |
| Restricted Stock Units [Member] | Former Chief Executive Officer [Member] | ||
| Summary of RSU Activity [Abstract] | ||
| Forfeited (in shares) | (100,842) | |
| Vested (in shares) | (33,614) | |
| Restricted Stock Units [Member] | 2020 Equity Incentive Plan [Member] | ||
| Restricted Stock Units [Abstract] | ||
| Weighted average grant date fair value of RSUs granted (in dollars per share) | $ 3.37 | |
| Summary of RSU Activity [Abstract] | ||
| Granted (in shares) | 936,156 | 0 |
| Restricted Stock Units [Member] | 2020 Equity Incentive Plan [Member] | Minimum [Member] | ||
| Restricted Stock Units [Abstract] | ||
| Vesting period | 6 months | |
| Restricted Stock Units [Member] | 2020 Equity Incentive Plan [Member] | Maximum [Member] | ||
| Restricted Stock Units [Abstract] | ||
| Vesting period | 4 years | |
Stock-based Compensation, Common Stock Issued for Services (Details) - USD ($) |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Common Stock Issued for Services [Abstract] | ||
| Granted stock for services performed (in shares) | 72,986 | 74,396 |
| Weighted grant date fair value per share of services granted (in dollars per share) | $ 3.77 | $ 2.04 |
| Share based compensation for services | $ 275,000 | $ 154,000 |
Stock-based Compensation, General (Details) $ in Millions |
12 Months Ended |
|---|---|
|
Dec. 31, 2023
USD ($)
shares
| |
| General [Abstract] | |
| Unrecognized stock-based compensation cost | $ | $ 6.4 |
| Weighted average period to recognized stock-based compensation | 1 year 9 months 18 days |
| 2020 Plan Evergreen Provision [Member] | |
| General [Abstract] | |
| Common stock available for future issuance (in shares) | 1,528,003 |
| 2018 Equity Incentive Plan [Member] | |
| General [Abstract] | |
| Common stock available for future issuance (in shares) | 1,528,003 |
Apexian Sublicense Agreement (Details) - Apexian Sublicense Agreement [Member] - USD ($) $ in Millions |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2020 |
|
| Sublicense Agreement [Abstract] | ||
| Common stock issued (in shares) | 891,422 | |
| Development and Regulatory Milestones [Member] | Maximum [Member] | ||
| Sublicense Agreement [Abstract] | ||
| Milestone payments | $ 11 | |
| Sales Milestones [Member] | Maximum [Member] | ||
| Sublicense Agreement [Abstract] | ||
| Milestone payments | $ 20 | |
Stockholders' Equity, Lincoln Park Purchase Agreement (Details) |
3 Months Ended | 12 Months Ended | ||
|---|---|---|---|---|
|
Aug. 10, 2023
USD ($)
Time
$ / shares
shares
|
Sep. 30, 2023
shares
|
Dec. 31, 2023
USD ($)
shares
|
Dec. 31, 2022
USD ($)
|
|
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Net proceeds | $ 9,227,000 | $ 4,428,000 | ||
| Issuance costs | $ 278,000 | $ 131,000 | ||
| Lincoln Park [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Term of purchase agreement | 30 months | |||
| Consideration shares of common stock (in shares) | shares | 246,792 | 0 | 1,300,000 | |
| Common stock fair value | $ 1,000,000 | |||
| Net proceeds | $ 4,500,000 | |||
| Issuance costs | $ 152,000 | |||
| Number of consecutive trading days | 10 days | |||
| Number of time shares purchased | Time | 3 | |||
| Percentage of common stock | 30.00% | |||
| Percentage of accelerated purchase | 96.50% | |||
| Lincoln Park [Member] | Closing Stock Price [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Measurement input | 4.14 | 3.01 | ||
| Lincoln Park [Member] | Stock Volatility Rate [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Measurement input | 0.825 | 0.775 | ||
| Lincoln Park [Member] | Expected Term [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Measurement input | 2.5 | 2.1 | ||
| Lincoln Park [Member] | Risk Free Interest Rate [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Measurement input | 0.046 | 0.042 | ||
| Lincoln Park [Member] | Maximum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Purchase of common stock | $ 50,000,000 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $0.25 [Member] | Maximum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Purchase of common stock (in shares) | shares | 50,000 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $0.25 [Member] | Minimum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Closing stock price (in dollars per share) | $ / shares | $ 0.25 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $5.00 [Member] | Maximum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Purchase of common stock (in shares) | shares | 60,000 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $5.00 [Member] | Minimum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Closing stock price (in dollars per share) | $ / shares | $ 5 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $7.50 [Member] | Maximum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Purchase of common stock (in shares) | shares | 70,000 | |||
| Lincoln Park [Member] | Closing Sale Price, not Below $7.50 [Member] | Minimum [Member] | ||||
| Lincoln Park Purchase Agreement [Abstract] | ||||
| Closing stock price (in dollars per share) | $ / shares | $ 7.5 | |||
Stockholders' Equity, At-The-Market Program (Details) - USD ($) |
12 Months Ended | |||
|---|---|---|---|---|
Mar. 11, 2021 |
Feb. 12, 2021 |
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| At-The-Market Program [Abstract] | ||||
| Gross proceeds | $ 9,227,000 | $ 4,428,000 | ||
| Issuance expenses | $ 278,000 | $ 131,000 | ||
| 2021 Shelf [Member] | Maximum [Member] | ||||
| At-The-Market Program [Abstract] | ||||
| Aggregate offering price | $ 125,000,000 | |||
| ATM [Member] | ||||
| At-The-Market Program [Abstract] | ||||
| Shares sold (in shares) | 1,417,446 | 1,848,980 | ||
| Gross proceeds | $ 4,700,000 | $ 4,400,000 | ||
| Issuance expenses | $ 136,000 | $ 133,000 | ||
| ATM [Member] | Maximum [Member] | ||||
| At-The-Market Program [Abstract] | ||||
| Aggregate offering price | $ 40,000,000 | |||
Stockholders' Equity, Registered Direct Offering (Details) - USD ($) $ / shares in Units, $ in Thousands |
12 Months Ended | ||
|---|---|---|---|
Jun. 08, 2021 |
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Registered Direct Offerings [Abstract] | |||
| Gross proceeds | $ 9,227 | $ 4,428 | |
| Issuance expenses | $ 278 | $ 131 | |
| Registered Direct Offering [Member] | |||
| Registered Direct Offerings [Abstract] | |||
| Gross proceeds | $ 15,000 | ||
| Registered Direct Offering [Member] | Common Stock [Member] | |||
| Registered Direct Offerings [Abstract] | |||
| Shares sold (in shares) | 3,076,923 | ||
| Warrants issued (in shares) | 1,538,461 | ||
| Offering price (in dollars per share) | $ 4.875 | ||
| Issuance expenses | $ 1,100 | ||
| Registered Direct Offering [Member] | RDO Warrants [Member] | |||
| Registered Direct Offerings [Abstract] | |||
| Number of RDO warrants sold at offering price (per unit) | 0.5 | ||
| Exercise price (in dollars per share) | $ 6.09 | ||
| Expiration period | 5 years | ||
| Warrants outstanding (in shares) | 1,538,461 | ||
| Minimum percentage of beneficial ownership | 4.99% | ||
| Maximum percentage of beneficial ownership limitation | 9.99% | ||
Stockholders' Equity, Pre-Merger Financing, Series A & B Warrants (Details) $ / shares in Units, $ in Thousands |
12 Months Ended | ||||
|---|---|---|---|---|---|
|
Jun. 29, 2020
USD ($)
Director
|
Dec. 31, 2023
$ / shares
shares
|
Dec. 31, 2022
shares
|
Nov. 19, 2020
$ / shares
shares
|
Nov. 05, 2020
shares
|
|
| Rexahn [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Exercise price (in dollars per share) | $ / shares | $ 38.4 | ||||
| Warrants outstanding (in shares) | 58,597 | 231,433 | |||
| Securities Purchase Agreement [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Number of directors | Director | 5 | ||||
| Securities Purchase Agreement [Member] | Rexahn [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Number of directors | Director | 1 | ||||
| Securities Purchase Agreement [Member] | Investors [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Total investment | $ | $ 21,150 | ||||
| Securities Purchase Agreement [Member] | Directors [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Total investment | $ | $ 300 | ||||
| Series A Warrants [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Exercise price (in dollars per share) | $ / shares | $ 4.4795 | ||||
| Exercisable term | 5 years | ||||
| Warrant issued (in shares) | 5,665,838 | ||||
| Series B Warrants [Member] | |||||
| Pre-Merger Financing [Abstract] | |||||
| Exercise price (in dollars per share) | $ / shares | $ 0.0001 | ||||
| Warrants outstanding (in shares) | 0 | ||||
| Warrants exercised (in shares) | 17,869 | 60,832 | |||
License and Collaboration Agreements, Viatris License Agreement (Details) $ in Thousands |
1 Months Ended | 12 Months Ended | |||
|---|---|---|---|---|---|
|
Sep. 25, 2023
USD ($)
|
Nov. 06, 2022
USD ($)
|
Nov. 30, 2022
USD ($)
|
Dec. 31, 2023
USD ($)
Performanceobligation
|
Dec. 31, 2022
USD ($)
|
|
| Reconciliation of Closing Balance of Contract Asset [Abstract] | |||||
| Revenue recognized | $ 19,049 | $ 39,850 | |||
| Viatris License Agreement [Member] | |||||
| Collaboration and License Agreement [Abstract] | |||||
| Non-refundable cash payment received | $ 35,000 | ||||
| Maximum amount of payments receivable for development, regulatory and commercial milestones | $ 130,000 | ||||
| First potential payments to be received | $ 10,000 | ||||
| Maximum percentage of tiered royalties receivable | 20.00% | ||||
| Number of distinct performance obligations | Performanceobligation | 2 | ||||
| Aggregate transaction price to be recognized | $ 10,000 | $ 40,000 | |||
| Period of non-cancellation window agreement | 120 days | ||||
| Milestone payment included in revenue recognization | 10,000 | ||||
| Reconciliation of Closing Balance of Contract Asset [Abstract] | |||||
| Balance as of beginning of period | 3,552 | 0 | |||
| Revenue recognized | 19,049 | 39,850 | |||
| Balance as of end of period | 1,407 | 3,552 | |||
| Viatris License Agreement [Member] | License Transfer Fee [Member] | |||||
| Collaboration and License Agreement [Abstract] | |||||
| Aggregate transaction price to be recognized | 35,000 | ||||
| Estimated standalone selling price for license agreement | $ 287,800 | ||||
| Transaction price allocation of ESSP obligations | 39,300 | ||||
| Reconciliation of Closing Balance of Contract Asset [Abstract] | |||||
| Revenue recognized | 0 | (35,000) | |||
| Reclassification to accounts receivable related to costs billed under the Viatris License Agreement | (21,194) | $ (1,298) | |||
| Viatris License Agreement [Member] | Research and Development Services [Member] | |||||
| Collaboration and License Agreement [Abstract] | |||||
| Aggregate transaction price to be recognized | 5,000 | ||||
| Estimated standalone selling price for license agreement | 5,000 | ||||
| Transaction price allocation of ESSP obligations | $ 700 | ||||
| Viatris License Agreement [Member] | Sales Milestones [Member] | |||||
| Collaboration and License Agreement [Abstract] | |||||
| Milestone payment requirements attributed to the FDA's approval | 10,000 | ||||
| Reconciliation of Closing Balance of Contract Asset [Abstract] | |||||
| Revenue recognized | $ 0 | ||||
License and Collaboration Agreements, BioSense License and Assignment Agreement (Details) |
12 Months Ended | ||
|---|---|---|---|
|
Mar. 10, 2020
USD ($)
|
Dec. 31, 2023
USD ($)
Milestone
Country
|
Dec. 31, 2021
USD ($)
|
|
| BioSense License and Assignments Agreement [Member] | |||
| Collaboration and License Agreement [Abstract] | |||
| Non-refundable cash payment received | $ 1,650,000 | ||
| Milestone payment achieved for performance obligation, recorded as collaboration revenue | $ 100,000 | ||
| Milestone payments received | $ 0 | ||
| Maximum amount of payments receivable for development, regulatory and commercial milestones | 84,500,000 | ||
| BioSense License and Assignments Agreement [Member] | Rexahn [Member] | |||
| Collaboration and License Agreement [Abstract] | |||
| Non-refundable cash payment received | $ 1,550,000 | ||
| Processa License Agreement [Member] | |||
| Collaboration and License Agreement [Abstract] | |||
| Milestone payments received | $ 0 | ||
| Number of times sales milestone payments | Milestone | 1 | ||
| Percentage of milestone payments eligible to receive on sub-license agreement | 32.00% | ||
| Number of countries | Country | 1 | ||
| Milestone period to administer drug clinical trial of licensed product on first patient | 3 years | ||
| Milestone period to administer drug clinical trial of licensed product on first patient for second indication | 5 years | ||
| Period of opportunity to cure breach of agreement | 120 days | ||
| Period of prior written notice to be served for termination of agreements | 120 days | ||
Net (loss) income per share, Computation of Diluted Net Income (Loss) Per Share (Details) - shares |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Computation of weighted average common shares [Abstract] | ||
| Basic common shares outstanding (in shares) | 21,589,821 | 19,931,080 |
| Dilutive stock options (in shares) | 0 | 589,165 |
| Dilutive warrants (in shares) | 0 | 76,967 |
| Diluted common shares outstanding (in shares) | 21,589,821 | 20,597,212 |
Net (loss) income per share, Anti-dilutive Securities Excluded from Computation of Net Income (Loss) per Share (Details) - shares |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Series A, Series B and RDO Warrants [Member] | ||
| Net Income (Loss) Per Share [Abstract] | ||
| Antidilutive securities excluded from computation of net loss per share (in shares) | 7,204,299 | 7,145,201 |
| Stock Options [Member] | ||
| Net Income (Loss) Per Share [Abstract] | ||
| Antidilutive securities excluded from computation of net loss per share (in shares) | 4,410,258 | 2,346,879 |
| RSUs [Member] | ||
| Net Income (Loss) Per Share [Abstract] | ||
| Antidilutive securities excluded from computation of net loss per share (in shares) | 801,700 | 0 |
| Former Rexahn Warrants [Member] | ||
| Net Income (Loss) Per Share [Abstract] | ||
| Antidilutive securities excluded from computation of net loss per share (in shares) | 58,597 | 60,713 |
Income Taxes, Reconciliation of Statutory to Effective Income Tax Rate (Details) |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Effective Federal Income Tax Rate Reconciliation [Abstract] | ||
| Income tax (benefit) provision at federal statutory rate | (21.00%) | 21.00% |
| Valuation allowance | 23.80% | (21.40%) |
| State income tax, net of federal benefit | (4.90%) | 4.90% |
| Financing contracts | 3.20% | 0.00% |
| Stock options | 1.00% | 0.40% |
| Research and development | (3.90%) | (3.10%) |
| Other | 1.90% | (0.10%) |
| Effective tax rate | 0.10% | 1.70% |
Income Taxes, Components of Income Tax Provision (Benefit) (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Components of Income Tax Provision (Benefit) [Abstract] | ||
| (Loss) income before income taxes | $ (9,974) | $ 18,203 |
| Current: | ||
| Federal | 2 | 279 |
| State | 10 | 36 |
| Total current tax provision (benefit) | 12 | 315 |
| Deferred: | ||
| Federal | 0 | 0 |
| State | 0 | 0 |
| Total tax provision (benefit) | $ 12 | $ 315 |
Income Taxes, Deferred Tax Assets and Liabilities (Details) - USD ($) $ in Thousands |
Dec. 31, 2023 |
Dec. 31, 2022 |
|---|---|---|
| Deferred Tax Assets [Abstract] | ||
| Federal and state operating loss carryforwards | $ 12,780 | $ 13,087 |
| Acquired intangibles | 547 | 547 |
| Deferral of research and development costs | 3,794 | 2,820 |
| Organizational costs | 6 | 7 |
| Other | 72 | 62 |
| Stock-based compensation | 1,835 | 1,152 |
| Research and development credit carryforward | 1,107 | 731 |
| Subtotal | 20,141 | 18,406 |
| Valuation allowance | (20,141) | (17,770) |
| Total deferred tax assets, net of valuation allowance | 0 | 636 |
| Deferred Tax Liabilities [Abstract] | ||
| Deferred revenue | 0 | (636) |
| Total deferred tax liabilities | 0 | (636) |
| Net deferred tax assets | $ 0 | $ 0 |
Income Taxes, Operating Loss Carryforwards (Details) - USD ($) $ in Thousands |
12 Months Ended | |
|---|---|---|
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Deferred Tax Assets, Operating Loss Carryforwards, Components [Abstract] | ||
| Federal net operating loss carryforward | $ 10,600 | $ 10,900 |
| Federal research credit carryforwards | 1,100 | 700 |
| State net operating loss carryforward | 2,100 | 2,200 |
| State research credit carryforwards | 0 | 0 |
| Federal operating loss carryforwards, tax effect | 200 | 4,800 |
| State operating loss carryforwards, tax effect | 100 | 1,400 |
| Utilized federal research credit carryforwards | 26 | 900 |
| Income Tax Uncertainties [Abstract] | ||
| Reserves or related accruals for interest and penalty on income tax expense | 0 | 0 |
| Deferred tax assets relating to net operating loss carryforwards [Abstract] | ||
| Deferred tax assets relating to net operating loss carryforwards | 12,780 | $ 13,087 |
| Rexahn stockholders [Member] | ||
| Deferred tax assets relating to net operating loss carryforwards [Abstract] | ||
| Deferred tax assets relating to net operating loss carryforwards | $ 10,300 | |
Deferred Compensation Plan (Details) - 401K Plan [Member] - USD ($) |
12 Months Ended | ||
|---|---|---|---|
Oct. 01, 2021 |
Dec. 31, 2023 |
Dec. 31, 2022 |
|
| Deferred Compensation Plan [Abstract] | |||
| Employer matching contribution, first match | 100.00% | ||
| Deferred compensation matched by employer, first match | 3.00% | ||
| Additional employer matching contribution | 50.00% | ||
| Additional deferred compensation matched by employer | 2.00% | ||
| Employer contribution | $ 99,000 | $ 76,000 | |
Subsequent Events (Details) $ / shares in Units, $ in Millions |
12 Months Ended | |||
|---|---|---|---|---|
|
Feb. 12, 2024
Intallment
$ / shares
shares
|
Jan. 10, 2024
USD ($)
|
Jan. 01, 2024
shares
|
Dec. 31, 2023 |
|
| 2020 Plan Evergreen Provision [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Percentage of common stock shares outstanding | 5.00% | |||
| 2020 Plan Evergreen Provision [Member] | Maximum [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Period of shares reserved under plan | 10 years | |||
| Subsequent Event [Member] | Maximum [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Aggregate offering price | $ | $ 175 | |||
| Subsequent Event [Member] | 2020 Plan Evergreen Provision [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Number of shares added (in shares) | 1,198,875 | |||
| Subsequent Event [Member] | Inducement Plan [Member] | Stock Options [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Shares issued (in shares) | 435,000 | |||
| Common stock exercise price of options (in dollars per share) | $ / shares | $ 2.66 | |||
| Vesting period | 4 years | |||
| Subsequent Event [Member] | Inducement Plan [Member] | Stock Options [Member] | Tranche One [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Vesting percentage | 25.00% | |||
| Subsequent Event [Member] | Inducement Plan [Member] | Stock Options [Member] | Tranche Two [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Vesting percentage | 75.00% | |||
| Subsequent Event [Member] | Inducement Plan [Member] | Restricted Stock Units [Member] | ||||
| Subsequent Events [Abstract] | ||||
| Shares issued (in shares) | 215,000 | |||
| Number of equal annual installments for vesting | Intallment | 4 |
1 Year Ocuphire Pharma Chart |
1 Month Ocuphire Pharma Chart |

It looks like you are not logged in. Click the button below to log in and keep track of your recent history.
Support: +44 (0) 203 8794 460 | support@advfn.com
By accessing the services available at ADVFN you are agreeing to be bound by ADVFN's Terms & Conditions