false2023-06-302023Q20001809196--12-31 0001809196 2023-06-30 0001809196 2022-12-31 0001809196 2022-01-01 2022-06-30 0001809196 2023-01-01 2023-06-30 0001809196 2022-04-01 2022-06-30 0001809196 2023-04-01 2023-06-30 0001809196 2022-06-30 0001809196 2021-12-31 0001809196 imtx:BMSCollaborationAgreementMember 2023-04-01 2023-06-30 0001809196 imtx:ResearchAndDevelopmentExpenseMember 2023-04-01 2023-06-30 0001809196 imtx:GeneralAndAdministrativeExpenseMember 2023-04-01 2023-06-30 0001809196 imtx:GenmabCollaborationAgreementMember country:DK 2023-04-01 2023-06-30 0001809196 imtx:BMSCollaborationAgreementMember country:US 2023-04-01 2023-06-30 0001809196 country:GB imtx:GSKCollaborationAgreementMember 2023-04-01 2023-06-30 0001809196 imtx:CollaborationAgreementsMember 2023-04-01 2023-06-30 0001809196 imtx:BMSOptInAgreementMember 2023-04-01 2023-06-30 0001809196 imtx:ServiceOptionsMember imtx:KeyManagementPersonnelAndBoardOfDirectorsMember 2023-04-01 2023-06-30 0001809196 imtx:ResearchAndDevelopmentExpenseMember 2022-04-01 2022-06-30 0001809196 imtx:GeneralAndAdministrativeExpenseMember 2022-04-01 2022-06-30 0001809196 country:DE imtx:ImmaticsBiotechnologiesGmbhMember 2022-04-01 2022-06-30 0001809196 country:DK imtx:GenmabCollaborationAgreementMember 2022-04-01 2022-06-30 0001809196 country:US imtx:BMSCollaborationAgreementMember 2022-04-01 2022-06-30 0001809196 imtx:GSKCollaborationAgreementMember country:GB 2022-04-01 2022-06-30 0001809196 imtx:CollaborationAgreementsMember 2022-04-01 2022-06-30 0001809196 ifrs-full:OtherReservesMember 2022-01-01 2022-06-30 0001809196 ifrs-full:RetainedEarningsMember 2022-01-01 2022-06-30 0001809196 ifrs-full:SharePremiumMember 2022-01-01 2022-06-30 0001809196 imtx:ResearchAndDevelopmentExpenseMember 2022-01-01 2022-06-30 0001809196 imtx:GeneralAndAdministrativeExpenseMember 2022-01-01 2022-06-30 0001809196 ifrs-full:IssuedCapitalMember 2022-01-01 2022-06-30 0001809196 srt:ScenarioPreviouslyReportedMember 2022-01-01 2022-06-30 0001809196 srt:RevisionOfPriorPeriodErrorCorrectionAdjustmentMember 2022-01-01 2022-06-30 0001809196 imtx:CurrentlyReportedMember 2022-01-01 2022-06-30 0001809196 imtx:GenmabCollaborationAgreementMember country:DK 2022-01-01 2022-06-30 0001809196 imtx:BMSCollaborationAgreementMember country:US 2022-01-01 2022-06-30 0001809196 country:GB imtx:GSKCollaborationAgreementMember 2022-01-01 2022-06-30 0001809196 imtx:CollaborationAgreementsMember 2022-01-01 2022-06-30 0001809196 ifrs-full:OtherReservesMember 2023-01-01 2023-06-30 0001809196 ifrs-full:RetainedEarningsMember 2023-01-01 2023-06-30 0001809196 ifrs-full:SharePremiumMember 2023-01-01 2023-06-30 0001809196 imtx:CelgeneSwitzerlandLlcAndGenmabAsMember 2023-01-01 2023-06-30 0001809196 imtx:BMSCollaborationAgreementMember 2023-01-01 2023-06-30 0001809196 imtx:NonInterestBearingLiabilityMember 2023-01-01 2023-06-30 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-01-01 2023-06-30 0001809196 imtx:MatchingStockOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:ConvertedOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:PerformanceBasedOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:GrantedOnMarchTwoThousandAndTwentyOneMember imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-01-01 2023-06-30 0001809196 imtx:GrantedOnSeptemberTwoThousandAndTwentyOneMember imtx:PerformanceBasedOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:GrantedOnJuneTwoThousandAndTwentyMember imtx:ConvertedOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:MatchingStockOptionsMember 2023-01-01 2023-06-30 0001809196 imtx:ResearchAndDevelopmentExpenseMember 2023-01-01 2023-06-30 0001809196 imtx:GeneralAndAdministrativeExpenseMember 2023-01-01 2023-06-30 0001809196 ifrs-full:IssuedCapitalMember 2023-01-01 2023-06-30 0001809196 imtx:BMSCollaborationAgreementMember imtx:Ima401Member 2023-01-01 2023-06-30 0001809196 imtx:AtTheMarketAgreementMember imtx:SvbSecuritiesLlcMember 2023-01-01 2023-06-30 0001809196 imtx:GenmabCollaborationAgreementMember country:DK 2023-01-01 2023-06-30 0001809196 country:US imtx:BMSCollaborationAgreementMember 2023-01-01 2023-06-30 0001809196 imtx:GSKCollaborationAgreementMember country:GB 2023-01-01 2023-06-30 0001809196 imtx:CollaborationAgreementsMember 2023-01-01 2023-06-30 0001809196 imtx:BMSOptInAgreementMember 2023-01-01 2023-06-30 0001809196 imtx:ServiceOptionsMember imtx:KeyManagementPersonnelAndBoardOfDirectorsMember 2023-01-01 2023-06-30 0001809196 ifrs-full:LeaseholdImprovementsMember 2023-01-01 2023-06-30 0001809196 imtx:RentalLandMember 2023-01-01 2023-06-30 0001809196 imtx:CelgeneSwitzerlandLlcAndGenmabAsMember 2022-12-31 0001809196 imtx:BondsMember ifrs-full:CarryingAmountMember 2022-12-31 0001809196 ifrs-full:AtFairValueMember imtx:BondsMember 2022-12-31 0001809196 ifrs-full:CarryingAmountMember 2022-12-31 0001809196 ifrs-full:AtFairValueMember 2022-12-31 0001809196 imtx:CashAndCashEquivalentsMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2022-12-31 0001809196 imtx:BondsMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2022-12-31 0001809196 ifrs-full:TradeReceivablesMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2022-12-31 0001809196 imtx:OtherCurrentNonCurrentAssetsMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2022-12-31 0001809196 ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2022-12-31 0001809196 ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember imtx:ShorttermDepositsMember 2022-12-31 0001809196 imtx:AccountsPayableMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2022-12-31 0001809196 imtx:OtherCurrentLiabilitiesMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2022-12-31 0001809196 imtx:LiabilitiesForWarrantsMember ifrs-full:FinancialLiabilitiesAtFairValueThroughProfitOrLossCategoryMember 2022-12-31 0001809196 ifrs-full:LeaseLiabilitiesMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2022-12-31 0001809196 ifrs-full:FinancialLiabilitiesAtFairValueThroughProfitOrLossCategoryMember 2022-12-31 0001809196 ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2022-12-31 0001809196 imtx:ReorganizationMember 2022-12-31 0001809196 imtx:CashAndCashEquivalentsMember 2022-12-31 0001809196 imtx:BondsMember 2022-12-31 0001809196 ifrs-full:TradeReceivablesMember 2022-12-31 0001809196 imtx:OtherCurrentNonCurrentAssetsMember 2022-12-31 0001809196 imtx:AccountsPayableMember 2022-12-31 0001809196 imtx:OtherCurrentLiabilitiesMember 2022-12-31 0001809196 imtx:LiabilitiesForWarrantsMember 2022-12-31 0001809196 ifrs-full:LeaseLiabilitiesMember 2022-12-31 0001809196 imtx:ShorttermDepositsMember 2022-12-31 0001809196 imtx:CelgeneSwitzerlandLlcAndGenmabAsMember 2023-06-30 0001809196 imtx:CashAndCashEquivalentsMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2023-06-30 0001809196 ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember imtx:ShorttermDepositsMember 2023-06-30 0001809196 ifrs-full:TradeReceivablesMember ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2023-06-30 0001809196 ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember imtx:OtherCurrentNonCurrentAssetsMember 2023-06-30 0001809196 ifrs-full:FinancialAssetsAtAmortisedCostCategoryMember 2023-06-30 0001809196 imtx:AccountsPayableMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2023-06-30 0001809196 imtx:OtherCurrentLiabilitiesMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2023-06-30 0001809196 imtx:LiabilitiesForWarrantsMember ifrs-full:FinancialLiabilitiesAtFairValueThroughProfitOrLossCategoryMember 2023-06-30 0001809196 ifrs-full:LeaseLiabilitiesMember ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2023-06-30 0001809196 ifrs-full:FinancialLiabilitiesAtFairValueThroughProfitOrLossCategoryMember 2023-06-30 0001809196 ifrs-full:FinancialLiabilitiesAtAmortisedCostCategoryMember 2023-06-30 0001809196 imtx:ReorganizationMember 2023-06-30 0001809196 imtx:MatchingStockOptionsMember 2023-06-30 0001809196 imtx:ConvertedOptionsMember 2023-06-30 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-06-30 0001809196 imtx:PerformanceBasedOptionsMember 2023-06-30 0001809196 imtx:PerformanceBasedOptionsMember imtx:TrancheOneMember 2023-06-30 0001809196 imtx:TrancheTwoMember imtx:PerformanceBasedOptionsMember 2023-06-30 0001809196 imtx:PerformanceBasedOptionsMember imtx:TrancheThreeMember 2023-06-30 0001809196 imtx:CashAndCashEquivalentsMember 2023-06-30 0001809196 imtx:ShorttermDepositsMember 2023-06-30 0001809196 ifrs-full:TradeReceivablesMember 2023-06-30 0001809196 imtx:OtherCurrentNonCurrentAssetsMember 2023-06-30 0001809196 imtx:AccountsPayableMember 2023-06-30 0001809196 imtx:OtherCurrentLiabilitiesMember 2023-06-30 0001809196 imtx:LiabilitiesForWarrantsMember 2023-06-30 0001809196 ifrs-full:LeaseLiabilitiesMember 2023-06-30 0001809196 ifrs-full:TopOfRangeMember 2023-06-30 0001809196 imtx:CollaborationAgreementMember 2023-06-30 0001809196 imtx:CelgeneSwitzerlandLlcAndGenmabAsMember 2022-01-01 2022-12-31 0001809196 imtx:AuthorizationOfFinancialStatementsMember 2023-08-17 2023-08-17 0001809196 imtx:IssueOfSecuritiesMember imtx:SvbSecuritiesLlcMember imtx:AtTheMarketAgreementMember 2023-08-17 2023-08-17 0001809196 ifrs-full:BottomOfRangeMember 2023-03-31 0001809196 ifrs-full:BottomOfRangeMember 2022-03-31 0001809196 ifrs-full:TopOfRangeMember 2022-06-30 0001809196 imtx:ImmaticsBiotechnologiesGmbhMember country:DE 2022-06-30 0001809196 imtx:IssueOfSecuritiesMember 2023-07-19 0001809196 imtx:IssueOfSecuritiesMember 2023-07-19 2023-07-19 0001809196 imtx:IssueOfSecuritiesMember imtx:SvbSecuritiesLlcMember imtx:AtTheMarketAgreementMember 2023-08-17 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-04-03 2023-04-03 0001809196 imtx:GrantedInJuneTwoThousandAndTwentyTwoMember imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-04-03 2023-04-03 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2023-06-27 2023-06-27 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember imtx:GrantedInJuneTwoThousandAndTwentyTwoMember 2023-06-27 2023-06-27 0001809196 ifrs-full:IssuedCapitalMember 2021-12-31 0001809196 ifrs-full:SharePremiumMember 2021-12-31 0001809196 ifrs-full:RetainedEarningsMember 2021-12-31 0001809196 ifrs-full:OtherReservesMember 2021-12-31 0001809196 ifrs-full:IssuedCapitalMember 2022-06-30 0001809196 ifrs-full:SharePremiumMember 2022-06-30 0001809196 ifrs-full:RetainedEarningsMember 2022-06-30 0001809196 ifrs-full:OtherReservesMember 2022-06-30 0001809196 srt:ScenarioPreviouslyReportedMember 2021-12-31 0001809196 srt:RevisionOfPriorPeriodErrorCorrectionAdjustmentMember 2021-12-31 0001809196 imtx:CurrentlyReportedMember 2021-12-31 0001809196 srt:ScenarioPreviouslyReportedMember 2022-06-30 0001809196 srt:RevisionOfPriorPeriodErrorCorrectionAdjustmentMember 2022-06-30 0001809196 imtx:CurrentlyReportedMember 2022-06-30 0001809196 ifrs-full:IssuedCapitalMember 2022-12-31 0001809196 ifrs-full:SharePremiumMember 2022-12-31 0001809196 ifrs-full:RetainedEarningsMember 2022-12-31 0001809196 ifrs-full:OtherReservesMember 2022-12-31 0001809196 imtx:MatchingStockOptionsMember 2022-12-31 0001809196 imtx:ConvertedOptionsMember 2022-12-31 0001809196 imtx:AdditionalGrantsUnderTwoThousandAndTwentyPlanMember 2022-12-31 0001809196 imtx:PerformanceBasedOptionsMember 2022-12-31 0001809196 ifrs-full:OtherReservesMember 2023-06-30 0001809196 ifrs-full:RetainedEarningsMember 2023-06-30 0001809196 ifrs-full:SharePremiumMember 2023-06-30 0001809196 ifrs-full:IssuedCapitalMember 2023-06-30 iso4217:EUR xbrli:shares xbrli:pure iso4217:USD utr:Year iso4217:EUR xbrli:shares iso4217:USD xbrli:shares imtx:Warrants utr:Y
SECURITIES AND EXCHANGE COMMISSION
REPORT OF FOREIGN PRIVATE ISSUER
Pursuant to Rule
13a-16
or
15d-16
of the Securities Exchange Act of 1934
Commission File Number:
001-39363
72076 Tübingen, Federal Republic of Germany
(Address of Principal Executive Office)
Indicate by check mark whether the registrant files or will file annual reports under cover Form
20-F
or Form
40-F.
INFORMATION
CONTAINED IN THIS REPORT ON FORM
6-K
On August 17, 2023, Immatics N.V. (the “Company”) issued an interim report for the three and
six-month
periods ended June 30, 2023, which is attached hereto as Exhibit 99.1, and issued a press release announcing the second quarter 2023 financial results for the Company, which is attached hereto as Exhibit 99.2. Additionally, the Company made available an updated investor presentation, which is attached hereto as Exhibit 99.3. The fact that the presentation is being made available and furnished herewith is not an admission as to the materiality of any information contained in the presentation. The information contained in the presentation is being provided as of November 17, 2022 and the Company does not undertake any obligation to update the presentation in the future or to update forward-looking statements to reflect subsequent actual results.
INCORPORATION BY REFERENCE
This
Report
on Form
6-K
(other than Exhibit 99.2 and Exhibit 99.3 hereto) including Exhibit 99.1 hereto, shall be deemed to be incorporated by reference into the registration statements on Form
S-8
(333-249408
and
333-265820)
and the registration statements on Form
F-3
(Registration Nos.
333-258351
and
333-240260)
of Immatics N.V. and to be a part thereof from the date on which this report is filed, to the extent not superseded by documents or reports subsequently filed or furnished.
1
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
|
|
|
|
|
| |
|
|
|
|
| |
|
|
|
| Date: August 17, 2023 |
|
|
|
by: |
|
/s/ Harpreet Singh |
| |
|
|
|
|
|
Harpreet Singh |
| |
|
|
|
|
|
Chief Executive Officer |
2
The unaudited condensed Consolidated Financial Statements for the three and
six-month
periods ended June 30, 2023, included herein, have been prepared in accordance with International Accounting Standard 34 (“Interim Financial Reporting”), as issued by the International Accounting Standards Board (“IASB”). The Consolidated Financial Statements are presented in euros. All references in this interim report to “$,” and “U.S. dollars” mean U.S. dollars and all references to “€” and “euros” mean euros, unless otherwise noted.
This interim report, including “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” contains statements that constitute forward-looking statements within the meaning of Section 21E of the Exchange Act and Section 27A of the Securities Act of 1933, as amended (the “Securities Act”). All statements other than statements of historical facts, including statements regarding our future results of operations and financial position, business and commercial strategy, potential market opportunities, products and product candidates, research pipeline, ongoing and planned preclinical studies and clinical trials, regulatory submissions and approvals, research and development costs, timing and likelihood of success, as well as plans and objectives of management for future operations are forward-looking statements. Many of the forward-looking statements contained in this interim report can be identified by the use of forward-looking words such as “anticipate,” “believe,” “could,” “expect,” “should,” “plan,” “intend,” “estimate,” “will” and “potential” among others. Forward-looking statements are based on our management’s beliefs and assumptions and on information available to our management at the time such statements are made. Such statements are subject to risks and uncertainties, and actual results may differ materially from those expressed or implied in the forward-looking statements due to various factors, including, but not limited to the macro-economic environment; inconclusive clinical trial results or clinical trials failing to achieve one or more endpoints, early data not being repeated in ongoing or future clinical trials, failures to secure required regulatory approvals, disruptions from failures by third-parties on whom we rely in connection with our clinical trials, delays or negative determinations by regulatory authorities, changes or increases in oversight and regulation; increased competition; manufacturing delays or problems, inability to achieve enrollment targets, disagreements with our collaboration partners or failures of collaboration partners to pursue product candidates, legal challenges, including product liability claims or intellectual property disputes, commercialization factors, including regulatory approval and pricing determinations, disruptions to access to raw materials or starting material, proliferation and continuous evolution of new technologies; disruptions to Immatics’ business; management changes; dislocations in the capital markets; and other important factors described under “Risk Factors” in our Annual Report on Form
20-F
for the year ended December 31, 2022, filed with the Securities and Exchange Commission on March 22, 2023 and those described in our other filings with the Securities and Exchange Commission. Forward-looking statements speak only as of the date on which they were made. Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements, whether as a result of any new information, future events, changed circumstances or otherwise.
We own various trademark registrations and applications, and unregistered trademarks, including Immatics
®
, XPRESIDENT
®
, ACTengine
®
, ACTallo
®
, ACTolog
®
, XCEPTOR
®
, TCER
®
, AbsQuant
®
, IMADetect
®
and our corporate logo. All other trade names, trademarks and service marks of other companies appearing in this interim report are the property of their respective owners. Solely for convenience, the trademarks and trade names in this interim report may be referred to without the
®
and
™
symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. We do not intend to use or display other companies’ trademarks and trade names to imply a relationship with, or endorsement or sponsorship of us by, any other companies.
As used in this interim report, the terms “Immatics,” “we,” “our,” “us,” “the Group” and “the Company” refer to Immatics N.V. and its subsidiaries, taken as a whole, unless the context otherwise requires. The unaudited condensed consolidated financial statements and Management’s Discussion & Analysis of Financial Condition and Results of Operations in this interim report are related to Immatics N.V. and its German subsidiary Immatics Biotechnologies GmbH as well as its U.S. subsidiary Immatics US Inc.
Unaudited Condensed Consolidated Statement of Profit/(Loss) of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
(Euros in thousands, except
per share data) |
|
|
(Euros in thousands, except
per share data) |
|
Revenue from collaboration agreements |
|
|
5 |
|
|
|
22,354 |
|
|
|
17,215 |
|
|
|
32,150 |
|
|
|
120,123 |
|
Research and development expenses |
|
|
|
|
|
|
(27,317 |
) |
|
|
(25,216 |
) |
|
|
(54,898 |
) |
|
|
(50,360 |
) |
General and administrative expenses |
|
|
|
|
|
|
(9,358 |
) |
|
|
(8,683 |
) |
|
|
(18,944 |
) |
|
|
(17,961 |
) |
Other income |
|
|
|
|
|
|
6 |
|
|
|
27 |
|
|
|
948 |
|
|
|
32 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Change in fair value of liabilities for warrants |
|
|
6 |
|
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
Other financial income |
|
|
6 |
|
|
|
3,954 |
|
|
|
7,015 |
|
|
|
6,748 |
|
|
|
8,774 |
|
Other financial expenses |
|
|
6 |
|
|
|
(1,144 |
) |
|
|
(407 |
) |
|
|
(4,653 |
) |
|
|
(1,524 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Profit/(loss) before taxes |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Taxes on income |
|
|
7 |
|
|
|
— |
|
|
|
(1,145 |
) |
|
|
— |
|
|
|
(1,145 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net profit/(loss) per share: |
|
|
17 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Basic |
|
|
|
|
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
Diluted |
|
|
|
|
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Unaudited Condensed Consolidated Statement of Comprehensive Income/(Loss) of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other comprehensive income/(loss) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Items that may be reclassified subsequently to profit or loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Currency translation differences from foreign operations |
|
|
14 |
|
|
|
(224 |
) |
|
|
778 |
|
|
|
340 |
|
|
|
1,338 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total comprehensive income/(loss) for the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Unaudited Condensed Consolidated Statement of Financial Position of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
16 |
|
|
|
130,405 |
|
|
|
148,519 |
|
Other financial assets |
|
|
16 |
|
|
|
217,222 |
|
|
|
213,686 |
|
Accounts receivables |
|
|
16 |
|
|
|
330 |
|
|
|
1,111 |
|
Other current assets |
|
|
9 |
|
|
|
16,668 |
|
|
|
13,838 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Property, plant and equipment |
|
|
10 |
|
|
|
27,188 |
|
|
|
13,456 |
|
Intangible assets |
|
|
10 |
|
|
|
1,655 |
|
|
|
1,632 |
|
|
|
|
10 |
|
|
|
14,749 |
|
|
|
13,033 |
|
|
|
|
9 |
|
|
|
1,972 |
|
|
|
2,545 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and shareholders’ equity |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Provisions |
|
|
11 |
|
|
|
3,117 |
|
|
|
— |
|
Accounts payables |
|
|
12 |
|
|
|
19,904 |
|
|
|
13,056 |
|
Deferred revenue |
|
|
5 |
|
|
|
67,997 |
|
|
|
64,957 |
|
Liabilities for warrants |
|
|
16 |
|
|
|
22,622 |
|
|
|
16,914 |
|
Lease liabilities |
|
|
16 |
|
|
|
2,737 |
|
|
|
2,159 |
|
Other current liabilities |
|
|
13 |
|
|
|
7,929 |
|
|
|
9,366 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Deferred revenue |
|
|
5 |
|
|
|
53,559 |
|
|
|
75,759 |
|
Lease liabilities |
|
|
16 |
|
|
|
14,085 |
|
|
|
12,403 |
|
Other non-current liabilities |
|
|
|
|
|
|
26 |
|
|
|
42 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Share capital |
|
|
14 |
|
|
|
804 |
|
|
|
767 |
|
Share premium |
|
|
14 |
|
|
|
763,206 |
|
|
|
714,177 |
|
Accumulated deficit |
|
|
14 |
|
|
|
(544,656 |
) |
|
|
(500,299 |
) |
Other reserves |
|
|
14 |
|
|
|
(1,141 |
) |
|
|
(1,481 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholders’ equity |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities and shareholders’ equity |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Unaudited Condensed Consolidated Statement of Cash Flows of Immatics N.V.
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
| |
|
|
|
Cash flows from operating activities |
|
|
|
|
|
|
|
|
Net profit/(loss) |
|
|
(44,357 |
) |
|
|
71,682 |
|
Taxes on income |
|
|
— |
|
|
|
1,145 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
|
(4,999 |
) |
|
|
(23 |
) |
Depreciation and amortization |
|
|
3,666 |
|
|
|
3,407 |
|
Interest expenses |
|
|
401 |
|
|
|
538 |
|
Equity settled share-based payment |
|
|
11,615 |
|
|
|
11,262 |
|
Net foreign exchange differences and expected credit losses |
|
|
4,081 |
|
|
|
(7,834 |
) |
Change in fair value of liabilities for warrants |
|
|
5,708 |
|
|
|
(13,743 |
) |
|
|
|
|
|
|
|
|
|
Decrease/(increase) in accounts receivables |
|
|
781 |
|
|
|
(280 |
) |
Decrease/(increase) in other assets |
|
|
765 |
|
|
|
(6,903 |
) |
(Decrease)/increase in deferred revenue, accounts payables and other liabilities |
|
|
(9,889 |
) |
|
|
96,933 |
|
Interest received |
|
|
2,051 |
|
|
|
23 |
|
Interest paid |
|
|
(146 |
) |
|
|
(434 |
) |
Income tax paid |
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
Net cash (used in)/provided by operating activities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from investing activities |
|
|
|
|
|
|
|
|
Payments for property, plant and equipment |
|
|
(15,004 |
) |
|
|
(1,965 |
) |
Payments for intangible assets |
|
|
(154 |
) |
|
|
(6 |
) |
Proceeds from disposal of property, plant and equipment |
|
|
— |
|
|
|
1 |
|
Payments for investments classified in Other financial assets |
|
|
(170,326 |
) |
|
|
(59,253 |
) |
Proceeds from maturity of investments classified in Other financial assets |
|
|
164,929 |
|
|
|
12,695 |
|
|
|
|
|
|
|
|
|
|
Net cash (used in)/provided by investing activities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities |
|
|
|
|
|
|
|
|
Proceeds from issuance of shares to equity holders |
|
|
38,608 |
|
|
|
17,112 |
|
Transaction costs deducted from equity |
|
|
(1,157 |
) |
|
|
(515 |
) |
Repayment of lease liabilities |
|
|
(1,866 |
) |
|
|
(1,394 |
) |
|
|
|
|
|
|
|
|
|
Net cash provided by/(used in) financing activities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net (decrease)/increase in cash and cash equivalents |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents at beginning of the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Effects of exchange rate changes and expected credit losses on cash and cash equivalents |
|
|
(2,821 |
) |
|
|
9,683 |
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents at end of the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Unaudited Condensed Consolidated Statement of Changes in Shareholders’ equity of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
share-
holders’
equity |
|
Balance as of January 1, 2022 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other comprehensive income |
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,338 |
|
|
|
1,338 |
|
| Net profit |
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
71,682 |
|
|
|
— |
|
|
|
71,682 |
|
Comprehensive income for the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Equity-settled share-based compensation |
|
|
8 |
|
|
|
— |
|
|
|
11,262 |
|
|
|
— |
|
|
|
— |
|
|
|
11,262 |
|
| Share options exercised |
|
|
|
|
|
|
— |
|
|
|
1 |
|
|
|
— |
|
|
|
— |
|
|
|
1 |
|
| Issue of share capital – net of transaction costs |
|
|
14 |
|
|
|
24 |
|
|
|
16,571 |
|
|
|
— |
|
|
|
— |
|
|
|
16,595 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance as of June 30, 2022 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance as of January 1, 2023 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other comprehensive income |
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
340 |
|
|
|
|
|
| Net loss |
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
(44,357 |
) |
|
|
— |
|
|
|
|
|
Comprehensive loss for the year |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Equity-settled share-based compensation |
|
|
8 |
|
|
|
— |
|
|
|
11,615 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
| Share options exercised |
|
|
|
|
|
|
— |
|
|
|
40 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
| Issue of share capital – net of transaction costs |
|
|
14 |
|
|
|
37 |
|
|
|
37,374 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance as of June 30, 2023 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The accompanying notes are an integral part of these condensed consolidated financial statements.
Notes to the Unaudited Condensed Consolidated Financial Statements of Immatics N.V.
Immatics N.V., together with its German subsidiary Immatics Biotechnologies GmbH and its U.S. subsidiary, Immatics US Inc., (“Immatics” or “the Group”) is a biotechnology group that is primarily engaged in the research and development of T cell redirecting immunotherapies for the treatment of cancer. Immatics N.V., a Dutch public limited liability company, was converted on July 1, 2020 from Immatics B.V., a Dutch company with limited liability. Immatics Biotechnologies GmbH (“Immatics GmbH”) and Immatics US Inc. became wholly-owned subsidiaries of Immatics N.V. as part of the ARYA Merger on July 1, 2020.
Immatics N.V. is registered with the commercial register at the Netherlands Chamber of Commerce under RSIN 861058926 with a corporate seat in Amsterdam and is located at Paul-Ehrlich Str. 15 in 72076 Tübingen, Germany.
These interim condensed consolidated financial statements of the Group for the three and six months ended June 30, 2023, were authorized for issue by the Audit Committee of Immatics N.V. on August 17, 2023.
2. Significant events and changes in the current reporting period
The following significant events or transactions occurred during the three and six months ended June 30, 2023.
Opt-In
of
TCR-T
Candidate from ongoing collaboration with BMS
Immatics GmbH entered into a License agreement (the “BMS
Opt-In
agreement”) with Bristol-Myer-Squibb Company (“BMS”). The agreement became effective on April 28, 2023. Pursuant to the BMS
Opt-In
agreement, the Group received an option exercise fee in the amount of $
15 million (€
13.7 million).
Under the 2019 agreement with BMS, Immatics granted BMS the option to enter into a
pre-negotiated
license agreement on a target-by-target basis. Immatics developed individual
TCR-T
products directed against targets under the terms of the 2019 agreement. Under the BMS
Opt-In
agreement signed on April 28, 2023, BMS exercised its first option and entered into an exclusive license agreement for one target.
The BMS
Opt-In
agreement created the right for BMS to receive the exclusive license and the right for Immatics to receive the
Opt-In
exercise fee as well as potential future milestones and royalties. Immatics promised an additional distinct performance obligation under the
Opt-in
agreement which is to issue the license to BMS. The price of the contract increases by an amount of consideration that reflects the entity’s stand-alone selling price of the license. The license grants BMS a right to use the license as no further work from Immatics is required under the agreement.
The potential milestone and royalty payments are accounted for under the most likely method. No variable payment is considered likely, therefore, no variable payments are considered as part of the transaction price.
For the three and six months ended June 30, 2023, the Group recognized €
13.7 million of revenue related to the BMS
Opt-In
agreement.
Macroeconomic environment
Currently, multiple global uncertainties are existing.
The conflict between Russia and Ukraine has resulted, and is expected to further result, in significant disruption, instability and volatility in global markets, as well as higher energy and other commodity prices. Since the Company is not currently conducting any business or receiving any material services from vendors located in Russia or Ukraine, it does not expect that the ongoing war will have a direct impact on its operations in the near term. However, the Company may be indirectly affected by price increases or certain policy changes, such as new tax legislation, economic sanctions and comparable measures. In addition, other geopolitical instabilities might impact the Group in the future.
During the six months ended June 30, 2023, Silicon Valley Bank and Credit Suisse, two large banks, as well as other smaller banks, were subject to liquidity problems. The Group does not hold deposits or securities with any of the affected banks. While the banking system remained stable overall, we will continue to closely monitor the situation.
While there is currently no material direct risk identified for the Group from
COVID-19,
Immatics will continue to closely monitor the effects of the pandemic as well.
3. Significant accounting policies
The interim condensed consolidated financial statements of the Group as of June 30, 2023 and for the three and six months ended June 30, 2023 and 2022 have been prepared on a going concern basis in accordance with International Accounting Standard 34 (“Interim Financial Reporting”), as issued by the International Accounting Standards Board (“IASB”) and have not been audited or reviewed by a statutory auditor.
In accordance with IAS 34, the interim condensed consolidated financial statements do not include all the information and disclosures required in the annual financial statements and should be read in conjunction with the Group’s annual financial statements for the year ended December 31, 2022, which have been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the IASB, taking into account the recommendations of the International Financial Reporting Standards Interpretations Committee (“IFRS IC”). In these condensed notes to the consolidated financial statements, information is provided primarily on the items for which there have been significant changes compared with the consolidated financial statements of the Group for fiscal year 2022.
The interim condensed consolidated financial statements are presented in Euros. Amounts are stated in thousands of Euros, unless otherwise indicated. For technical reasons, the information provided in these financial statements may contain rounding differences of +/- one unit.
The accounting policies adopted in the preparation of the interim condensed consolidated financial statements are consistent with those followed in the preparation of the Group’s annual consolidated financial statements for the year ended December 31, 2022. The new and amended standards and interpretations applicable for the first time as of January 1, 2023, as disclosed in the notes to the consolidated financial statements for the year ended December 31, 2022, had no impact on the interim condensed consolidated financial statements of the Group for the three and six months ended June 30, 2023.
Estimates and assumptions have to be made in the interim consolidated financial statements as of June 30, 2023. These have an impact on the amount and disclosure of the recognized assets and liabilities, income and expenses, and contingent liabilities. The estimates and judgments are basically unchanged from the circumstances described in the consolidated financial statements of the Group for the fiscal year 2022. Developments deviating from this may result in the amounts that arise deviating from the original estimates. These possible developments are outside the sphere of influence of the management.
Revision of previously issued financial statements
During the preparation of the unaudited interim consolidated financial statements for the three and nine months ended September 30, 2022, the Group identified an error in the presentation of ‘Net foreign exchange differences’ and ‘Effects of exchange rate changes on cash and cash equivalents’ in the statement of cash flows. The error resulted in a presentation of effects from exchange rate changes on
non-functional
currency denominated cash and cash equivalents in Immatics N.V. and Immatics GmbH as operating cash flow instead of the presentation as
non-cash
items in effects of exchange rate changes on cash and cash equivalents.
This error had no impact on the Company’s consolidated statements of financial position, of profit/(loss), of comprehensive income/(loss) and of consolidated statements of changes in equity. The Company assessed the materiality of these errors on the previously issued consolidated financial statements and concluded that the errors were not material to any period presented. The impact of the revision of the previously issued financial statements is as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, 2022 |
|
| |
|
|
|
|
|
|
|
|
|
Net foreign exchange differences and expected credit losses |
|
|
115 |
|
|
|
(7,949 |
) |
|
|
(7,834 |
) |
Net cash provided by/(used in) operating activities |
|
|
163,722 |
|
|
|
(7,949 |
) |
|
|
155,773 |
|
Net cash (used in)/provided by investing activities |
|
|
(48,528 |
) |
|
|
— |
|
|
|
(48,528 |
) |
Net cash (used in)/provided by financing activities |
|
|
15,203 |
|
|
|
— |
|
|
|
15,203 |
|
Net increase/(decrease) in cash and cash equivalents |
|
|
130,397 |
|
|
|
(7,949 |
) |
|
|
122,448 |
|
Cash and cash equivalents at beginning of period |
|
|
132,994 |
|
|
|
— |
|
|
|
132,994 |
|
Effects of exchange rate changes on cash and cash equivalents |
|
|
1,734 |
|
|
|
7,949 |
|
|
|
9,683 |
|
Cash and cash equivalents at end of period |
|
|
265,125 |
|
|
|
— |
|
|
|
265,125 |
|
The Group manages its operations as a single segment for the purpose of assessing performance and making operating decisions. The Group’s focus is on the research and development of T cell redirecting immunotherapies for the treatment of cancer. The Chief Executive Officer is the chief operating decision maker who regularly reviews the consolidated operating results and makes decisions about the allocation of the Group’s resources.
5. Revenue from collaboration agreements
The Group currently earns revenue through strategic collaboration agreements with third party pharmaceutical and biotechnology companies. As of June 30, 2023, the Group had four strategic collaboration agreements in place after the collaboration with GSK plc (“GSK”) was terminated in 2022. Three of the four collaboration programs are still at
pre-clinical
development stage and IMA401, which is subject of a collaboration with Bristol Myers Squibb (“BMS”) is in clinical development.
Revenue from collaboration agreements were realized with the following partners:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
| Genmab, Denmark |
|
|
915 |
|
|
|
4,125 |
|
|
|
215 |
|
|
|
7,044 |
|
| BMS, United States |
|
|
21,439 |
|
|
|
12,107 |
|
|
|
31,935 |
|
|
|
110,532 |
|
| GSK, United Kingdom |
|
|
— |
|
|
|
983 |
|
|
|
— |
|
|
|
2,547 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
As of June 30, 2023, the Group has not recognized any milestone revenue under the collaboration agreements, due to the scientific uncertainty of achieving the milestones or the successful commercialization of a product. As of June 30, 2023, Immatics had not received any milestone or royalty payments in connection with the collaboration agreements. The Group expects to recognize the remaining deferred revenue balance as revenue as it performs the related performance obligations under each contract.
The revenue for the three and six months ended June 30, 2023 from the collaboration agreement with BMS includes the
Opt-in
payment of €
13.7 million. The remaining revenue for the three and six months ended June 30, 2023 from the collaboration with BMS is the revenue recognized over time on a
basis. The revenue for the six months ended June 30, 2023 from the collaboration agreement with Genmab decreases in comparison to the revenue for the three months ended June 30, 2023, due to the negative revenue for the three months ended March 31, 2023, which is a result of changes to the inputs in the
model resulting from an increase in the expected cost of the collaboration. The revenue from collaboration agreements with BMS includes the revenue regarding the
license for IMA401 amounting to €
91.3 million for the six months ended June 30, 2022.
Deferred revenue related to the collaboration agreements consists of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Current |
|
|
67,997 |
|
|
|
64,957 |
|
| |
|
|
53,559 |
|
|
|
75,759 |
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
Deferred revenues are contract liabilities within the scope of IFRS 15. The Group recognized expenses related to the amortization of capitalized cost of obtaining a contract of €0.1 million and €0.2 million for the three months ended June 30, 2023 and June 30, 2022.
The Group recognized expenses related to the amortization of capitalized cost of obtaining a contract of €0.1 million and €0.3 million for the six months ended June 30, 2023 and June 30, 2022.
Other financial income and financial expenses consist of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Interest income |
|
|
2,744 |
|
|
|
69 |
|
|
|
4,999 |
|
|
|
75 |
|
| Foreign currency gains |
|
|
1,210 |
|
|
|
6,946 |
|
|
|
1,749 |
|
|
|
8,699 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Interest expenses |
|
|
(206 |
) |
|
|
(376 |
) |
|
|
(401 |
) |
|
|
(538 |
) |
| Foreign currency losses |
|
|
(805 |
) |
|
|
(31 |
) |
|
|
(4,119 |
) |
|
|
(986 |
) |
| Losses on financial instruments |
|
|
(133 |
) |
|
|
— |
|
|
|
(133 |
) |
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Change in fair value of liabilities for warrants |
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income mainly results from short-term deposits as well as cash balances for the three and six months ended June 30, 2023. Interest expenses mainly results from leases.
Foreign currency gains and losses mainly consist of realized and unrealized gains and losses in connection with our USD holdings of cash and cash equivalents, short-term deposits as well as bonds.
The fair value of the warrants decreased from €2.35 ($2.51) per warrant as of December 31, 2022 to €1.32 ($1.44) as of March 31, 2023 and increased to €3.15 ($3.42) as of June 30, 2023. The result is an increase in fair value of warrant liabilities of €13.1 million for the three months ended June 30, 2023 and an increase in fair value of warrant liabilities of €5.7 million for the six months ended June 30, 2023.
The fair value of the warrants decreased from €3.88 ($4.39) per warrant as of December 31, 2021 to €1.58 ($1.75) as of March 31, 2022 and increased to €1.96 ($2.04) as of June 30, 2022. The result is an increase in fair value of warrant liabilities of €2.8 million for the three months ended June 30, 2022 and a decrease in fair value of warrant liabilities of €13.7 million for the six months ended June 30, 2022.
During the three and six months ended June 30, 2023, the Group generated a net loss. The Group correspondingly recognized no income tax expense and no equivalent current tax liability for the three and six months ended June 30, 2023. During the three months ended March 31, 2022, the Group generated a net income due to the recognition of revenue in connection with the license component of the BMS collaboration agreement on IMA401. This
one-time
revenue is not accounted for under German GAAP and consequently under German tax accounting. Instead, the Group recognizes revenue for the BMS agreement over the period of the clinical trial service.
The deferred tax liability arising from the temporary difference related to delayed revenue recognition under German tax accounting is offset by deferred tax assets on tax losses carried forward that were previously not capitalized due to the Groups expectation of generating taxable losses in the foreseeable future. During the three and six months ended June 30, 2023 and 2022, the Group’s German operations were subject to a statutory tax rate of 28.5% and the Group´s U.S. operations were subject to a federal corporate income tax rate of 21%.
For Immatics GmbH, the Group recognized an income tax expense and an equivalent current tax liability in the amount of €
1.2 million for the three and six months ended June 30, 2022. The income tax expense is calculated based on taxable income of Immatics GmbH for the three and six months ended June 30, 2022 and does not take into account potential income or loss of the following quarters. The Group applied the estimated effective tax rate
for
the financial year 2022 to the taxable income for the three and six months ended June 30, 2022. Since no deferred tax assets have been recognized as of December 31, 2021, the Group took into account the tax losses carried forward that can be used to offset the taxable income generated in the three and six months ended June 30, 2022. In accordance with §10d para 2 EStG (German income tax code),
60% of an income of a given year can be offset with tax losses carried forward. Accordingly,
40% of the income before tax of Immatics GmbH are subject to income tax.
As the profit generated in the three and six months ended June 30, 2022, is considered a
one-time
profit, no deferred tax assets exceeding the deferred tax liability for temporary differences have been recognized in respect of tax losses carried forward. The current assessment regarding the usability of deferred tax assets may change, depending on the Group’s taxable income in future years, which could result in the recognition of deferred tax assets.
The Group continued to generate losses for all entities within the Group during the three and six months ended June 30, 2023 as well as for all entities apart from Immatics GmbH during the three and six months ended June 30, 2022.
Due to changes in ownership in prior periods, there are certain limitations on tax losses carried forward for net operating losses incurred by Immatics US, Inc., under Section 382 of the U.S. Internal Revenue Code.
Immatics N.V. has two share-based payment plans. In June 2020, Immatics N.V. established an initial equity incentive plan (“2020 Equity Plan”). At the Annual General Meeting on June 13, 2022, Immatics´s shareholders approved the Company’s 2022 stock option and incentive plan (“2022 Equity Plan”). The 2022 Equity Plan allows the company to grant additional options, other than that, it does not materially differ from the 2020 Equity Plan.
Immatics GmbH previously issued share-based awards to employees under two different plans. Under the GmbH Stock Appreciation Program 2010 (the “2010 Plan”), the Company issued stock appreciation rights (“SARs”), which the Group accounted for as cash-settled awards. Under the Immatics Biotechnologies 2016 Equity Incentive Plan (“2016 Plan”), the Company issued tandem awards, which contained the possibility to function as either a SAR or a stock option.
The Group accounted for awards issued under the 2016 Plan, which were redeemable in either cash or equity shares at the Group’s discretion, as equity settled.
As part of the ARYA Merger, all outstanding awards under the 2010 Plan and 2016 Plan were replaced by a combination of cash payments and share-based awards under the 2020 Equity Plan in Immatics N.V. Under the 2020 Plan, management and employees have been granted different types of options, all of which are equity-settled transactions. As part of the replacement, active employees and management members received stock options (“Matching Stock Options”) to acquire shares in Immatics N.V. The Matching Stock Options have an exercise price of $
10.00 and vested fully on
July 31, 2021. The awards have a
ten-year
contract life.
Matching Stock Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
Weighted average
exercise price in USD |
|
|
|
|
| Matching Stock Options outstanding on January 1, |
|
|
10.00 |
|
|
|
1,348,004 |
|
| Matching Stock Options forfeited |
|
|
— |
|
|
|
— |
|
| Matching Stock Options exercised |
|
|
10.00 |
|
|
|
720 |
|
| Matching Stock Options expired |
|
|
10.00 |
|
|
|
4,280 |
|
| Matching Stock Options outstanding on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Matching Stock Options exercisable on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Weighted average remaining contract life (years) |
|
|
7.01 |
|
|
|
|
|
For any outstanding 2016 Plan and 2010 Plan awards scheduled to vest on or after January 1, 2021, employees received replacement stock options (“Converted Options”) to acquire shares in Immatics N.V. The Converted Options have comparable terms to previous awards, with revised exercise prices reflecting the reorganized capital structure of Immatics. The options granted under the 2020 Equity Plan that gives employees the right to acquire shares in Immatics N.V., are accounted for as a modification under IFRS 2, with the incremental fair value expensed over the remaining vesting period.
The incremental fair value is the difference between the fair value of the options to purchase ordinary shares under the 2020 Equity Plan to acquire shares in Immatics N.V., and the fair value of the exchanged unvested SAR (both measured at the date on which the replacement award is issued).
Converted Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Converted Options outstanding on January 1, |
|
|
2.74 |
|
|
|
525,181 |
|
| Converted Options forfeited |
|
|
1.14 |
|
|
|
909 |
|
| Converted Options exercised |
|
|
1.28 |
|
|
|
11,341 |
|
| Converted Options expired |
|
|
1.17 |
|
|
|
8 |
|
| Converted Options outstanding on June 30, |
|
|
2.78 |
|
|
|
512,923 |
|
| Converted Options exercisable on June 30, |
|
|
2.78 |
|
|
|
469,161 |
|
| Weighted average remaining contract life (years) |
|
|
4.51 |
|
|
|
|
|
Under the 2020 Plan and the 2022 Plan, Immatics also issues employee stock options with a service requirement (“Service Options”), to acquire shares of Immatics N.V. The service-based options for employees including management will vest on a four-year time-based vesting schedule. Under the 2022 Plan, annual service options for members of the Board of Directors will vest entirely after one year. Service Options are granted on a recurring basis.
The Company granted Service Options on April 3, 2023 and on June 27, 2023, which were accounted for using the respective grant date fair value. Immatics applied a Black Scholes pricing model to estimate the fair value of the Service Options, with a weighted average fair value of $6.97 for Service Options granted during the six months ended June 30, 2023.
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Exercise price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Underlying share price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Volatility |
|
|
84.84 |
% |
|
|
86.76 |
% |
| Time period (years) |
|
|
6.11 |
|
|
|
5.74 |
|
| Risk free rate |
|
|
3.47 |
% |
|
|
3.94 |
% |
| Dividend yield |
|
|
0.00 |
% |
|
|
0.00 |
% |
Service Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Service Options outstanding on January 1, |
|
|
10.07 |
|
|
|
6,129,160 |
|
| Service Options granted in 2023 |
|
|
9.48 |
|
|
|
506,163 |
|
| Service Options forfeited |
|
|
9.80 |
|
|
|
145,494 |
|
| Service Options exercised |
|
|
9.51 |
|
|
|
2,707 |
|
| Service Options expired |
|
|
10.75 |
|
|
|
17,825 |
|
| Service Options outstanding on June 30, |
|
|
10.02 |
|
|
|
6,469,297 |
|
| Service Options exercisable on June 30, |
|
|
10.07 |
|
|
|
2,135,617 |
|
| Weighted average remaining contract life (years) |
|
|
8.57 |
|
|
|
|
|
In addition, after the closing of the ARYA Merger certain executive officers and key personnel of the Group received under the 2020 Equity Plan performance-based options (“PSUs”), vesting based on both the achievement of market capitalization milestones and satisfaction of a four-year time-based vesting schedule. The PSUs are split into three equal tranches. The performance criteria for each of the three respective tranches requires Immatics to achieve a market capitalization of at least $1.5 billion, $2 billion and $3 billion, respectively.
A Monte-Carlo simulation model has been used to measure the fair value at grant date of the PSUs. This model incorporates the impact of the performance criteria regarding market capitalization in the calculation of the award’s fair value at grant date.
PSUs outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| PSUs outstanding on January 1, |
|
|
10.08 |
|
|
|
3,666,000 |
|
| PSUs granted in 2023 |
|
|
— |
|
|
|
— |
|
| PSUs forfeited |
|
|
10.00 |
|
|
|
12,000 |
|
| PSUs outstanding on June 30, |
|
|
10.08 |
|
|
|
3,654,000 |
|
| PSUs exercisable on June 30, |
|
|
— |
|
|
|
— |
|
| Weighted average remaining contract life (years) |
|
|
7.05 |
|
|
|
|
|
The Group recognized total employee-related share-based compensation expenses from all plans, during the three and six months ended June 30, 2023 and 2022 as set out below:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Research and development expenses |
|
|
(3,282 |
) |
|
|
(3,107 |
) |
|
|
(6,815 |
) |
|
|
(6,375 |
) |
| General and administrative expenses |
|
|
(2,231 |
) |
|
|
(2,453 |
) |
|
|
(4,800 |
) |
|
|
(4,887 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
9. Other current and
non-current
assets
Other current assets consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Prepaid expenses |
|
|
9,546 |
|
|
|
10,450 |
|
| Value added tax receivables |
|
|
1,605 |
|
|
|
1,031 |
|
| Other assets |
|
|
5,517 |
|
|
|
2,357 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses include expenses for licenses and software of €6.7 million as of June 30, 2023 and €7.4 million as of December 31, 2022 and prepaid insurance expenses of €0.2 million as of June 30, 2023 and €1.2 million as of December 31, 2022. The Group accrued €0.3 million as of June 30, 2023 and €0.4 million as of December 31, 2022 of incremental cost for the successful arrangement of the BMS collaboration signed in 2019 and the Genmab collaboration agreement.
Additionally, prepaid expenses include expenses for maintenance of €0.7 million as of June 30, 2023 and €0.7 million as of December 31, 2022. The remaining amount is mainly related to prepaid expenses for contract research organizations and travel expenses.
Other assets include receivables from lease incentive, capital gains tax and prepaid deposit expenses.
Other
non-current
assets consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Prepaid expenses |
|
|
1,365 |
|
|
|
1,906 |
|
| Other assets |
|
|
607 |
|
|
|
639 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Prepaid expenses include the
non-current
portion of prepayments for licensing agreements of €
1.1 million as of June 30, 2023 and €
1.5 million as of December 31, 2022 , prepaid maintenance expenses of €
0.2 million as of June 30, 2023 and €
0.3 million as of December 31, 2022 and accrued incremental cost of the BMS and Genmab collaboration agreement of €
0.1 million as of June 30, 2023 and €
0.1 million as of December 31, 2022. Other assets include the
non-current
portion for prepaid deposit expenses.
10. Property, plant and equipment, intangible assets and
assets
During the three months ended June 30, 2023 and June 30, 2022, the Group acquired property, plant and equ
ip
ment and intangible assets in the amount of €
11.6 million and €
1.1 million, respectively.
During the six months ended June 30, 2023 and June 30, 2022, the Group acquired property, plant and equipment and intangible assets in the amount of €15.7 million and €2.2 million, respectively.
The Group’s additions include leasehold improvements for the research and commercial GMP manufacturing facility construction in Houston, Texas of €
9.8 million for the six months ended June 30, 2023. During the six months ended June 30, 2023, a new office space in Tübingen, Germany, lease term extensions and rent increases to existing lease agreements resulted in an addition in
assets and corresponding lease liability in the amount of €
3.8 million.
Provisions consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Provision for bonuses |
|
|
3,117 |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
These amounts include provisions for the Group’s annual employee bonuses.
Accounts payables consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Accounts payables |
|
|
5,473 |
|
|
|
4,025 |
|
| Accrued liabilities |
|
|
14,431 |
|
|
|
9,031 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
13. Other current liabilities
Other current liabilities consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Income tax liability |
|
|
4,298 |
|
|
|
4,298 |
|
| Payroll tax |
|
|
1,840 |
|
|
|
3,426 |
|
| Accrual for vacation |
|
|
1,400 |
|
|
|
806 |
|
| Accrued bonuses |
|
|
— |
|
|
|
680 |
|
| Other liabilities |
|
|
391 |
|
|
|
156 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other current liabilities are
non-interest-bearing
and are due within
one year. The carrying amounts of other current liabilities represents fair values due to their short-term nature.
As of June 30, 2023 and December 31, 2022, the total number of ordinary shares of Immatics N.V. outstanding is 80,397,851 and 76,670,699 with a par value of €0.01, respectively.
During the three months ended June 30, 2023, the Group issued
3.7 million shares under the ATM agreement with SVB Securities LLC and collected a gross amount of €
38.6 million less transaction costs of €
1.2 million, resulting in an increase in share capital of €
37 thousand and share premium of €
37.4 million. Additionally, the number of ordinary shares increased during the six months ended June 30, 2023, due to exercised share
options
from the Group’s equity incentive plan.
Other reserves are related to accumulated foreign currency translation amounts associated with the Group’s U.S. operations.
15. Related party disclosures
During the three and six months ended June 30, 2023, the Group did not enter into any new related-party transactions with its key management personnel or with related entities other than the granting of a total of
175,000 Service options to its Board of Directors for the
three and
six months ended June 30, 2023.
16. Financial Instruments
Set out below are the carrying amounts and fair values of the Group’s financial instruments that are carried in the interim condensed consolidated financial statements.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Carrying amount per measurement category |
|
| |
|
|
|
|
|
|
|
|
At fair value
through profit
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
130,405 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
217,222 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
330 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
5,158 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
19,212 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
58 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
22,622 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
16,822 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Carrying amount per measurement category |
|
|
|
|
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
148,519 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
154,930 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
58,756 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
1,111 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
2,402 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
11,735 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
54 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
16,914 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
14,563 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| * |
“Short-term deposits” and “Bonds” are classified within the balance sheet item “Other financial assets” |
In all valuation categories with the exception of Bonds, the carrying amount represents a reasonable approximation of the fair value based on the short-term maturities of these instruments. Set out below are the carrying amounts and fair values of the Group’s Bonds as of June 30, 2023 and December 31, 2022. The fair values of the financial assets and liabilities are included at the amount at which the instrument could be exchanged in a current transaction between willing parties, other than in a forced or liquidation sale.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bonds |
|
|
— |
|
|
|
— |
|
|
|
58,756 |
|
|
|
58,300 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The following methods and assumptions were used to estimate the fair values: All financial assets are categorized based on Level 1 inputs and are therefore valued using quoted (unadjusted) market prices. All financial liabilities are also categorized based on Level 1 inputs.
The bonds’ contractual cash flows represent solely payments of principal and interest and Immatics intends to hold the bonds to collect the contractual cash flows. The Group therefore accounts for the bonds as a financial asset at amortized cost. Bonds are classified as Level 1 of the fair value hierarchy, as they are listed on publicly traded markets.
Liabilities for warrants are comprised of the Immatics Warrants issued to investors with a cashless exercise mechanism as a current liability which the Company accounted for according to provisions of IAS 32. The Company measures the warrants at fair value by using the closing price of warrants at Nasdaq. The warrants are measured in each reporting period. Changes in the fair value are recognized in the Company’s Consolidated Statement of Profit/(Loss) as financial income or expenses, as appropriate. The warrants are classified as Level 1 of the fair value hierarchy. The maturity of the liabilities for warrants is dependent on the development of the share price as well as the decisions by the Immatics Warrants holders.
17. Earnings and Loss per Share
The Group reported basic and diluted loss and earnings per share during the three and six months ended June 30, 2023 and 2022. Basic and diluted loss per share and basic earnings per share are calculated by dividing the net profit or loss by the weighted-average number of ordinary shares outstanding for the reporting period. Diluted earnings per share for the six months ended June 30, 2022, are calculated by adjusting the weighted-average number of ordinary shares outstanding for any dilutive effects resulting from equity awards granted to the Board and employees of the Group as well as from publicly traded Immatics Warrants. The Group’s equity awards and Immatics Warrants for which the exercise price is exceeding the Group’s weighted average share price for the six months ended June 30, 2022, are anti-dilutive instruments and are excluded in the calculation of diluted weighted average number of ordinary shares. The Group was loss-making during the three and six months ended June 30, 2023 and during the three months ended June 30, 2022, therefore all instruments are anti-dilutive instruments and are excluded in the calculation of diluted weighted average number of ordinary shares outstanding, including the outstanding equity awards and the 7,187,500 Immatics Warrants issued in 2020 and outstanding as of June 30, 2023.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
(Euros in thousands, except share and per share data) |
|
|
(Euros in thousands, except share and per share data) |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
| Diluted |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
Weighted average shares outstanding: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
63,932,449 |
|
| Diluted |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
64,477,256 |
|
18. Events occurring after the reporting period
The Group evaluated subsequent events for recognition or disclosure through August 17, 2023.
On July 19, 2023, the Group closed a private placement transaction of
2,419,818 ordinary shares with a subscription price of $
14.46 per ordinary share with BMS. The Group received gross proceeds of approximately $
35 million, before deducting transaction expenses and intends to use the net proceeds to fund the continued research and development of the
Group’s
pipeline, the manufacturing and production of product candidates and for working capital.
After the reporting period, the Group issued 1.8 million shares under the ATM agreement with SVB Securities LLC and collected a gross amount of €20.4 million ($22.2 million).
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND
RESULTS OF OPERATIONS
The following discussion and analysis is based on the financial information of Immatics N.V., together with its German subsidiary Immatics Biotechnologies GmbH and its U.S. subsidiary, Immatics US, Inc. (“Immatics”, the “Company”, the “Group”, “we”, “our”). You should read the following discussion and analysis of our financial condition and results of operations together with our unaudited interim condensed consolidated financial statements for the three and six month-period ended June 30, 2023 and 2022 included in this interim report. You should also read our operating and financial review and prospects and our Consolidated Financial Statements for fiscal year 2022, and the notes thereto, in our Annual Report on Form 20-F for the year ended December 31, 2022, filed with the SEC on March 22, 2023 (the “Annual Report”). The following discussion is based on the financial information of Immatics prepared in accordance with International Financial Reporting Standards (“IFRS”), which may differ in material respects from generally accepted accounting principles in other jurisdictions, including U.S. generally accepted accounting principles.
Overview
We are a clinical-stage biotechnology company dedicated to the development of T cell receptor (“TCR”)-based immunotherapies for the treatment of cancer. Our purpose is to deliver a meaningful impact on the lives of cancer patients by developing novel TCR-based immunotherapies that are designed to achieve effect beyond an incremental clinical benefit. Our focus is the development of product candidates for the treatment of patients with solid tumors, who are inadequately served by existing treatment modalities. We strive to become an industry leading, fully integrated global biopharmaceutical company engaged in developing, manufacturing and commercializing TCR immunotherapies for the benefit of cancer patients, our employees, our shareholders and our partners.
By utilizing TCR-based therapeutics, we are able to direct T cells to intracellular cancer targets that are not accessible through classical antibody-based or CAR-T therapies. We believe that by identifying what we call true cancer targets and the right TCRs, we are well positioned to transform current solid tumor treatment paradigms by delivering cellular and bispecific product candidates that have the potential to substantially improve the lives of cancer patients.
We are developing our targeted immunotherapy product candidates through two distinct treatment modalities: TCR-engineered autologous (“ACTengine”) or allogeneic (“ACTallo”) Adoptive Cell Therapies (“ACT”) and antibody-like Bispecifics, also called T cell Engaging Receptors (“TCER”). Each modality is designed with distinct attributes and mechanisms of action to produce the desired therapeutic effect for a variety of cancer patient populations with different unmet medical needs. Our current pipeline comprises several proprietary TCR-based product candidates in clinical and preclinical development. In addition to our proprietary pipeline, we are collaborating with industry-leading partners, including Bristol Myers Squibb (“BMS”), Editas Medicine and Genmab, to develop multiple additional therapeutic programs covering ACT and Bispecifics.
Since our inception, we have focused on developing our technologies and executing our preclinical and clinical research programs with the aim to deliver the power of T cells to cancer patients. We do not have any products approved for sale. We have funded our operations primarily through equity financing and through payments from our collaboration partners.
We have assembled a team of 418 and 380 FTEs as of June 30, 2023 and December 31, 2022, respectively.
Through June 30, 2023 we have raised approximately €981.7 million through licensing payments from our collaborators and through private and public placements of securities. We are holding Cash and cash equivalents and Other financial assets of €347.6 million as of June 30, 2023. We believe that we have sufficient capital resources to fund our operations through at least the next 12 months.
Since our inception, we have incurred net losses, which have been significant in recent periods. The net profit for the year ended December 31, 2022 was due to a one-time upfront payment. We expect to continue to incur significant expenses and increasing net losses for the foreseeable future as we continue our research and development efforts and seek to obtain regulatory approval for and commercialize our product candidates. Our future profitability will be dependent upon the successful development, approval and commercialization of our product candidates and achieving a level of revenues adequate to support our cost structure. We may never achieve profitability and, unless and until we do, we will continue to need to raise additional capital. Our net losses may fluctuate significantly from period to period and year to year.
Global Developments
Currently, multiple global uncertainties are existing.
The conflict between Russia and Ukraine has resulted, and is expected to further result, in significant disruption, instability and volatility in global markets, as well as higher energy and other commodity prices. Since the Company is not currently conducting any business or receiving any material services from vendors located in Russia or Ukraine, it does not expect that the ongoing war will have a direct impact on its operations in the near term. However, the Company may be indirectly affected by price increases or certain policy changes in Germany, such as new tax legislation, economic sanctions and comparable measures. In addition, other geopolitical instabilities might impact the Group in the future.
During the six months ended June 30, 2023, Silicon Valley Bank as well as Credit Suisse, two large banks, as well as other smaller banks, were subject to liquidity problems. The Group does not hold deposits or securities with any of the affected banks. While the banking system remained stable overall, we will continue to closely monitor the situation.
While there is currently no material direct risk identified for the Group from COVID-19, Immatics will continue to closely monitor the effects of the pandemic as well.
Our Strategy
Our mission is to deliver the power of T cells to cancer patients. We seek to execute the following strategy to develop TCR-based immunotherapies for the treatment of cancer, maximizing the value of our technology platforms and the broad portfolio of product candidates:
| |
• |
|
Realize the full multi-cancer opportunity of PRAME. We believe PRAME (Preferentially Expressed Antigen in Melanoma) is one of the most promising and most prevalent, clinically validated solid tumor targets known to date. To leverage its full potential and maximize patient reach, we are: (1) focusing and accelerating the development of our ACTengine IMA203 TCR-T towards pivotal trials, (2) expanding the patient population that might benefit from a PRAME-targeting therapy by developing an off-the-shelf biologic TCER IMA402 with a different mechanism of action without the requirement for administration at specialized medical centers and (3) expanding beyond HLA-A*02 by investigating new target-TCR pairs for PRAME epitopes binding to other HLA types. |
| |
• |
|
Advance our pipeline of innovative ACTengine TCR-T product candidates. In addition to our most advanced TCR-T product candidate ACTengine IMA203, our pipeline is strengthened by innovative cell therapy programs in development. ACTengine IMA204 is directed against the novel tumor stroma target COL6A3 that is highly prevalent across many different solid tumor types and provides a promising and innovate therapeutic opportunity for a broad patient population as monotherapy or in combination with TCR-T cells directed against targets presented on tumor cells. IMA204 uses an affinity maturated CD8-independent, next-generation TCR that engages both CD4 and CD8 T cells without the need of CD8 co-transduction. Moreover, we continue to actively investigate multiple other next-generation enhancement and combination strategies to render ACTengine T cells even more potent to combat solid tumors and enhance tolerability and ease of use of our product candidates. |
| |
• |
|
Advance our pipeline of next-generation, half-life extended TCR Bispecifics. In addition to PRAME TCER IMA402 which entered clinical development in August 2023, we have a broad portfolio of clinical and preclinical TCR Bispecifics. Phase 1 clinical development commenced in May 2022 for our most advanced TCER program IMA401 targeting MAGEA4/8. IMA401 is being developed in collaboration with BMS and we seek to deliver clinical PoC for IMA401 and thus our TCER platform as fast as possible. We also continue development of several innovative preclinical TCER product candidates against so far undisclosed targets for our proprietary and/or partnered pipeline. IMA403 is in advanced preclinical stage with PoC studies ongoing. Additionally, TCER engineering and preclinical testing is ongoing for further TCER candidates, IMA40x, targeting peptides presented by HLA-A*02:01 and other HLA-types. Our next-generation, half-life extended TCER format used in all our candidates is designed to safely apply high drug doses for activity in a broad range of tumors, even with low target density, and to achieve a patient-convenient dosing schedule. |
| |
• |
|
Further enhance our cell therapy manufacturing capabilities. Our proprietary ACTengine manufacturing process is generating TCR-T cells that have been shown to achieve a high rate of objective responses, infiltrate the patient’s tumor and function in the solid tumor microenvironment. With a manufacturing time of approximately one week and an accelerated product release time, we are aiming at shortening the vein-to-vein time and to provide products to patients as fast as possible. We have implemented several manufacturing enhancements in our IMA203 Phase 1b trial (including monocyte depletion) that enhanced key features of the cell product and were focused on robustness, quality, and speed of product release. We continue to implement minor improvements to prepare for pivotal trials and potential commercialization. We are currently expanding our cell therapy manufacturing capabilities with construction of a state-of-the-art GMP manufacturing facility for registration-directed and commercial production of ACTengine TCR-T products, including IMA203. The manufacturing facility is expected to be operational in 2024. |
| |
• |
|
Develop allogeneic off-the-shelf cell therapies. We aim to increase the commercial opportunity of cell therapies by supplying products to patients more quickly and at lower cost with our off-the-shelf cell therapy approach, ACTallo. ACTallo is our proprietary allogeneic adoptive cell therapy platform based on gamma delta T cells sourced from healthy donors and designed to create hundreds of doses from one single donor leukapheresis. In June 2022, we entered into strategic collaborations with Bristol Myers Squibb and Editas Medicine with the goal to develop transformative next-generation allogeneic gamma delta TCR-T/CAR-T programs with enhanced persistence, safety and potency by combining our proprietary ACTallo platform with Bristol Myers Squibb’s next-gen technologies and Editas Medicine’s CRISPR gene editing technology. |
| |
• |
|
Leverage the full potential of strategic collaborations. We have entered strategic collaborations with key industry partners to maintain our leadership position in the TCR therapeutics field and to strengthen our proprietary pipeline. We intend to generate value from these strategic collaborations by developing transformative, cutting-edge therapeutics through the combination of synergistic capabilities and technologies, and we benefit through upfront payments and potential milestone payments and royalties for product candidates that our partners successfully advance into and through clinical development and towards commercial launch. We are in discussions to potentially establish a strategic collaboration that may encompass any, some or all of the following: exclusive development and out-licensing agreement of therapeutics against a limited number of preclinical-stage TCER targets, limited access to our XPRESIDENT technology platform and/or the development of combination therapeutics. |
| |
• |
|
Strengthen our intellectual property portfolio. We intend to continuously build and maintain our intellectual property portfolio.The protection of our intellectual property assets is a foundational element of our ability to not only strengthen our product pipeline, but also to successfully defend and strengthen our position in the field of TCR therapies. |
| |
• |
|
Enhance the competitive edge of our technology platforms. Our target and TCR discovery platforms XPRESIDENT and XCEPTOR are the foundation for the further strengthening our product pipeline and our position in the field of TCR-based therapies. We have developed a suite of proprietary technologies to identify what we refer to as “true targets” and “right TCRs.” True targets are (i) naturally occurring at significant levels on native tumor tissue (in contrast to being in silico predicted or identified from cell line cultures), and (ii) highly specific to cancer cells. Right TCRs are (i) high-affinity TCRs, and (ii) highly specific to the respective cancer target, with no or minimized cross-reactivities to healthy tissues. We leverage this unique knowledge to develop a pipeline of transformative TCR-based product candidates. Our goal is to maintain and expand our competitive edge in highly differentiated platform technologies aimed at developing additional, better and highly innovative product candidates within shorter development timelines, for mid- and long-term value generation as part of our own or partnered pipeline. |
Portfolio Update
Adoptive Cell Therapy Programs
ACTengine® IMA203: ACTengine® IMA203 TCR-T against PRAME is currently being evaluated in an ongoing Phase 1b dose expansion trial.
| |
• |
|
As per the latest data cut-off of April 4, 2023, ACTengine® IMA203 TCR-T monotherapy Cohort A showed a 67% confirmed objective response rate (cORR) in an interim clinical update announced on May 2, 2023. The data covered 11 heavily pre-treated patients; the median duration of response was not reached at a median follow-up time of 8.5 months. Patients were infused with IMA203 TCR-T cells at dose level (DL) 4 or DL5 with a mean total infused dose of 3.67x109 TCR-T cells (range 1.30-8.84x109 TCR-T cells). |
| |
• |
|
Cohort A IMA203 monotherapy TCR-T treatment continues to show manageable tolerability with no high-grade CRS and no ICANS; all 11 patients experienced expected cytopenia (Grade 1-4) associated with lymphodepletion. 10 patients (91%) had a low to moderate (Grade 1-2) cytokine release syndrome (CRS), of which 5 patients (45%) had Grade 1, and 5 patients (45%) had Grade 2 CRS. |
| |
• |
|
Objective responses were observed independent of tumor type including checkpoint-refractory and BRAF inhibitor-refractory cutaneous melanoma, platinum-resistant ovarian cancer, uveal melanoma, head and neck cancer and synovial sarcoma. Longest duration of responses were observed in cutaneous and uveal melanoma with ongoing responses at 6, 9 and 10 months post infusion at data cut-off. |
| |
• |
|
IMA203 in combination with nivolumab (Cohort B) has been de-prioritized in the last-line setting. Such a combination is being considered for the front-line setting. |
| |
• |
|
IMA203CD8 (Cohort C) is a next-generation monotherapy where IMA203 engineered T cells are co-transduced with a CD8αß co-receptor. IMA203CD8 is currently being explored in DL4a (up to 0.8x109 TCR-T cells/m2 BSA). |
| |
• |
|
Next update on Immatics’ IMA203 Phase 1b cohorts, including the projected clinical development path for PRAME-targeted TCR-T monotherapy towards registration-directed trials is planned for 4Q 2023. Immatics’ IMA203 development strategy to realize the multi-cancer opportunity of targeting PRAME is based on two pillars aimed at: |
| |
1. |
maximizing speed to market in one to two last-line solid cancer types focusing on cutaneous melanoma, uveal melanoma and potentially other tumor types with high PRAME prevalence where clinical proof-of-concept has been demonstrated, and |
| |
2. |
broad development with expansion to other cancer types, such as ovarian cancer, uterine cancer, lung cancer, breast cancer, head and neck cancer and other tumor types having a broad patient reach. |
TCR-T pipeline
| |
• |
|
Earlier this year, Bristol Myers Squibb exercised its first option and entered into a global license agreement with Immatics for the most advanced TCR-T product candidate. As part of the agreement, Immatics received an option payment of $15 million and is eligible for up to $490 million in milestone payments in addition to tiered royalties on net sales of the product. |
TCR Bispecifics Programs
Immatics’ T cell engaging receptor (TCER®) candidates are next-generation, half-life extended TCR Bispecific molecules designed to maximize efficacy while minimizing toxicities in patients through Immatics’ proprietary format using a low-affinity T cell recruiter and a high-affinity TCR domain.
| |
• |
|
TCER® IMA401 (MAGEA4/8) – Phase 1 trial to evaluate safety, tolerability and initial anti-tumor activity of TCER® IMA401 in patients with recurrent and/or refractory solid tumors is ongoing. IMA401 targets an HLA-A*02:01-presented peptide that occurs identically in two different proteins, MAGEA4 and MAGEA8. This target peptide has been selected based on natural expression in native solid tumors at particularly high target density (peptide copy number per tumor cell identified by Immatics’ proprietary quantitative mass spectrometry engine XPRESIDENT®). MAGEA4 and MAGEA8 are expressed in multiple solid cancers including lung cancer, head and neck cancer, melanoma, ovarian cancer, sarcoma and others. IMA401 is being developed in collaboration with Bristol Myers Squibb. |
| |
• |
|
TCER® IMA402 (PRAME) – Immatics submitted a clinical trial application (Clinical Trial Application (CTA) is the European equivalent of an Investigational New Drug (IND) application) to the Paul-Ehrlich-Institute (PEI) in April 2023. Following CTA acceptance, Immatics initiated the Phase 1/2 trial investigating the company’s fully owned TCER® candidate IMA402 in patients with recurrent and/or refractory solid tumors in August. Initial focus indications are cutaneous and uveal melanoma, ovarian cancer, lung cancer, uterine cancer and synovial sarcoma, among others. A first clinical data update is planned for 2024. IMA402 targets an HLA-A*02:01-presented peptide derived from the tumor antigen PRAME. This target peptide has been selected based on natural expression in native solid primary tumors and metastases at particularly high target density (peptide copy number per tumor cell identified by Immatics’ proprietary quantitative mass spectrometry engine XPRESIDENT®). |
Components of Operating Results
Revenue from Collaboration Agreements
To date, we have not generated any revenue from the sale of pharmaceutical products. Our revenue has been solely derived from our collaboration agreements, such as with BMS and Genmab.
Our revenue from collaboration agreements consists of upfront payments as well as reimbursement of research and development expenses. Upfront payments allocated to the obligation to perform research and development services are initially recorded on our statement of financial position as deferred revenue and are subsequently recognized as revenue on a cost-to-cost measurement basis, in accordance with our accounting policy as described further under “Critical Accounting Estimates.”
As part of the collaboration arrangements, we grant exclusive licensing rights for the development and commercialization of future product candidates, developed for specified targets defined in the respective collaboration agreement. We carry out our research activities using our proprietary technology and know-how, participate in joint steering committees, and prepare data packages. In three of our four current collaboration agreements, these commitments represent one combined performance obligation, because the research activities are mutually dependent and the collaborator is unable to derive significant benefit from our access to these targets without our research activities, which are highly specialized and cannot be performed by other organizations. For the collaboration signed with BMS in December 2021, we identified two separate performance obligations, because the license is a distinct obligation and the running of the clinical trial services will not result in a modification of the license.
The collaboration agreements resulted in €412.7 million of payments through June 30, 2023. We received a €13.7 million Opt-in payment from our collaboration partner BMS in 2023. As part of the agreements, we contribute our XPRESIDENT and other technologies, as well as commit to participating in joint research activities. In addition, we agree to license certain target rights and the potential product candidates developed under the collaboration.
Under each of our collaboration agreements, we are entitled to receive payments for certain development and commercial milestone events, in addition to royalty payments upon successful commercialization of a product. The uncertainty of achieving these milestones significantly impacts our ability to generate revenue.
Our ability to generate revenue from sales of pharmaceutical products and to become profitable depends on the successful commercialization of product candidates by us and/or by our collaboration partners. In the foreseeable future, we do not expect revenue from product sales. To the extent that existing or potential future collaborations generate revenue, our revenue may vary due to many uncertainties in the development of our product candidates and other factors.
Research and Development Expenses
Research and development expenses consist primarily of personnel-related costs (including share-based compensation) for the various research and development departments, intellectual property (“IP”) expenses, facility-related costs and amortization as well as direct expenses for clinical and preclinical programs.
Our core business is focused on the following initiatives with the goal of providing novel TCR-based immunotherapies to cancer patients:
| |
• |
|
Realize the full multi-cancer opportunity of PRAME by (1) focusing and accelerating the development of our ACTengine IMA203 TCR-T towards pivotal trials, (2) expanding the patient population that might benefit from a PRAME-targeting therapy by developing an off-the-shelf biologic TCER IMA402 and (3) expanding beyond HLA-A*02 by investigating new target-TCR pairs for PRAME epitopes binding to other HLA types; |
| |
• |
|
Advance our pipeline of innovative ACTengine TCR-T product candidates; |
| |
• |
|
Advance our pipeline of next-generation, half-life extended TCR Bispecifics; |
| |
• |
|
Enhance the commercial opportunities of cell therapies; |
| |
• |
|
Further enhance our cell therapy manufacturing capabilities; |
| |
• |
|
Leverage the full potential of strategic collaborations; |
| |
• |
|
Strengthen our intellectual property portfolio; and |
| |
• |
|
Enhance the competitive edge of our technology platforms. |
Research expenses are defined as costs incurred for current or planned investigations undertaken with the prospect of gaining new scientific or technical knowledge and understanding. All research and development costs are expensed as incurred due to scientific uncertainty.
We expect our research and development expenses to increase substantially in the future as we advance existing and future proprietary product candidates into and through clinical studies and pursue regulatory approval. The process of conducting the necessary clinical studies to obtain regulatory approval is costly and time-consuming. We expect to increase our headcount to support our continued research activities and to advance the development of our product candidates. Clinical studies generally become larger and more costly to conduct as they advance into later stages and, in the future, we will be required to make estimates for expense accruals related to clinical study expenses. At this time, we cannot reasonably estimate or know the nature, timing and estimated costs of the efforts that will be necessary to complete the development of any product candidates that we develop from our programs. Our research and development programs are at an early stage. We must demonstrate our products’ safety and efficacy through extensive clinical testing. We may experience numerous unforeseen events during, or as a result of, the testing process that could delay or prevent commercialization of our products, including but not limited to the following:
| |
• |
|
after reviewing trial results, we or our collaborators may abandon projects previously believed to be promising; |
| |
• |
|
we, our collaborators, or regulators may suspend or terminate clinical trials if the participating subjects or patients are being exposed to unacceptable health risks; |
| |
• |
|
our potential products may not achieve the desired effects or may include undesirable side effects or other characteristics that preclude regulatory approval or limit their commercial use if approved; |
| |
• |
|
we or our manufacturers may not meet the necessary standards for the production of the product candidates or may not be able to supply the product candidates in a sufficient quantity; |
| |
• |
|
regulatory authorities may find that our clinical trial design or conduct does not meet the applicable approval requirements; and |
| |
• |
|
safety and efficacy results in various human clinical trials reported in scientific and medical literature may not be indicative of results we obtain in our clinical trials. |
Clinical testing is very expensive, can take many years, and the outcome is uncertain. It could take several years before we learn the results from any clinical trial using ACT or TCR Bispecifics. The data collected from our clinical trials may not be sufficient to support approval by the FDA, the EMA or comparable regulatory authorities of our ACT or TCR Bispecific product candidates for the treatment of solid tumors. The clinical trials for our products under development may not be completed on schedule and the FDA, EMA or regulatory authorities in other countries may not ultimately approve any of our product candidates for commercial sale. If we fail to adequately demonstrate the safety and effectiveness of any product candidate under development, we may not receive regulatory approval for those product candidates, which would prevent us from generating revenues or achieving profitability.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel-related costs (including share-based compensation) for finance, legal, human resources, business development and other administrative and operational functions, professional fees, accounting and legal services, information technology and facility-related costs. These costs relate to the operation of the business, unrelated to the research and development function or any individual program.
Due to our planned increase in research and development activities as explained above, we also expect that our general and administrative expenses might increase. We might incur increased accounting, audit, legal, regulatory, compliance, director and officer insurance costs. Additionally, if and when a regulatory approval of a product candidate appears likely, we anticipate an increase in payroll and expenses as a result of our preparation for commercial operations.
Financial Result
Financial result consists of both other financial income and other financial expenses. Other financial income results primarily from interest income and foreign exchange gains. Other financial expenses consist of interest expenses related to lease liabilities, foreign exchange losses and expected credit losses. Additionally, our warrants are classified as liabilities for warrants. The change in fair value of warrant liabilities consists of the change in fair value of these warrants.
Results of Operations
Comparison of the Three and Six Months Ended June 30, 2023 and June 30, 2022
The following table summarizes our consolidated statements of operations for each period presented:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months
ended June 30, |
|
|
Six months
ended June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| |
|
(Euros in thousands, except per share data) |
|
|
(Euros in thousands, except per share data) |
|
| Revenue from collaboration agreements |
|
|
22,354 |
|
|
|
17,215 |
|
|
|
32,150 |
|
|
|
120,123 |
|
| Research and development expenses |
|
|
(27,317 |
) |
|
|
(25,216 |
) |
|
|
(54,898 |
) |
|
|
(50,360 |
) |
| General and administrative expenses |
|
|
(9,358 |
) |
|
|
(8,683 |
) |
|
|
(18,944 |
) |
|
|
(17,961 |
) |
| Other income |
|
|
6 |
|
|
|
27 |
|
|
|
948 |
|
|
|
32 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Operating result |
|
|
(14,315 |
) |
|
|
(16,657 |
) |
|
|
(40,744 |
) |
|
|
51,834 |
|
| Change in fair value of liabilities for warrants |
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
| Other financial income |
|
|
3,954 |
|
|
|
7,015 |
|
|
|
6,748 |
|
|
|
8,774 |
|
| Other financial expenses |
|
|
(1,144 |
) |
|
|
(407 |
) |
|
|
(4,653 |
) |
|
|
(1,524 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Financial result |
|
|
(10,295 |
) |
|
|
3,822 |
|
|
|
(3,613 |
) |
|
|
20,993 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Profit/(loss) before taxes |
|
|
(24,610 |
) |
|
|
(12,835 |
) |
|
|
(44,357 |
) |
|
|
72,827 |
|
| Taxes on income |
|
|
— |
|
|
|
(1,145 |
) |
|
|
— |
|
|
|
(1,145 |
) |
| Net profit/(loss) |
|
|
(24,610 |
) |
|
|
(13,980 |
) |
|
|
(44,357 |
) |
|
|
71,682 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net profit/(loss) per share: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
| Diluted |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
Revenue from Collaboration Agreements
The following table summarizes our collaboration revenue for the periods indicated:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months
ended
June 30, |
|
|
Six months
ended
June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
(Euros in thousands) |
|
|
(Euros in thousands) |
|
| Genmab, Denmark |
|
|
915 |
|
|
|
4,125 |
|
|
|
215 |
|
|
|
7,044 |
|
| BMS, United States |
|
|
21,439 |
|
|
|
12,107 |
|
|
|
31,935 |
|
|
|
110,532 |
|
| GSK, United Kingdom |
|
|
— |
|
|
|
983 |
|
|
|
— |
|
|
|
2,547 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Total |
|
|
22,354 |
|
|
|
17,215 |
|
|
|
32,150 |
|
|
|
120,123 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Our Revenue from collaboration agreements increased from €17.2 million for the three months ended June 30, 2022 to €22.4 million for the three months ended June 30, 2023. The increase in revenue of €5.2 million is mainly due to the revenue recognized regarding the BMS Opt-in payment of €13.7 million for the three months ended June 30, 2023, partially offset by lower revenue from the Genmab collaboration due to lower progress of the collaboration.
Our Revenue from collaboration agreements decreased from €120.1 million for the six months ended June 30, 2022 to €32.2 million for the six months ended June 30, 2023. The decrease in revenue of €87.9 million is mainly due to the recognized revenue regarding the right-to-use license for IMA401 amounting to €91.3 million for the six months ended June 30, 2022, partially offset by revenue recognized regarding the BMS Opt-in payment of €13.7 million for the six months ended June 30, 2023. The revenue for the six months ended June 30, 2023 from the collaboration agreement with Genmab decreased in comparison to the revenue for the three months ended June 30, 2023, due to the negative revenue for the three months ended March 31, 2023, which was a result of changes to the inputs in the cost-to-cost model resulting from an increase in the expected cost of the collaboration. The collaboration with GSK was terminated in 2022, so no further revenue was recognized for the three and six months ended June 30, 2023.
We did not achieve any milestones or receive any royalty payments in connection with our collaboration agreements during the presented periods.
Research and Development Expenses
The following table summarizes our research and development expenses for the periods indicated:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three Months
Ended
June 30, |
|
|
Six Months
Ended
June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| (Euros in thousands) |
|
|
|
|
|
|
|
|
|
|
|
|
| Direct external research and development expenses by program: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| ACT Programs |
|
|
(5,204 |
) |
|
|
(3,910 |
) |
|
|
(8,803 |
) |
|
|
(8,667 |
) |
| TCR Bispecifics Programs |
|
|
(1,274 |
) |
|
|
(1,028 |
) |
|
|
(3,590 |
) |
|
|
(2,090 |
) |
| Other programs |
|
|
(1,441 |
) |
|
|
(2,010 |
) |
|
|
(3,032 |
) |
|
|
(3,232 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sub-total direct external expenses |
|
|
(7,919 |
) |
|
|
(6,947 |
) |
|
|
(15,425 |
) |
|
|
(13,990 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Indirect research and development expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Personnel related (excluding share-based compensation) |
|
|
(9,973 |
) |
|
|
(9,265 |
) |
|
|
(19,882 |
) |
|
|
(18,244 |
) |
| Share-based compensation expenses |
|
|
(3,282 |
) |
|
|
(3,107 |
) |
|
|
(6,815 |
) |
|
|
(6,375 |
) |
| IP Expenses |
|
|
(2,213 |
) |
|
|
(2,051 |
) |
|
|
(4,563 |
) |
|
|
(4,364 |
) |
| Facility and depreciation |
|
|
(1,956 |
) |
|
|
(1,718 |
) |
|
|
(3,732 |
) |
|
|
(3,414 |
) |
| Other indirect costs |
|
|
(1,974 |
) |
|
|
(2,128 |
) |
|
|
(4,481 |
) |
|
|
(3,974 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Sub-total indirect expenses |
|
|
(19,398 |
) |
|
|
(18,268 |
) |
|
|
(39,473 |
) |
|
|
(36,371 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Total |
|
|
(27,317 |
) |
|
|
(25,216 |
) |
|
|
(54,898 |
) |
|
|
(50,360 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Direct external research and development expenses for our ACT programs increased from €3.9 million for the three months ended June 30, 2022 to €5.2 million for the three months ended June 30, 2023. This increase mainly resulted from increased activities in our clinical trials during the second quarter of 2023, which was triggered in part by an increased number of patients recruited. Direct external research and development expenses for our TCR Bispecifics programs increased from €1.0 million for the three months ended June 30, 2022 to €1.3 million for the three months ended June 30, 2023. This increase mainly resulted from additional activities in our preclinical studies for IMA402 for which we applied for clinical trial approval in April 2023 and are preparing the start of the clinical trial.
Direct external research and development expenses for our other programs such as technology platforms and collaboration agreements decreased from €2.0 million for the three months ended June 30, 2022 to €1.4 million for the three months ended June 30, 2023. This decrease mainly resulted from collaboration timeline extensions which resulted in cost shifts as well as the termination of the GSK collaboration which was effective in the fourth quarter of 2022.
Direct external research and development expenses for our ACT programs increased from €8.7 million for the six months ended June 30, 2022 to €8.8 million for the six months ended June 30, 2023. This increase mainly resulted from increased activities in our clinical trials. Direct external research and development expenses for our TCR Bispecifics programs increased from €2.1 million for the six months ended June 30, 2022 to €3.6 million for the six months ended June 30, 2023. This increase mainly resulted from additional activities in our preclinical studies for IMA402 for which we applied for clinical trial approval in April 2023.
Direct external research and development expenses for our other programs such as technology platforms and collaboration agreements decreased from €3.2 million for the six months ended June 30, 2022 to €3.0 million for the six months ended June 30, 2023. This decrease mainly resulted from collaboration timeline extensions which resulted in cost shifts as well as the termination of the GSK collaboration which was effective in the fourth quarter of 2022.
We do not allocate indirect research and development expenses by program, as our research and development personnel work across programs. Our intellectual property expenses are incurred for the protection of cancer antigen targets, T cell receptors, antibodies, bispecific molecules, and antigen discovery platforms which are beneficial to the whole research and development group rather than for specific programs. Our programs use common research and development facilities and laboratory equipment, and we also incur other costs such as general laboratory material or maintenance expenses that are incurred for commonly used activities within the whole research and development group.
Personnel-related expenses increased from €9.3 million for the three months ended June 30, 2022 to €10.0 million for the three months ended June 30, 2023. This increase resulted from our headcount growth due to our increased research and development activities including clinical trials. Share-based compensation expenses increased from €3.1 million for the three months ended June 30, 2022 to €3.3 million for the three months ended June 30, 2023. IP expenses increased from €2.1 million for the three months ended June 30, 2022 to €2.2 million for the three months ended June 30, 2023 due to our ongoing expansion of our IP portfolio. Facility and depreciation expenses increased from €1.7 million for the three months ended June 30, 2022 to €2.0 million for the three months ended June 30, 2023. This increase resulted from the acquisition of laboratory equipment and leasehold improvements. Other indirect expenses decreased from €2.1 million for the three months ended June 30, 2022 to €2.0 million for the three months ended June 30, 2023.
Personnel-related expenses increased from €18.2 million for the six months ended June 30, 2022 to €19.9 million for the six months ended June 30, 2023. This increase resulted from our headcount growth due to our increased research and development activities including clinical trials. Share-based compensation expenses increased from €6.4 million for the six months ended June 30, 2022 to €6.8 million for the six months ended June 30, 2023. IP expenses increased from €4.4 million for the six months ended June 30, 2022 to €4.6 million for the six months ended June 30, 2023 due to our ongoing expansion of our IP portfolio. Facility and depreciation expenses increased from €3.4 million for the six months ended June 30, 2022 to €3.7 million for the six months ended June 30, 2023. This increase resulted from the acquisition of laboratory equipment and leasehold improvements. Other indirect expenses increased from €4.0 million for the six months ended June 30, 2022 to €4.5 million for the six months ended June 30, 2023.
General and Administrative Expenses
The following table summarizes our General and administrative expenses for the periods indicated:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three Months Ended
June 30, |
|
|
Six Months Ended
June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| (Euros in thousands) |
|
|
|
|
|
|
|
|
|
|
|
|
| Share-based compensation expenses |
|
|
(2,231 |
) |
|
|
(2,453 |
) |
|
|
(4,800 |
) |
|
|
(4,887 |
) |
| Personnel related (excluding share-based compensation) |
|
|
(2,891 |
) |
|
|
(2,626 |
) |
|
|
(6,441 |
) |
|
|
(5,240 |
) |
| Professional and consulting fees |
|
|
(1,698 |
) |
|
|
(1,789 |
) |
|
|
(2,658 |
) |
|
|
(3,079 |
) |
| Other external general and administrative expenses |
|
|
(2,538 |
) |
|
|
(1,816 |
) |
|
|
(5,045 |
) |
|
|
(4,759 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Total |
|
|
(9,358 |
) |
|
|
(8,683 |
) |
|
|
(18,944 |
) |
|
|
(17,961 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
General and administrative expenses increased from €8.7 million for the three months ended June 30, 2022 to €9.4 million for the three months ended June 30, 2023.
Share-based compensation expenses decreased from €2.5 million for the three months ended June 30, 2022 to €2.2 million for the three months ended June 30, 2023.
Personnel related general and administrative expenses, excluding share-based compensation, increased from €2.6 million for the three months ended June 30, 2022 to €2.9 million for the three months ended June 30, 2023. The increase mainly resulted from an increased headcount in our finance, IT, human resources and communications functions.
Professional and consulting fees decreased from €1.8 million for the three months ended June 30, 2022 to €1.7 million for the three months ended June 30, 2023. The decrease in professional and consulting fees resulted mainly from lower legal expenses and consulting expenses.
Other external expenses increased from €1.8 million for the three months ended June 30, 2022 to €2.5 million for the three months ended June 30, 2023. The increase in other expenses mainly resulted from increased other operating expenses.
General and administrative expenses increased from €18.0 million for the six months ended June 30, 2022 to €18.9 million for the six months ended June 30, 2023.
Share-based compensation expenses decreased from €4.9 million for the six months ended June 30, 2022 to €4.8 million for the six months ended June 30, 2023.
Personnel related general and administrative expenses, excluding share-based compensation, increased from €5.2 million for the six months ended June 30, 2022 to €6.4 million for the six months ended June 30, 2023. The increase mainly resulted from an increased headcount in our finance, IT, human resources and communications functions.
Professional and consulting fees decreased from €3.1 million for the six months ended June 30, 2022 to €2.7 million for the six months ended June 30, 2023. The decrease in professional and consulting fees resulted mainly from lower legal expenses and consulting expenses.
Other external expenses increased from €4.8 million for the six months ended June 30, 2022 to €5.0 million for the six months ended June 30, 2023. The increase in other expenses mainly resulted from increased other operating expenses.
Other Financial Income and Other Financial Expenses
Other financial income decreased from €7.0 million for the three months ended June 30, 2022 to €4.0 million for the three months ended June 30, 2023. The decrease mainly resulted from lower foreign exchange gains.
Other financial expenses increased from €0.4 million for the three months ended June 30, 2022 to €1.1 million for the three months ended June 30, 2023. The increase mainly resulted from higher foreign exchange losses.
Other financial income decreased from €8.8 million for the six months ended June 30, 2022 to €6.7 million for the six months ended June 30, 2023. The decrease mainly resulted from lower foreign exchange gains.
Other financial expenses increased from €1.5 million for the six months ended June 30, 2022 to €4.7 million for the six months ended June 30, 2023. The increase mainly resulted from higher foreign exchange losses.
Change in fair value of warrant liabilities
Subsequent to the Business Combination, there were 7,187,500 warrants outstanding, which were classified as financial liabilities through profit and loss. The warrants entitle the holder to purchase one ordinary share at an exercise price of $11.50 per share. The warrants will expire five years after the completion of the Business Combination or earlier upon redemption or liquidation in accordance with their terms.
The fair value of the warrants decreased from €2.35 ($2.51) per warrant as of December 31, 2022 to €1.32 ($1.44) as of March 31, 2023 and increased to €3.15 ($3.42) as of June 30, 2023. The result is an increase in fair value of warrant liabilities of €13.1 million for the three months ended June 30, 2023 and an increase in fair value of warrant liabilities of €5.7 million for the six months ended June 30, 2023.
The fair value of the warrants decreased from €3.88 ($4.39) per warrant as of December 31, 2021 to €1.58 ($1.75) as of March 31, 2022 and increased to €1.96 ($2.04) as of June 30, 2022. The result is an increase in fair value of warrant liabilities of €2.8 million for the three months ended June 30, 2022 and a decrease in fair value of warrant liabilities of €13.7 million for the six months ended June 30, 2022.
Liquidity and Capital Resources
Sources of Liquidity
We have incurred losses since inception, with the exception of the year ended December 31, 2022. We have negative cash flows from operations for the six months ended June 30, 2023 and positive cash flows from operations for the six months ended June 30, 2022. As of June 30, 2023, we had an accumulated deficit of €544.7 million.
We have funded our operations primarily from public offerings and private placements of our ordinary shares, upfront payments from collaborations agreements, and the net proceeds generated from the ARYA Merger and PIPE Financing that closed on July 1, 2020 and our public offering in October 2022.
Cash and cash equivalents decreased from €148.5 million as of December 31, 2022 to €130.4 million as of June 30, 2023.
We received a €13.7 million Opt-in payment from our collaboration partner BMS during the three months ended June 30, 2023. We received €212.4 million in connection with the strategic collaboration agreements with BMS and €106.2 million from a public offering of 10,905,000 ordinary shares during the year ended December 31, 2022.
We believe our existing cash, cash equivalents and other financial assets will be sufficient to fund our operating expenses and capital expenditure requirements through at least the next 12 months. We may consider raising additional capital to pursue strategic investments, to take advantage of financing opportunities or for other reasons. Additionally, in 2021, we established an at-the-market (“ATM”) offering program pursuant to which we may, from time to time, issue and sell shares that have an aggregate offering price of $100 million. For the six months ended June 30, 2023, 3.7 million shares had been sold under the ATM agreement with SVB Securities LLC and collected a gross amount of €38.6 million ($41.8 million).
The Company closed a private placement transaction of 2,419,818 ordinary shares with a subscription price of $14.46 per ordinary share with BMS and received gross proceeds of approximately $35 million after the reporting period.
We plan to utilize the existing Cash, cash equivalents and Other financial assets on hand primarily to fund our operating activities associated with our research and development initiatives to continue or commence clinical trials and seek regulatory approval for our product candidates. We also expect to make capital expenditures in the near term related to the expansion of our laboratory spaces in Tübingen, Germany and our new GMP manufacturing facility in Houston metropolitan area, Texas and expect to continue investing in laboratory and manufacturing equipment and operations to support our anticipated growth. Cash in excess of immediate requirements is invested in accordance with our investment policy with an emphasis on liquidity and capital preservation and consist primarily of cash in banks, short-term deposits and AAA rated bonds.
Cash Flows
The following table summarizes our cash flows for each period presented:
|
|
|
|
|
|
|
|
|
| |
|
Six Months Ended June 30, |
|
| |
|
2023 |
|
|
2022 |
|
| (Euros in thousands) |
|
|
|
|
|
|
| Net cash provided by / (used in): |
|
|
|
|
|
|
|
|
| Operating activities* |
|
|
(30,323 |
) |
|
|
155,773 |
|
| Investing activities |
|
|
(20,555 |
) |
|
|
(48,528 |
) |
| Financing activities |
|
|
35,585 |
|
|
|
15,203 |
|
|
|
|
|
|
|
|
|
|
| Total* |
|
|
(15,293 |
) |
|
|
122,448 |
|
|
|
|
|
|
|
|
|
|
| * |
See Note 3 of the Notes to the Unaudited Condensed Consolidated Financial Statements of Immatics N.V. for details regarding the revision of prior period numbers as a result of a correction in presentation of net foreign exchange differences and effects of exchange rate changes on cash and cash equivalents |
Operating Activities
We primarily derive cash from our collaboration agreements. Our cash used in operating activities is significantly influenced by our use of cash for operating expenses and working capital to support the business. Historically we experienced negative cash flows from operating activities as we have invested in the development of our technologies in our clinical and preclinical development of our product candidates. During the six months ended June 30, 2022, our cash flow from operating activities was positive, as we received an upfront payment from our collaboration partner BMS under the BMS IMA401 collaboration agreement.
Our net cash outflow from operating activities for the six months ended June, 2023 was €30.3 million. This was comprised of a loss of €44.4 million, an increase in working capital of €8.3 million and other effects of €2.7 million related to accrued interest income, partly offset by non-cash expense of €5.7 million related to the change in fair value of the warrants, non-cash charges from equity settled share-based compensation expenses for employees of €11.6 million, depreciation and amortization charge of €3.7 million and net foreign exchange differences and expected credit losses of €4.1 million. The increase in working capital mainly resulted from a decrease in deferred revenue, accounts payable and other liabilities of €9.9 million, partly offset by a decrease in accounts receivable of €0.8 million and a decrease in other assets and prepayments of €0.8 million.
Our net cash inflow from operating activities for the six months ended June, 2022 was €155.8 million. This was comprised of a net profit of €71.7 million, a decrease in working capital of €90.9 million, non-cash charges from equity settled share-based compensation expenses for employees of €11.3 million and depreciation and amortization charge of €3.4 million, partly offset by a non-cash income of €13.7 million related to the change in fair value of the warrants and net foreign exchange differences and expected credit losses of €7.8 million. The decrease in working capital mainly resulted from an increase in deferred revenue, accounts payable and other liabilities of €98.1 million, partly offset by an increase in accounts receivable of €0.3 million and a decrease in other assets and prepayments of €6.9 million.
Investing Activities
Our net outflow of cash from investing activities for the six months ended June 30, 2023 was €20.6 million. This consisted primarily of cash paid in the amount of €170.3 million for short-term deposit investments that are classified as Other financial assets and held with financial institutions to finance the company, €15.2 million as payment for new equipment and intangible assets, partially offset by cash received from maturity of bonds and short-term deposits of €164.9 million.
Our net outflow of cash from investing activities for the six months ended June 30, 2022 was €48.5 million. This consisted primarily of cash paid in the amount of €59.2 million for bond investments that are classified as Other financial assets and held with financial institutions to finance the company, €2.0 million as payment for new equipment and intangible assets, partially offset by cash received from maturity of bonds of €12.7 million.
Financing Activities
During the six months ended June 30, 2023, net cash provided from financing activities amounted to €35.6 million. As of June 30, 2023, 3.7 million shares had been sold under the ATM agreement with SVB Securities LLC and resulted in net proceeds of €37.4 million. This was partially offset by the principal portion of payments in connection with lease contracts.
During the six months ended June 30, 2022, net cash provided from financing activities amounted to €15.2 million. As of June 30, 2022, 2.4 million shares had been sold under the ATM agreement with SVB Securities LLC and resulted in net proceeds of €16.6 million. This was partially offset by the principal portion of payments in connection with lease contracts.
Operation and Funding Requirements
Historically, we have incurred significant losses due to our substantial research and development expenses. We have an accumulated deficit of €544.7 million as of June 30, 2023. We expect our expenses to increase in connection with our ongoing activities, particularly as we continue the research and development of, continue or commence clinical trials including GMP manufacturing of, and seek regulatory approval for, our product candidates. We believe that we have sufficient financial resources available to fund our projected operating requirements for at least the next twelve months. Because the outcome of our current and planned clinical trials is highly uncertain, we cannot reasonably estimate the actual amounts necessary to successfully complete the development and commercialization of our product candidates. For example, our costs will increase if we experience any delays in our current and planned clinical trials. Our future funding requirements will depend on many factors, including, but not limited to:
| |
1. |
progress, timing, scope and costs of our clinical trials, including the ability to timely initiate clinical sites, enroll patients and manufacture ACT and TCR Bispecific product candidates for our ongoing, planned and potential future clinical trials; |
| |
2. |
time and cost to conduct IND- or CTA-enabling studies for our preclinical programs; |
| |
3. |
time and costs required to perform research and development to identify and characterize new product candidates from our research programs; |
| |
4. |
time and cost necessary to obtain regulatory authorizations and approvals that may be required by regulatory authorities to execute clinical trials or commercialize our products; |
| |
5. |
our ability to successfully commercialize our product candidates, if approved; |
| |
6. |
our ability to have clinical and commercial products successfully manufactured consistent with FDA, the EMA and comparable regulatory authorities’ regulations; |
| |
7. |
amount of sales and other revenues from product candidates that we may commercialize, if any, including the selling prices for such potential products and the availability of adequate third-party coverage and reimbursement for patients; |
| |
8. |
sales and marketing costs associated with commercializing our products, if approved, including the cost and timing of building our marketing and sales capabilities; |
| |
9. |
cost of building, staffing and validating our manufacturing processes, which may include capital expenditure; |
| |
10. |
terms and timing of our current and any potential future collaborations, licensing or other arrangements that we have established or may establish; |
| |
11. |
cash requirements of any future acquisitions or the development of other product candidates; |
| |
12. |
costs of operating as a public company; |
| |
13. |
time and cost necessary to respond to technological, regulatory, political and market developments; |
| |
14. |
costs of filing, prosecuting, defending and enforcing any patent claims and other IP rights; and |
| |
15. |
costs associated with any potential business or product acquisitions, strategic collaborations, licensing agreements or other arrangements that we may establish. |
Identifying potential product candidates and conducting preclinical studies and clinical trials is a time-consuming, expensive and uncertain process that takes many years to complete, and we may never generate the necessary data or results required to obtain regulatory approval and commercialize our product candidates. In addition, our product candidates, if approved, may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of products that we do not expect to be commercially available for many years, if at all. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all.
Unless and until we can generate sufficient revenue to finance our cash requirements, which may never happen, we may seek additional capital through a variety of means, including through public and private equity offerings and debt financings, credit and loan facilities and additional collaborations. If we raise additional capital through the sale of equity or convertible debt securities, our existing shareholders’ ownership interest will be diluted, and the terms of such equity or convertible debt securities may include liquidation or other preferences that are senior to or otherwise adversely affect the rights of our existing shareholders. If we raise additional capital through the sale of debt securities or through entering into credit or loan facilities, we may be restricted in our ability to take certain actions, such as incurring additional debt, making capital expenditures, acquiring or licensing IP rights, declaring dividends or encumbering our assets to secure future indebtedness. Such restrictions could adversely impact our ability to conduct our operations and execute our business plan. If we raise additional capital through collaborations with third parties, we may be required to relinquish valuable rights to our IP or product candidates or we may be required to grant licenses for our IP or product candidates on unfavorable terms. If we are unable to raise additional capital when needed, we may be required to delay, limit, reduce or terminate our product development efforts or we may be required to grant rights to third parties to develop and market our product candidates that we would otherwise prefer to develop and market ourselves. For more information as to the risks associated with our future funding needs, see “Risk Factors—Risks Related to Our Financial Position” in our Annual Report.
Critical Accounting Estimates
Our unaudited interim condensed consolidated financial statements for the three and six month-period ended June 30, 2023 and 2022, respectively, have been prepared in accordance with International Accounting Standard 34 (Interim Financial Reporting), as issued by the International Accounting Standards Board.
The preparation of the consolidated financial statements in accordance with IFRS requires the use of estimates and assumptions, which affect the value of assets and liabilities, as well as contingent assets and liabilities, as reported on the balance sheet date, and revenues and expenses arising during the fiscal year.
The preparation of the consolidated financial statements for the fiscal year ended December 31, 2022 and the three and six months ended June 30, 2023 in accordance with IFRS required the use of estimates and assumptions by the management that affect the value of assets and liabilities—as well as contingent assets and liabilities—as reported on the balance sheet date, and revenues and expenses arising during the year. The main areas in which assumptions, estimates and the exercising of a degree of discretion are appropriate relate to the determination of revenue recognition, research and development expenses, and share-based compensations as well as income taxes.
Our estimates are based on historical experience and other assumptions that are considered appropriate in the circumstances, and parameters available when the consolidated financial statements were prepared. Existing circumstances and assumptions about future developments, however, may change due to market changes or circumstances arising that are beyond our control. Hence, our estimates may vary from the actual values.
While our significant accounting policies are more fully discussed in our consolidated financial statements included in our Annual Report, we believe that the following accounting policies are critical to the process of making significant judgments and estimates in the preparation of our interim condensed consolidated financial statements.
Revenue Recognition for Collaboration Agreements
We recognize revenue through collaboration and license agreements and reimbursement for research and development costs.
Under our collaboration and license agreements, we may receive upfront licensing payments, milestone payments and reimbursement of research and development expenses. Such collaboration agreements also include licenses of certain of our IP to the respective collaborators. As these agreements are comprised of several commitments, it must be assessed whether these commitments are capable of being distinct within the context of the contract. For three of our four collaboration agreements, we determined that the commitments included in each agreement represented single combined performance obligations, with a single measure of progress. The performance obligation is accounted for as a performance obligation satisfied over time on a cost-to-cost basis, as our collaboration partner simultaneously receives and consumes the benefit from our performance. Upfront licensing payments and reimbursement for development expenses are initially deferred on our statement of financial position and subsequently recognized as revenue over time as costs are incurred.
For our collaboration with BMS regarding IMA-401 that was signed in December 2021, we concluded that the commitments from the collaboration agreement represented two distinct performance obligations. The granted license is transferred at a point in time at the effective date of the agreement and we recognized the revenue allocated to the license at the effective date. The performance obligation related to promised clinical trial services is satisfied over time. We transfer control of these agreed services over time and therefore recognize revenue over time on a cost-to-cost basis. The transaction price allocated to the commitment for clinical trial services is initially deferred on our statement of financial position and subsequently recognized as revenue as costs are incurred.
Milestone payments are generally included in the transaction price at the amount stipulated in the respective agreement and recognized to the extent that it is highly probable that a significant reversal in the amount of cumulative revenue recognized will not occur. To date, no milestone payment has been included in the transaction price and recognized into revenue.
We provide development and manufacturing work to our collaboration partners and recognize revenue over time using an input-based method to measure progress toward complete satisfaction of the service, because the collaboration partner simultaneously receives and consumes the benefits provided. Forecast values are used for the calculation of expected future revenue for the remaining term of the contract. These costs estimated as part of the budgeting process must be reviewed and approved before we can use them for recognition purposes. Significant management judgment is required to determine the level of effort required under an arrangement, and the period over which we expect to complete our performance obligations under the arrangement which includes total internal personnel costs and external costs to be incurred. Changes in these estimates can have a material effect on revenue recognized.
Share-based Compensation
The Company offers a share-based compensation plan that includes PSUs and service options including a conversion of previous share-based compensation arrangements entered into by Immatics GmbH.
The costs of equity-settled transactions are determined by the fair value at grant date, using an appropriate valuation model. Share-based expenses for the respective vesting periods, are recognized in research and development expenses and general and administrative expenses, reflecting a corresponding increase in equity.
Income Taxes
Uncertainties exist with respect to the interpretation of complex tax regulations, changes in tax laws, and the amount and timing of future taxable income. Given the wide range and complexity of existing contractual agreements, differences arising between the actual results and the assumptions made, or future changes to such assumptions, could necessitate future adjustments to tax income and expenses already recorded. Deferred tax assets are recognized for unused tax losses to the extent that it is probable that taxable profit will be available which can be utilized against the losses. Significant management judgement is required to determine the amount of deferred tax assets that can be recognized, based upon the likely timing and the level of future taxable profits together with future tax planning strategies. Due to our history of loss-making over the last several years as well as our expectation for the foreseeable future, we have not recognized any deferred tax assets on tax losses carried forward despite the net income for the year ended December 31, 2022. Changes in the estimation of our potential to use of tax losses carried forward can have a material effect on our net income.
Recently Issued and Adopted Accounting Pronouncement
New standards and interpretations applied for the first time as of January 1, 2023 and 2022 had no material effect on the consolidated financial statements of the Group.
Quantitative and Qualitative Disclosures about Market Risk
We are exposed to various risks in relation to financial instruments. Our principal financial instruments comprise cash and cash equivalents, short-term deposits, accounts receivables and bonds. The main purpose of these financial instruments is to invest the proceeds of capital contributions and upfront payments from collaboration agreements. We have various other financial instruments such as other receivables and trade accounts payables, which arise directly from its operations.
The main risks arising from our financial instruments are market risk and liquidity risk. The Board of Management reviews and agrees on policies for managing these risks as summarized below. We also monitor the market price risk arising from all financial instruments.
Interest rate risk
Our exposure to changes in interest rates relates to investments in deposits, bonds and to changes in the interest for overnight deposits. Changes in the general level of interest rates may lead to an increase or decrease in the fair value of these investments. Regarding the liabilities shown in the Consolidated Statement of Financial Position, we are currently not subject to interest rate risks.
Credit risk
Financial instruments that potentially subject us to concentrations of credit and liquidity risk consist primarily of cash and cash equivalents, accounts receivables, short-term deposits and bonds. Our cash and cash equivalents, bonds and short-term deposits are denominated in Euros and US Dollars and maintained with three financial institutions in Germany and two in the United States. Our accounts receivables are denominated in Euros.
We continually monitor our positions with, and the credit quality of, the financial institutions and corporation, which are counterparts to our financial instruments and we are not anticipating non-performance. The maximum default risk corresponds to the carrying amount of the financial assets shown in the statement of financial position. We monitor the risk of a liquidity shortage. The main factors considered here are the maturities of financial assets, as well as expected cash flows from equity measures.
Currency risk
Currency risk shows the risk that the value of a financial instrument will fluctuate due to changes in foreign exchange rates. In particular it poses a threat if the value of the currency in which liabilities are priced appreciates relative to the currency of the assets. Our business transactions are generally conducted in Euros and U.S. dollars. We aim to match EUR cash inflows with EUR cash outflows and U.S. dollar cash inflows with U.S. Dollar cash outflows where possible. Our objective of currency risk management is to identify, manage and control currency risk exposures within acceptable parameters.
Our cash and cash equivalents were €130.4 million as of June 30, 2023. Approximately 73% of our cash and cash equivalents were held in Germany, of which approximately 52% were denominated in Euros and 48% were denominated in U.S. Dollars. The remainder of our cash and cash equivalents are held in the United States and denominated in U.S. Dollars. Additionally, we have short-term deposits classified as Other financial assets denominated in Euros in the amount of €111.7 million and U.S. Dollars in the amount of €105.5 million as of June 30, 2023.
Market risk and currency risk of warrants
The Group’s activities expose it to the financial risks of changes in price of the warrants. As the warrants are recognized at fair value on the consolidated statement of financial position of the Group, the Group’s exposure to market risks results from the volatility of the warrants price. The Warrants are publicly traded at the NASDAQ Stock Exchange. A reasonable increase (decrease) in the warrant price by 10%, with all other variables held constant, would lead to a (loss) gain before tax of €2.3 million with a corresponding effect in the equity as of June 30, 2023.
OTHER INFORMATION
Legal Proceedings
From time to time, we may be subject to various legal proceedings and claims that arise in the ordinary course of our business activities. For example, in September 2020, we filed an opposition and in October 2020 we commenced a cancellation proceeding against Immunocore Limited which challenges its IMMTAX trademark in various jurisdictions. In November 2020, Immunocore Limited filed counterclaims against our registered trademark IMMATICS and IMTX. This matter has now been amicably resolved.
Risk Factors
There have been no material changes from the risk factors described in the section titled “Risk Factors” in our Annual Report.
Exhibit 99.2

PRESS RELEASE
Immatics Announces Second Quarter 2023
Financial Results and Business Update
| |
• |
|
Phase 1/2 clinical trial initiated evaluating Immatics’ second next-generation half-life extended TCR
Bispecific program, TCER® IMA402 targeting PRAME |
| |
• |
|
ACTengine® IMA203
TCR-T monotherapy against PRAME showed 67% confirmed ORR in an interim clinical update on 11 heavily pre-treated patients in Phase 1b dose expansion Cohort A with median
duration of response not reached at a median follow-up time of 8.5 months at data cut-off; next update in 4Q 2023 |
| |
• |
|
Bristol Myers Squibb exercised first opt-in into 2019 cell therapy
collaboration ($15 million option fee received) and made a $35 million equity investment |
| |
• |
|
Cash and cash equivalents as well as other financial assets not including the recent equity investment by Bristol
Myers Squibb amount to $377.7 million1 (€347.6) as of June 30, 2023; updated projected cash runway is late 2025 |
Tuebingen, Germany and Houston, TX, August 17, 2023 – Immatics N.V. (NASDAQ: IMTX; “Immatics”), a
clinical-stage biopharmaceutical company active in the discovery and development of T cell-redirecting cancer immunotherapies, today provided a business update and reported financial results for the quarter ended June 30, 2023.
“The interim clinical data update for IMA203 monotherapy demonstrated an encouraging initial objective response rate in a range of solid cancer
indications including durable responses supporting fast-tracking IMA203 to patients, starting with high-need solid cancers such as checkpoint-refractory melanoma and uveal melanoma,” said Harpreet Singh, Ph.D., CEO and Co-Founder of Immatics. “Beyond our recent IMA203 updates, we are pleased to report that we closed the second quarter with a cash position funding operations into late 2025. With this revised runway, we
anticipate reaching our most critical milestones including the initiation of registration-directed trials for IMA203, as well as delivering meaningful data to assess clinical proof of concept for both TCER® programs IMA401 and IMA402.”
| 1 |
All amounts translated using the exchange rate published by the European Central Bank in effect as of
June 30, 2023 (1 EUR = 1.0866 USD). |
|
|
|
| Immatics Press Release August 17, 2023 |
|
1 | 11 |

Second Quarter 2023 and Subsequent Company Progress
Adoptive Cell Therapy Programs
ACTengine® IMA203: ACTengine® IMA203 TCR-T against PRAME is currently being evaluated in an ongoing Phase 1b
dose expansion trial.
| |
• |
|
As per the latest data cut-off of April 4, 2023, ACTengine® IMA203 TCR-T monotherapy Cohort A showed a 67% confirmed objective response rate (cORR) in an interim clinical update announced on May 2, 2023. The
data covered 11 heavily pre-treated patients; the median duration of response was not reached at a median follow-up time of 8.5 months. Patients were infused with IMA203
TCR-T cells at dose level (DL) 4 or DL5 with a mean total infused dose of 3.67x109 TCR-T cells (range 1.30-8.84x109 TCR-T cells). |
| |
• |
|
Cohort A IMA203 monotherapy TCR-T treatment continues to show manageable
tolerability with no high-grade CRS and no ICANS; all 11 patients experienced expected cytopenia (Grade 1-4) associated with lymphodepletion. 10 patients (91%) had a low to moderate (Grade 1-2) cytokine release syndrome (CRS), of which 5 patients (45%) had Grade 1, and 5 patients (45%) had Grade 2 CRS. |
| |
• |
|
Objective responses were observed independent of tumor type including checkpoint-refractory and BRAF
inhibitor-refractory cutaneous melanoma, platinum-resistant ovarian cancer, uveal melanoma, head and neck cancer and synovial sarcoma. Longest duration of responses were observed in cutaneous and uveal melanoma with ongoing responses at 6, 9 and 10
months post infusion at data cut-off. |
| |
• |
|
IMA203 in combination with nivolumab (Cohort B) has been de-prioritized
in the last-line setting. Such a combination is being considered for the front-line setting. |
| |
• |
|
IMA203CD8 (Cohort C) is a next-generation monotherapy where IMA203 engineered T cells are co-transduced with a CD8αß co-receptor. IMA203CD8 is currently being explored in DL4a (up to 0.8x109 TCR-T cells/m2 BSA). |
| |
• |
|
Next update on Immatics’ IMA203 Phase 1b cohorts, including the projected clinical development path for
PRAME-targeted TCR-T monotherapy towards registration-directed trials is planned for 4Q 2023. Immatics’ IMA203 development strategy to realize the multi-cancer opportunity of targeting PRAME is based on
two pillars aimed at: |
| |
1. |
maximizing speed to market in one to two last-line solid cancer types focusing on cutaneous melanoma, uveal
melanoma and potentially other tumor types with high PRAME prevalence where clinical proof-of-concept has been demonstrated, and |
| |
2. |
broad development with expansion to other cancer types, such as ovarian cancer, uterine cancer, lung cancer,
breast cancer, head and neck cancer and other tumor types having a broad patient reach. |
|
|
|
| Immatics Press Release August 17, 2023 |
|
2 | 11 |

TCR-T pipeline
| |
• |
|
Earlier this year, Bristol Myers Squibb exercised its first option and entered into a global license agreement
with Immatics for the most advanced TCR-T product candidate. As part of the agreement, Immatics received an option payment of $15 million and is eligible for up to $490 million in milestone payments
in addition to tiered royalties on net sales of the product. |
TCR Bispecifics Programs
Immatics’ T cell engaging receptor (TCER®) candidates are next-generation, half-life extended TCR
Bispecific molecules designed to maximize efficacy while minimizing toxicities in patients through Immatics’ proprietary format using a low-affinity T cell recruiter and a high-affinity TCR domain.
| |
• |
|
TCER® IMA401 (MAGEA4/8) –
Phase 1 trial to evaluate safety, tolerability and initial anti-tumor activity of TCER® IMA401 in patients with recurrent and/or refractory solid tumors is ongoing. IMA401 targets an HLA-A*02:01-presented peptide that occurs identically in two different proteins, MAGEA4 and MAGEA8. This target peptide has been selected based on natural expression in native
solid tumors at particularly high target density (peptide copy number per tumor cell identified by Immatics’ proprietary quantitative mass spectrometry engine XPRESIDENT®). MAGEA4 and
MAGEA8 are expressed in multiple solid cancers including lung cancer, head and neck cancer, melanoma, ovarian cancer, sarcoma and others. IMA401 is being developed in collaboration with Bristol Myers Squibb. |
| |
• |
|
TCER® IMA402 (PRAME) –
Immatics submitted a clinical trial application (CTA2) to the Paul-Ehrlich-Institute (PEI) in April 2023. Following CTA acceptance, Immatics initiated the Phase 1/2 trial investigating the
company’s fully owned TCER® candidate IMA402 in patients with recurrent and/or refractory solid tumors in August. Initial focus indications are cutaneous and uveal melanoma,
ovarian cancer, lung cancer, uterine cancer and synovial sarcoma, among others. A first clinical data update is planned for 2024. IMA402 targets an HLA-A*02:01-presented
peptide derived from the tumor antigen PRAME. This target peptide has been selected based on natural expression in native solid primary tumors and metastases at particularly high target density (peptide copy number per tumor cell identified by
Immatics’ proprietary quantitative mass spectrometry engine XPRESIDENT®). |
| 2 |
Clinical Trial Application (CTA) is the European equivalent of an Investigational New Drug (IND) application.
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
3 | 11 |

Corporate Updates
| |
• |
|
On July 24, 2023, Bristol Myers Squibb purchased 2,419,818 ordinary shares in a private placement
transaction at a subscription price per share of $14.463. Additionally, Bristol Myers Squibb will appoint a member to the Immatics Scientific Advisory Board. |
Second Quarter 2023 Financial Results
Equity: The
Company raised a total of $64 million in June through August through its ATM facility.
Cash Position: Cash and cash equivalents as well as
other financial assets total €347.6 million ($377.7 million1) as of June 30, 2023, compared to €362.2 million ($393.6
million1) as of December 31, 2022. The decrease is mainly due to our ongoing research and development activities, partially offset by the option fee received by Bristol Myers Squibb and funds
raised in the period. The Company projects an updated cash runway into late 2025.
Revenue: Total revenue, consisting of revenue from collaboration
agreements, was €22.4 million ($24.3 million1) for the three months ended June 30, 2023, compared to €17.2 million ($18.7 million1) for the three months ended June 30, 2022. The increase is mainly related to the recognition of revenue for the opt-in agreement with Bristol Myers Squibb
signed during the three months ended June 30, 2023.
Research and Development Expenses: R&D expenses were €27.3 million ($29.7
million1) for the three months ended June 30, 2023, compared to €25.2 million ($27.4 million1) for the three months ended
June 30, 2022. The increase mainly resulted from higher costs associated with the advancement of the clinical and pre-IND pipeline of ACTengine®
and TCER® candidates.
General and Administrative Expenses: G&A expenses were
€9.4 million ($10.2 million1) for the three months ended June 30, 2023, compared to €8.7 million ($9.5 million1) for
the three months ended June 30, 2022.
Net Profit and Loss: Net loss was €24.6 million ($26.7 million1) for the three months ended June 30, 2023, compared to a net loss of €14.0 million ($15.2 million1) for the three months ended
June 30, 2022. The increased net loss mainly resulted from non-cash fair value adjustments of outstanding warrants.
| 3 |
Exact price per share $14.4639 |
|
|
|
| Immatics Press Release August 17, 2023 |
|
4 | 11 |

Upcoming Investor Conferences
| |
• |
|
Jefferies Cell & Genetic Medicine Summit, New York, NY – September
26-27, 2023 |
| |
• |
|
Jefferies London Healthcare Conference, London, U.K. – November
14-16, 2023 |
To see the full list of events and presentations, visit
www.investors.immatics.com/events-presentations.
About Immatics
Immatics combines the discovery of true targets for cancer immunotherapies with the development of the right T cell receptors with the goal of enabling a
robust and specific T cell response against these targets. This deep know-how is the foundation for our pipeline of Adoptive Cell Therapies and TCR Bispecifics as well as our partnerships with global leaders
in the pharmaceutical industry. We are committed to delivering the power of T cells and to unlocking new avenues for patients in their fight against cancer.
Immatics intends to use its website www.immatics.com as a means of disclosing material non-public information.
For regular updates you can also follow us on Twitter, Instagram and LinkedIn.
Forward-Looking Statements:
Certain statements in this press release may be considered forward-looking statements. Forward-looking statements generally relate to future events or
Immatics’ future financial or operating performance. For example, statements concerning the timing of product candidates and Immatics’ focus on partnerships to advance its strategy are forward-looking statements. In some cases, you can
identify forward-looking statements by terminology such as “may”, “should”, “expect”, “intend”, “will”, “estimate”, “anticipate”, “believe”, “predict”,
“potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward-looking statements are subject to risks, uncertainties, and other factors which could cause actual results to
differ materially from those expressed or implied by such forward-looking statements. These forward-looking statements are based upon estimates and assumptions that, while considered reasonable by Immatics and its management, are inherently
uncertain. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Factors that may cause actual results to differ materially from current expectations include, but are not limited to,
various factors beyond management’s control including general economic conditions and other risks, uncertainties and factors set forth in filings with the SEC. Nothing in this press release should be regarded as a representation by any person
that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as
of the date they are made. Immatics undertakes no duty to update these forward-looking statements.
|
|
|
| Immatics Press Release August 17, 2023 |
|
5 | 11 |

For more information, please contact:
|
| Media and Investor Relations Contact |
| Eva Mulder or Charlotte Spitz |
| Trophic Communications |
| Phone: +31 6 52 33 15 79 |
immatics@trophic.eu
|
|
|
|
|
| Immatics N.V. |
|
|
| Anja Heuer |
|
Sabrina Schecher, Ph.D. |
| Senior Director, Corporate Communications |
|
Senior Director, Investor Relations |
| Phone: +49 89 540415-606 |
|
Phone: +49 89 262002433 |
| media@immatics.com |
|
InvestorRelations@immatics.com |
|
|
|
| Immatics Press Release August 17, 2023 |
|
6 | 11 |

Unaudited Condensed Consolidated Statement of Profit/(Loss) of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| |
|
(Euros in thousands, except
per share data) |
|
|
(Euros in thousands, except
per share data) |
|
| Revenue from collaboration agreements |
|
|
22,354 |
|
|
|
17,215 |
|
|
|
32,150 |
|
|
|
120,123 |
|
| Research and development expenses |
|
|
(27,317 |
) |
|
|
(25,216 |
) |
|
|
(54,898 |
) |
|
|
(50,360 |
) |
| General and administrative expenses |
|
|
(9,358 |
) |
|
|
(8,683 |
) |
|
|
(18,944 |
) |
|
|
(17,961 |
) |
| Other income |
|
|
6 |
|
|
|
27 |
|
|
|
948 |
|
|
|
32 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Operating result |
|
|
(14,315 |
) |
|
|
(16,657 |
) |
|
|
(40,744 |
) |
|
|
51,834 |
|
| Change in fair value of liabilities for warrants |
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
| Other financial income |
|
|
3,954 |
|
|
|
7,015 |
|
|
|
6,748 |
|
|
|
8,774 |
|
| Other financial expenses |
|
|
(1,144 |
) |
|
|
(407 |
) |
|
|
(4,653 |
) |
|
|
(1,524 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Financial result |
|
|
(10,295 |
) |
|
|
3,822 |
|
|
|
(3,613 |
) |
|
|
20,993 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Profit/(loss) before taxes |
|
|
(24,610 |
) |
|
|
(12,835 |
) |
|
|
(44,357 |
) |
|
|
72,827 |
|
| Taxes on income |
|
|
— |
|
|
|
(1,145 |
) |
|
|
— |
|
|
|
(1,145 |
) |
| Net profit/(loss) |
|
|
(24,610 |
) |
|
|
(13,980 |
) |
|
|
(44,357 |
) |
|
|
71,682 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Net profit/(loss) per share: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
| Diluted |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
7 | 11 |

Unaudited Condensed Consolidated Statement of Comprehensive Income/(Loss) of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
2023 |
|
|
2022 |
|
|
2023 |
|
|
2022 |
|
| |
|
(Euros in thousands) |
|
|
(Euros in thousands) |
|
| Net profit/(loss) |
|
|
(24,610 |
) |
|
|
(13,980 |
) |
|
|
(44,357 |
) |
|
|
71,682 |
|
| Other comprehensive income/(loss) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Items that may be reclassified subsequently to profit or loss |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Currency translation differences from foreign operations |
|
|
(224 |
) |
|
|
778 |
|
|
|
340 |
|
|
|
1,338 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Total comprehensive income/(loss) for the year |
|
|
(24,834 |
) |
|
|
(13,202 |
) |
|
|
(44,017 |
) |
|
|
73,020 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
8 | 11 |

Unaudited Condensed Consolidated Statement of Financial Position of Immatics N.V.
|
|
|
|
|
|
|
|
|
| |
|
As of |
|
| |
|
June 30, 2023 |
|
|
December 31, 2022 |
|
| |
|
(Euros in thousands) |
|
| Assets |
|
|
|
|
|
|
|
|
| Current assets |
|
|
|
|
|
|
|
|
| Cash and cash equivalents |
|
|
130,405 |
|
|
|
148,519 |
|
| Other financial assets |
|
|
217,222 |
|
|
|
213,686 |
|
| Accounts receivables |
|
|
330 |
|
|
|
1,111 |
|
| Other current assets |
|
|
16,668 |
|
|
|
13,838 |
|
|
|
|
|
|
|
|
|
|
| Total current assets |
|
|
364,625 |
|
|
|
377,154 |
|
| Non-current assets |
|
|
|
|
|
|
|
|
| Property, plant and equipment |
|
|
27,188 |
|
|
|
13,456 |
|
| Intangible assets |
|
|
1,655 |
|
|
|
1,632 |
|
| Right-of-use
assets |
|
|
14,749 |
|
|
|
13,033 |
|
| Other non-current assets |
|
|
1,972 |
|
|
|
2,545 |
|
|
|
|
|
|
|
|
|
|
| Total non-current assets |
|
|
45,564 |
|
|
|
30,666 |
|
|
|
|
|
|
|
|
|
|
| Total assets |
|
|
410,189 |
|
|
|
407,820 |
|
|
|
|
|
|
|
|
|
|
| Liabilities and shareholders’ equity |
|
|
|
|
|
|
|
|
| Current liabilities |
|
|
|
|
|
|
|
|
| Provisions |
|
|
3,117 |
|
|
|
— |
|
| Accounts payables |
|
|
19,904 |
|
|
|
13,056 |
|
| Deferred revenue |
|
|
67,997 |
|
|
|
64,957 |
|
| Liabilities for warrants |
|
|
22,622 |
|
|
|
16,914 |
|
| Lease liabilities |
|
|
2,737 |
|
|
|
2,159 |
|
| Other current liabilities |
|
|
7,929 |
|
|
|
9,366 |
|
|
|
|
|
|
|
|
|
|
| Total current liabilities |
|
|
124,306 |
|
|
|
106,452 |
|
| Non-current liabilities |
|
|
|
|
|
|
|
|
| Deferred revenue |
|
|
53,559 |
|
|
|
75,759 |
|
| Lease liabilities |
|
|
14,085 |
|
|
|
12,403 |
|
| Other non-current liabilities |
|
|
26 |
|
|
|
42 |
|
|
|
|
|
|
|
|
|
|
| Total non-current liabilities |
|
|
67,670 |
|
|
|
88,204 |
|
| Shareholders’ equity |
|
|
|
|
|
|
|
|
| Share capital |
|
|
804 |
|
|
|
767 |
|
| Share premium |
|
|
763,206 |
|
|
|
714,177 |
|
| Accumulated deficit |
|
|
(544,656 |
) |
|
|
(500,299 |
) |
| Other reserves |
|
|
(1,141 |
) |
|
|
(1,481 |
) |
|
|
|
|
|
|
|
|
|
| Total shareholders’ equity |
|
|
218,213 |
|
|
|
213,164 |
|
|
|
|
|
|
|
|
|
|
| Total liabilities and shareholders’ equity |
|
|
410,189 |
|
|
|
407,820 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
9 | 11 |

Unaudited Condensed Consolidated Statement of Cash Flows of Immatics N.V.
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, |
|
| |
|
2023 |
|
|
2022 |
|
| |
|
(Euros in thousands) |
|
| Cash flows from operating activities |
|
|
|
|
|
|
|
|
| Net profit/(loss) |
|
|
(44,357 |
) |
|
|
71,682 |
|
| Taxes on income |
|
|
— |
|
|
|
1,145 |
|
| Profit/(loss) before tax |
|
|
(44,357 |
) |
|
|
72,827 |
|
| Adjustments for: |
|
|
|
|
|
|
|
|
| Interest income |
|
|
(4,999 |
) |
|
|
(23 |
) |
| Depreciation and amortization |
|
|
3,666 |
|
|
|
3,407 |
|
| Interest expenses |
|
|
401 |
|
|
|
538 |
|
| Equity settled share-based payment |
|
|
11,615 |
|
|
|
11,262 |
|
| Net foreign exchange differences and expected credit losses |
|
|
4,081 |
|
|
|
(7,834 |
) |
| Change in fair value of liabilities for warrants |
|
|
5,708 |
|
|
|
(13,743 |
) |
| Changes in: |
|
|
|
|
|
|
|
|
| Decrease/(increase) in accounts receivables |
|
|
781 |
|
|
|
(280 |
) |
| Decrease/(increase) in other assets |
|
|
765 |
|
|
|
(6,903 |
) |
| (Decrease)/increase in deferred revenue, accounts payables and other liabilities |
|
|
(9,889 |
) |
|
|
96,933 |
|
| Interest received |
|
|
2,051 |
|
|
|
23 |
|
| Interest paid |
|
|
(146 |
) |
|
|
(434 |
) |
| Income tax paid |
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
| Net cash (used in)/provided by operating activities |
|
|
(30,323 |
) |
|
|
155,773 |
|
|
|
|
|
|
|
|
|
|
| Cash flows from investing activities |
|
|
|
|
|
|
|
|
| Payments for property, plant and equipment |
|
|
(15,004 |
) |
|
|
(1,965 |
) |
| Payments for intangible assets |
|
|
(154 |
) |
|
|
(6 |
) |
| Proceeds from disposal of property, plant and equipment |
|
|
— |
|
|
|
1 |
|
| Payments for investments classified in Other financial assets |
|
|
(170,326 |
) |
|
|
(59,253 |
) |
| Proceeds from maturity of investments classified in Other financial assets |
|
|
164,929 |
|
|
|
12,695 |
|
|
|
|
|
|
|
|
|
|
| Net cash (used in)/provided by investing activities |
|
|
(20,555 |
) |
|
|
(48,528 |
) |
|
|
|
|
|
|
|
|
|
| Cash flows from financing activities |
|
|
|
|
|
|
|
|
| Proceeds from issuance of shares to equity holders |
|
|
38,608 |
|
|
|
17,112 |
|
| Transaction costs deducted from equity |
|
|
(1,157 |
) |
|
|
(515 |
) |
| Repayment of lease liabilities |
|
|
(1,866 |
) |
|
|
(1,394 |
) |
|
|
|
|
|
|
|
|
|
| Net cash provided by/(used in) financing activities |
|
|
35,585 |
|
|
|
15,203 |
|
|
|
|
|
|
|
|
|
|
| Net (decrease)/increase in cash and cash equivalents |
|
|
(15,293 |
) |
|
|
122,448 |
|
|
|
|
|
|
|
|
|
|
| Cash and cash equivalents at beginning of the year |
|
|
148,519 |
|
|
|
132,994 |
|
|
|
|
|
|
|
|
|
|
| Effects of exchange rate changes and expected credit losses on cash and cash equivalents |
|
|
(2,821 |
) |
|
|
9,683 |
|
|
|
|
|
|
|
|
|
|
| Cash and cash equivalents at end of the year |
|
|
130,405 |
|
|
|
265,125 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
10 | 11 |

Unaudited Condensed Consolidated Statement of Changes in Shareholders’ equity of Immatics N.V.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| (Euros in thousands) |
|
Share
capital |
|
|
Share
premium |
|
|
Accumulated
deficit |
|
|
Other
reserves |
|
|
Total
share-
holders’
equity |
|
| Balance as of January 1, 2022 |
|
|
629 |
|
|
|
565,192 |
|
|
|
(537,813 |
) |
|
|
(3,945 |
) |
|
|
24,063 |
|
| Other comprehensive income |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,338 |
|
|
|
1,338 |
|
| Net profit |
|
|
— |
|
|
|
— |
|
|
|
71,682 |
|
|
|
— |
|
|
|
71,682 |
|
| Comprehensive income for the year |
|
|
— |
|
|
|
— |
|
|
|
71,682 |
|
|
|
1,338 |
|
|
|
73,020 |
|
| Equity-settled share-based compensation |
|
|
— |
|
|
|
11,262 |
|
|
|
— |
|
|
|
— |
|
|
|
11,262 |
|
| Share options exercised |
|
|
— |
|
|
|
1 |
|
|
|
— |
|
|
|
— |
|
|
|
1 |
|
| Issue of share capital - net of transaction costs |
|
|
24 |
|
|
|
16,571 |
|
|
|
— |
|
|
|
— |
|
|
|
16,595 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance as of June 30, 2022 |
|
|
653 |
|
|
|
593,026 |
|
|
|
(466,131 |
) |
|
|
(2,607 |
) |
|
|
124,941 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance as of January 1, 2023 |
|
|
767 |
|
|
|
714,177 |
|
|
|
(500,299 |
) |
|
|
(1,481 |
) |
|
|
213,164 |
|
| Other comprehensive income |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
340 |
|
|
|
340 |
|
| Net loss |
|
|
— |
|
|
|
— |
|
|
|
(44,357 |
) |
|
|
— |
|
|
|
(44,357 |
) |
| Comprehensive loss for the year |
|
|
— |
|
|
|
— |
|
|
|
(44,357 |
) |
|
|
340 |
|
|
|
(44,017 |
) |
| Equity-settled share-based compensation |
|
|
— |
|
|
|
11,615 |
|
|
|
— |
|
|
|
— |
|
|
|
11,615 |
|
| Share options exercised |
|
|
— |
|
|
|
40 |
|
|
|
— |
|
|
|
— |
|
|
|
40 |
|
| Issue of share capital - net of transaction costs |
|
|
37 |
|
|
|
37,374 |
|
|
|
— |
|
|
|
— |
|
|
|
37,411 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Balance as of June 30, 2023 |
|
|
804 |
|
|
|
763,206 |
|
|
|
(544,656 |
) |
|
|
(1,141 |
) |
|
|
218,213 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Immatics Press Release August 17, 2023 |
|
11 | 11 |

Immatics Corporate Presentation August
17, 2023 Exhibit 99.3

Forward-Looking Statement This
presentation (“Presentation”) is provided by Immatics N.V. (“Immatics” or the “Company”) for informational purposes only. The information contained herein does not purport to be all-inclusive and none of Immatics,
any of its affiliates, any of its or their respective control persons, officers, directors, employees or representatives makes any representation or warranty, express or implied, as to the accuracy, completeness or reliability of the information
contained in this Presentation. Forward-Looking Statements. Certain statements in this presentation may be considered forward-looking statements. Forward-looking statements generally relate to future events or the Company’s future financial or
operating performance. For example, statements concerning timing of data read-outs for product candidates, the timing of IND or CTA filing for pre-clinical stage product candidates, the Company’s focus on partnerships to advance its strategy,
and other metrics are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “may”, “should”, “expect”, “intend”, “will”,
“estimate”, “anticipate”, “believe”, “predict”, “potential” or “continue”, or the negatives of these terms or variations of them or similar terminology. Such forward-looking
statements are subject to risks, uncertainties, and other factors which could cause actual results to differ materially from those expressed or implied by such forward looking statements. These forward-looking statements are based upon estimates and
assumptions that, while considered reasonable, Immatics and its management, are inherently uncertain. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Factors that may cause
actual results to differ materially from current expectations include, but are not limited to, various factors beyond management's control including general economic conditions and other risks, uncertainties and factors set forth in the
Company’s Annual report on Form 20-F and other filings with the Securities and Exchange Commission (SEC). Nothing in this presentation should be regarded as a representation by any person that the forward-looking statements set forth herein
will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. You should not place undue reliance on forward-looking statements, which speak only as of the date they are made. The Company undertakes no
duty to update these forward-looking statements. No Offer or Solicitation. This communication is for informational purposes only and does not constitute, or form a part of, an offer to sell or the solicitation of an offer to sell or an offer to buy
or the solicitation of an offer to buy any securities, and there shall be no sale of securities, in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any
such jurisdiction. No offer of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933, as amended, or in an offering exempt from registration. Certain information contained in
this Presentation relates to or is based on studies, publications, surveys and the Company’s own internal estimates and research. In addition, all of the market data included in this presentation involves a number of assumptions and
limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while the Company believes its internal research is reliable, such research has not been verified by any independent source. All the
scientific and clinical data presented within this presentation are – by definition prior to completion of the clinical trial and a clinical study report – preliminary in nature and subject to further quality checks including customary
source data verification.

Therapeutic Opportunity Potential for
addressing large patient populations with high prevalence targets in solid tumors Two Clinical-Stage Modalities Pipeline of TCR-T and TCR Bispecific product candidates in clinical & preclinical development Building a Leading TCR Therapeutics
Company Intro Differentiated Platforms Unique technologies to identify true cancer targets and right TCRs Clinical PoC for Cell Therapy Anti-tumor activity and durability of response across multiple solid tumors in early TCR-T clinical
development
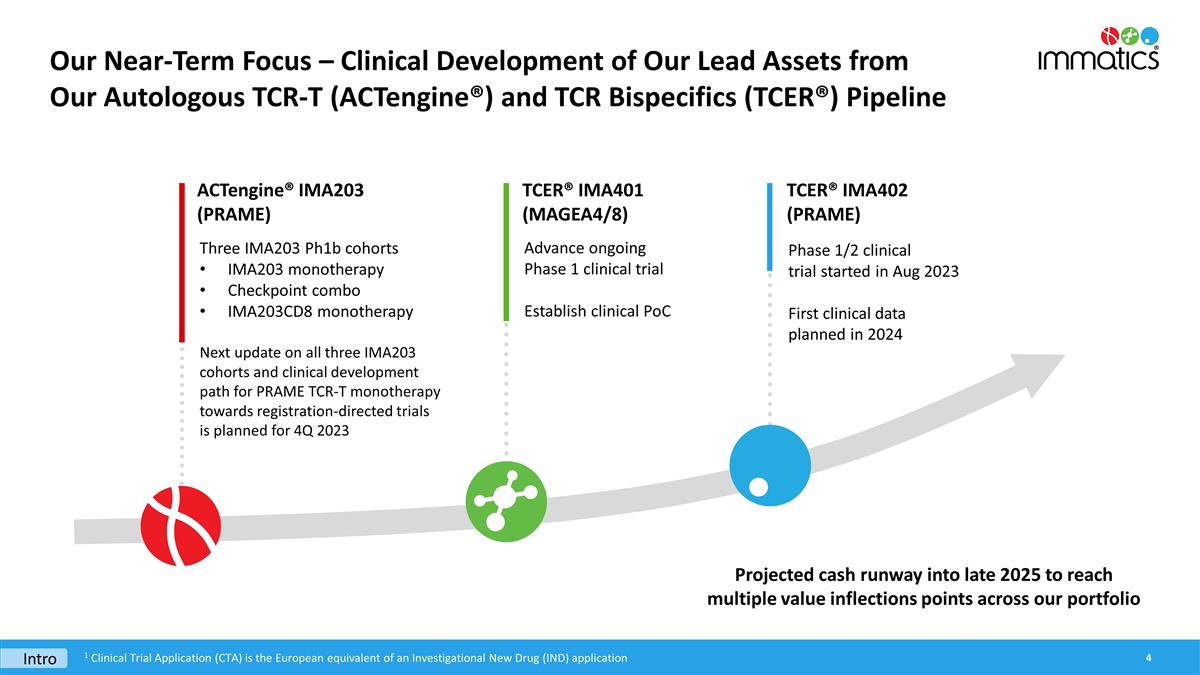
Three IMA203 Ph1b cohorts IMA203
monotherapy Checkpoint combo IMA203CD8 monotherapy Next update on all three IMA203 cohorts and clinical development path for PRAME TCR-T monotherapy towards registration-directed trials is planned for 4Q 2023 ACTengine® IMA203 (PRAME) Advance
ongoing Phase 1 clinical trial Establish clinical PoC TCER® IMA401 (MAGEA4/8) Phase 1/2 clinical trial started in Aug 2023 First clinical data planned in 2024 TCER® IMA402 (PRAME) Intro Projected cash runway into late 2025 to reach
multiple value inflections points across our portfolio Our Near-Term Focus – Clinical Development of Our Lead Assets from Our Autologous TCR-T (ACTengine®) and TCR Bispecifics (TCER®) Pipeline 1 Clinical Trial Application (CTA) is
the European equivalent of an Investigational New Drug (IND) application
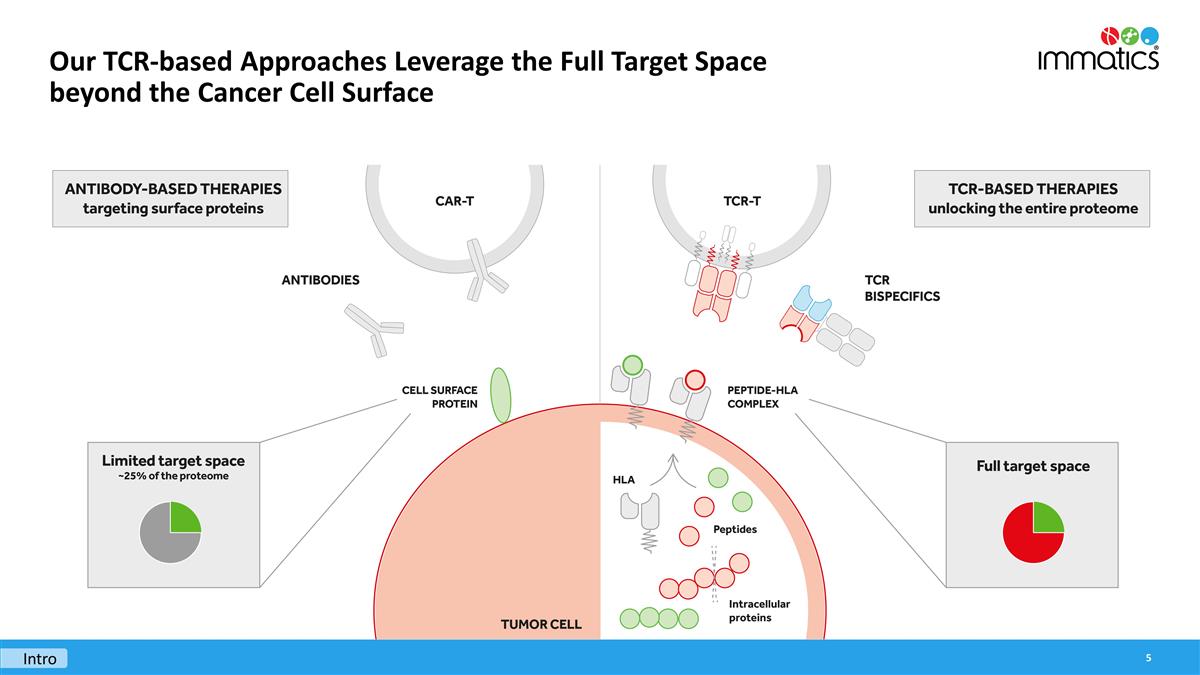
Our TCR-based Approaches Leverage the
Full Target Space beyond the Cancer Cell Surface Intro
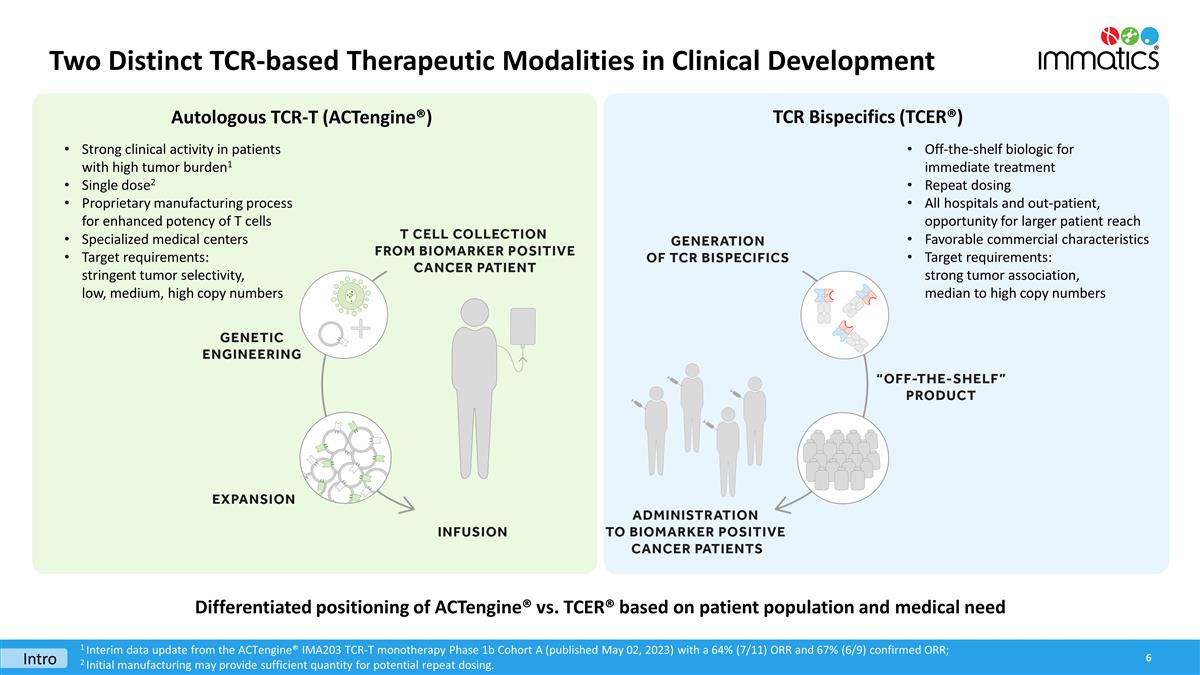
Two Distinct TCR-based Therapeutic
Modalities in Clinical Development Differentiated positioning of ACTengine® vs. TCER® based on patient population and medical need Intro 1 Interim data update from the ACTengine® IMA203 TCR-T monotherapy Phase 1b Cohort A (published
May 02, 2023) with a 64% (7/11) ORR and 67% (6/9) confirmed ORR; 2 Initial manufacturing may provide sufficient quantity for potential repeat dosing. Autologous TCR-T (ACTengine®) TCR Bispecifics (TCER®) Strong clinical activity in
patients with high tumor burden1 Single dose2 Proprietary manufacturing process for enhanced potency of T cells Specialized medical centers Target requirements: stringent tumor selectivity, low, medium, high copy numbers Off-the-shelf biologic for
immediate treatment Repeat dosing All hospitals and out-patient, opportunity for larger patient reach Favorable commercial characteristics Target requirements: strong tumor association, median to high copy numbers
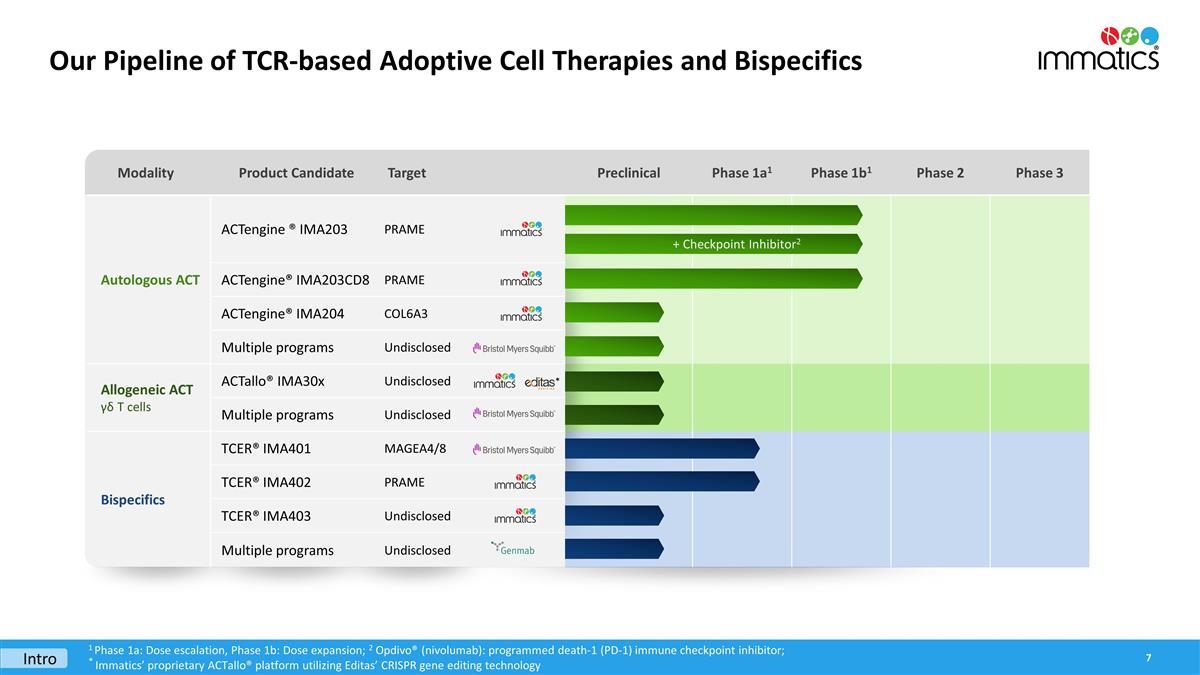
Modality Product Candidate Target
Preclinical Phase 1a1 Phase 1b1 Phase 2 Phase 3 Autologous ACT ACTengine ® IMA203 PRAME ACTengine® IMA203CD8 PRAME ACTengine® IMA204 COL6A3 Autologous ACT Multiple programs Undisclosed Allogeneic ACT γδ T cells ACTallo®
IMA30x Undisclosed Multiple programs Undisclosed Bispecifics TCER® IMA401 MAGEA4/8 TCER® IMA402 PRAME TCER® IMA403 Undisclosed Multiple programs Undisclosed Our Pipeline of TCR-based Adoptive Cell Therapies and Bispecifics Intro 1
Phase 1a: Dose escalation, Phase 1b: Dose expansion; 2 Opdivo® (nivolumab): programmed death-1 (PD-1) immune checkpoint inhibitor; * Immatics’ proprietary ACTallo® platform utilizing Editas’ CRISPR gene editing technology +
Checkpoint Inhibitor2 *
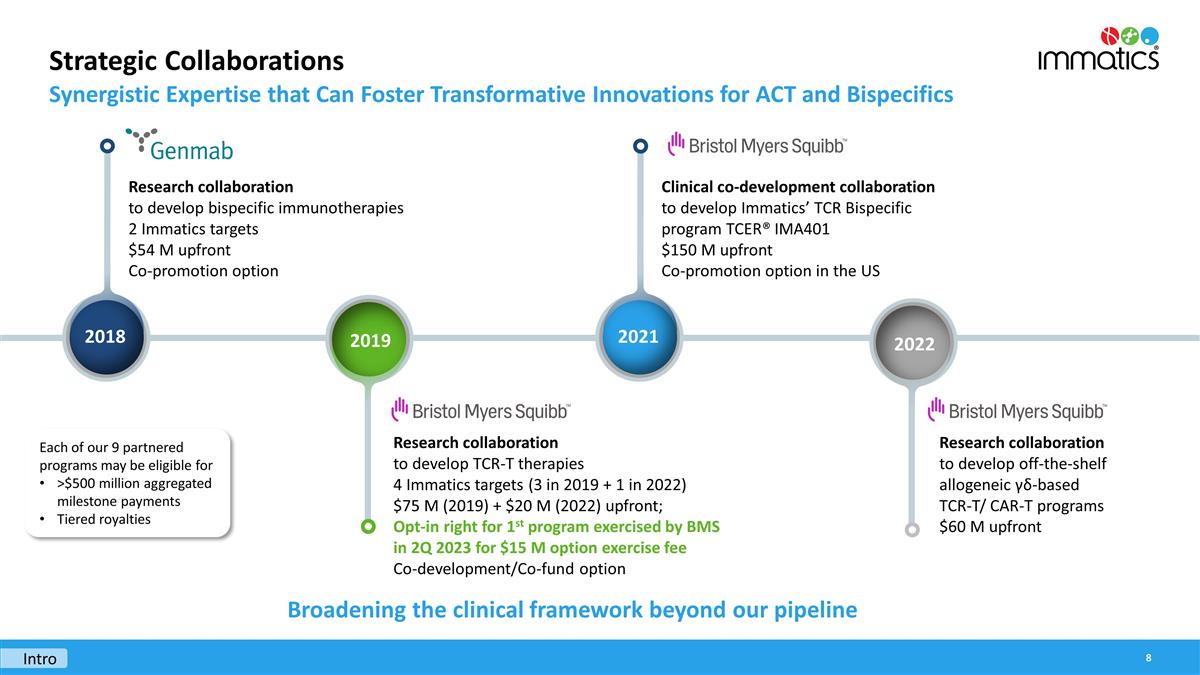
Strategic Collaborations Synergistic
Expertise that Can Foster Transformative Innovations for ACT and Bispecifics Research collaboration to develop bispecific immunotherapies 2 Immatics targets $54 M upfront Co-promotion option Broadening the clinical framework beyond our pipeline
Research collaboration to develop TCR-T therapies 4 Immatics targets (3 in 2019 + 1 in 2022) $75 M (2019) + $20 M (2022) upfront; Opt-in right for 1st program exercised by BMS in 2Q 2023 for $15 M option exercise fee Co-development/Co-fund option
Research collaboration to develop off-the-shelf allogeneic γδ-based TCR-T/ CAR-T programs $60 M upfront Clinical co-development collaboration to develop Immatics’ TCR Bispecific program TCER® IMA401 $150 M upfront Co-promotion
option in the US 2022 2021 2018 2019 Each of our 9 partnered programs may be eligible for >$500 million aggregated milestone payments Tiered royalties Intro
Potential for Large Patient
Populations across Multiple Solid Cancers Uterine Carcinoma – 100% Uterine Carcinosarcoma – 100% Sarcoma Subtypes – up to 100% Cut. Melanoma – 95% Uveal Melanoma1 – 90% Ovarian Carcinoma – 80% Squamous NSCLC
– 65% TNBC – 60% Small Cell Lung Cancer – 55% Kidney Carcinoma – up to 45% Cholangiocarcinoma – 35% Adeno NSCLC – 25% Breast Carcinoma– 25% HNSCC – 25% Esophageal Carcinoma – 20% HCC
– 20% Bladder Carcinoma – 20% Sarcoma Subtypes – up to 80% Squamous NSCLC – 50% HNSCC – 35% Bladder Carcinoma – 30% Esophageal Carcinoma – 25% Uterine Carcinosarcoma – 25% Ovarian Carcinoma – 20%
Melanoma – 20% IMA203 / IMA402 PRAME IMA401 MAGEA4/8 IMA204 COL6A3 Exon 6 Intro Pancreatic Carcinoma – 80% Breast Carcinoma – 75% Stomach Carcinoma – 65% Sarcoma – 65% Esophageal Carcinoma – 60% Squamous
NSCLC– 55% Adeno NSCLC– 55% HNSCC – 55% Uterine Carcinosarcoma – 55% Colorectal Carcinoma – 45% Mesothelioma – 45% Cholangiocarcinoma – 40% Ovarian Carcinoma – 40% Melanoma – 35% Bladder
Carcinoma – 35% ACTengine® and TCER® targets demonstrate high prevalence in multiple solid cancers Target prevalence for selected solid cancer indications are based on TCGA (for SCLC: in-house) RNAseq data combined with a proprietary
mass spec-guided RNA expression threshold; 1 Uveal melanoma target prevalence is based on IMADetect® qPCR testing of screening biopsies from clinical trial patients (n=21)
Realizing the Full Multi-Cancer
Opportunity of PRAME ACTengine® IMA203 (TCR-T) and TCER® IMA402 (TCR Bispecific) Indication % PRAME positive patients1 Uterine Carcinoma Uterine Carcinosarcoma Sarcoma Subtypes Cut. Melanoma Uveal Melanoma2 Ovarian Carcinoma Squamous NSCLC
TNBC Small Cell Lung Cancer Kidney Carcinoma Cholangiocarcinoma Adeno NSCLC Breast Carcinoma HNSCC Esophageal Carcinoma HCC Bladder Carcinoma 100% 100% up to 100% 95% 90% 80% 65% 60% 55% up to 45% 35% 25% 25% 25% 20% 20% 20% ACTengine® IMA203
(TCR-T) Cancer Cell Death PRAME is one of the most promising and most prevalent, clinically validated solid tumor targets known to date Leverage the full potential of targeting PRAME by continued evaluation of the best suited therapeutic modality
(ACTengine® vs. TCER® or both) for each cancer type 1 PRAME target prevalence is based on TCGA (for SCLC: in-house) RNAseq data combined with a proprietary mass spec-guided RNA expression threshold; 2 Uveal melanoma target prevalence is
based on IMADetect® qPCR testing of screening biopsies from clinical trial patients (n=21); NSCLC: Non-small cell lung cancer, TNBC: Triple-negative breast cancer, HNSCC: Head and neck squamous cell carcinoma; HCC: Hepatocellular carcinoma
Intro Phase 1b dose expansion ongoing Initiation of Phase 1/2 trial Aug 2023 TCER® IMA402 (TCR Bispecific)

ACTengine® IMA203 –
TCR-T Targeting PRAME
ACTengine® IMA203 Targeting
PRAME – Mechanism of Action Immatics’ Leading TCR-T Approach IMA203

Key Pillars of Developing a
Successful TCR-T Product Candidate Summary of Interim Update on IMA203 TCR-T Phase 1b Cohort A as of April 2023 Safety Anti-Tumor Activity Durability Product Quality Broad Reach Manageable tolerability at doses as high as ~9x109 TCR-T cells High
rate of objective responses: 64% (7/11) ORR1 67% (6/9) cORR2 Ongoing durable responses at 9+ months mDOR: Not reached min 1.3+, max 8.8+ mFU: 8.5 months Rapid manufacturing time of 7 days (+ 7-day release testing), manufacturing success rate of 94%
Confirmed objective responses in broad range of solid cancer types at low, medium and high PRAME levels above threshold 1 Initial ORR: Objective response rate according to RECIST 1.1 at first scan post infusion at ~week 6; 2 Confirmed ORR (cORR):
Confirmed objective response rate according to RECIST 1.1 for patients with available second scan post infusion at ~month 3 or patients with progressive disease (PD) at any timepoint before this scan; mDOR: median duration of response; mFU: median
follow-up Data cut-off Apr 04, 2023 IMA203
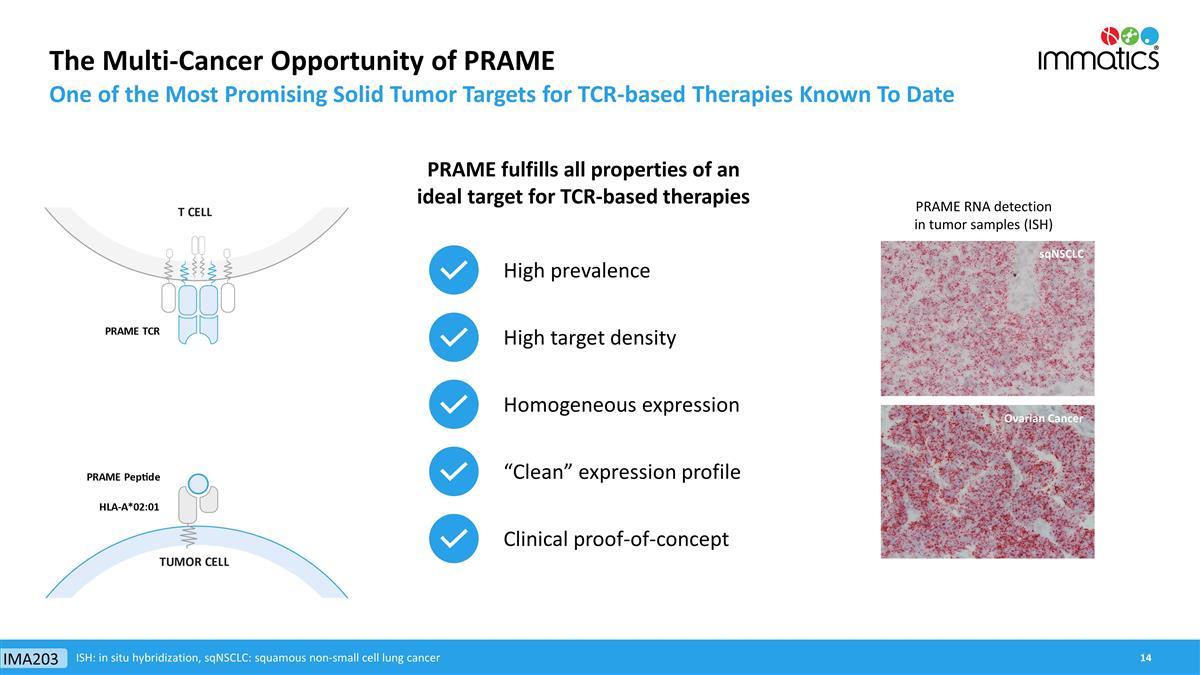
The Multi-Cancer Opportunity of
PRAME One of the Most Promising Solid Tumor Targets for TCR-based Therapies Known To Date High prevalence High target density Homogeneous expression “Clean” expression profile Clinical proof-of-concept sqNSCLC Ovarian Cancer PRAME
fulfills all properties of an ideal target for TCR-based therapies PRAME RNA detection in tumor samples (ISH) ISH: in situ hybridization, sqNSCLC: squamous non-small cell lung cancer IMA203
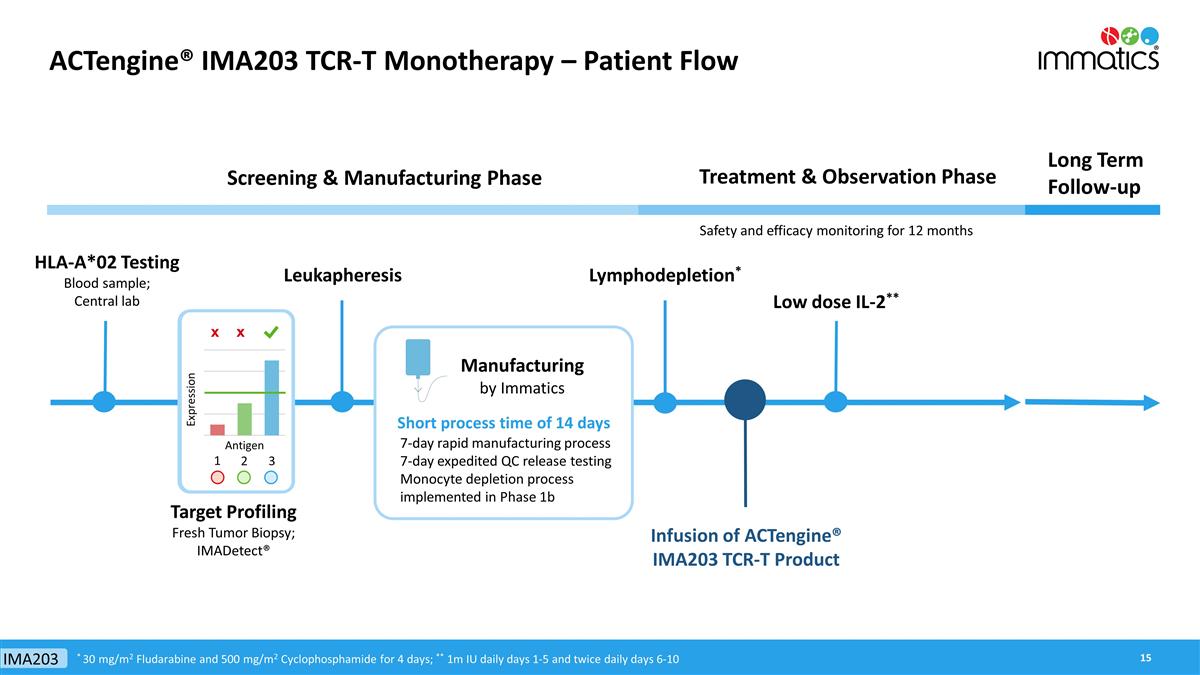
ACTengine® IMA203 TCR-T
Monotherapy – Patient Flow HLA-A*02 Testing Blood sample; Central lab Treatment & Observation Phase Long Term Follow-up Screening & Manufacturing Phase Manufacturing by Immatics Infusion of ACTengine® IMA203 TCR-T Product
Lymphodepletion* Target Profiling Fresh Tumor Biopsy; IMADetect® Low dose IL-2** Safety and efficacy monitoring for 12 months Leukapheresis x x 1 3 2 Short process time of 14 days * 30 mg/m2 Fludarabine and 500 mg/m2 Cyclophosphamide for 4
days; ** 1m IU daily days 1-5 and twice daily days 6-10 7-day rapid manufacturing process 7-day expedited QC release testing Monocyte depletion process implemented in Phase 1b IMA203
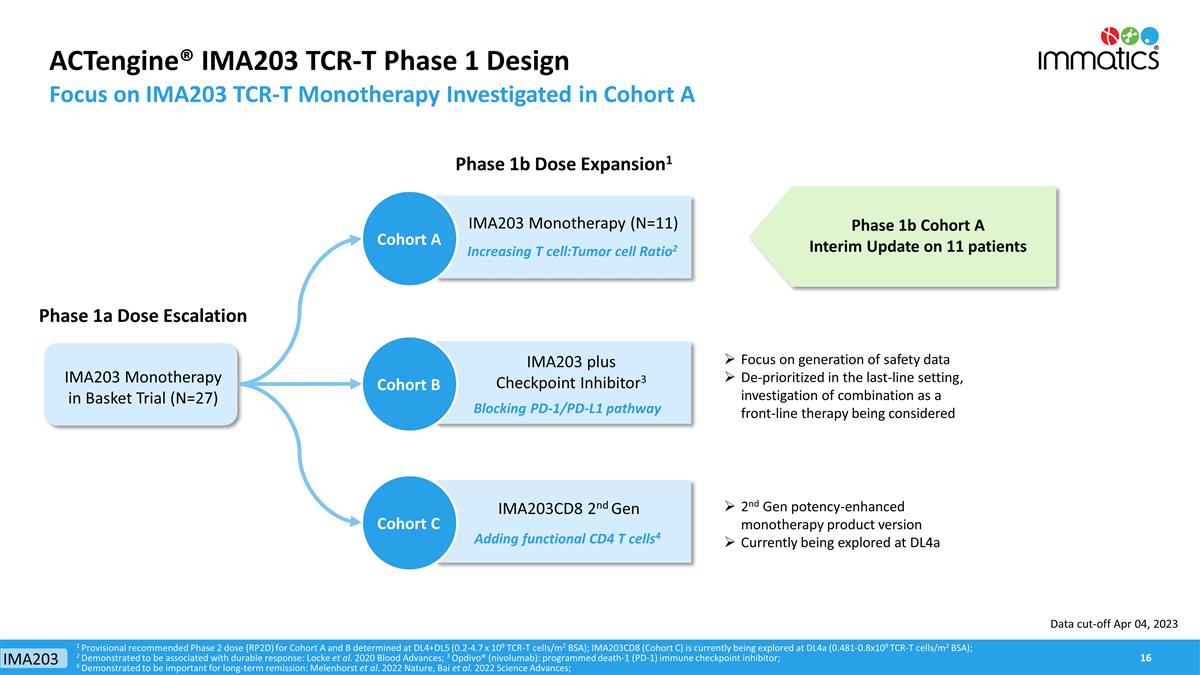
ACTengine® IMA203 TCR-T Phase
1 Design Focus on IMA203 TCR-T Monotherapy Investigated in Cohort A Cohort A Phase 1b Cohort A Interim Update on 11 patients Focus on generation of safety data De-prioritized in the last-line setting, investigation of combination as a front-line
therapy being considered Phase 1b Dose Expansion1 Phase 1a Dose Escalation Cohort A IMA203 Monotherapy (N=11) Increasing T cell:Tumor cell Ratio2 IMA203 Monotherapy in Basket Trial (N=27) 2nd Gen potency-enhanced monotherapy product version
Currently being explored at DL4a Data cut-off Apr 04, 2023 Cohort B Cohort C IMA203 Adding functional CD4 T cells4 IMA203CD8 2nd Gen IMA203 plus Checkpoint Inhibitor3 Blocking PD-1/PD-L1 pathway 1 Provisional recommended Phase 2 dose (RP2D) for
Cohort A and B determined at DL4+DL5 (0.2-4.7 x 109 TCR-T cells/m2 BSA); IMA203CD8 (Cohort C) is currently being explored at DL4a (0.481-0.8x109 TCR-T cells/m2 BSA); 2 Demonstrated to be associated with durable response: Locke et al. 2020 Blood
Advances; 3 Opdivo® (nivolumab): programmed death-1 (PD-1) immune checkpoint inhibitor; 4 Demonstrated to be important for long-term remission: Melenhorst et al. 2022 Nature, Bai et al. 2022 Science Advances;
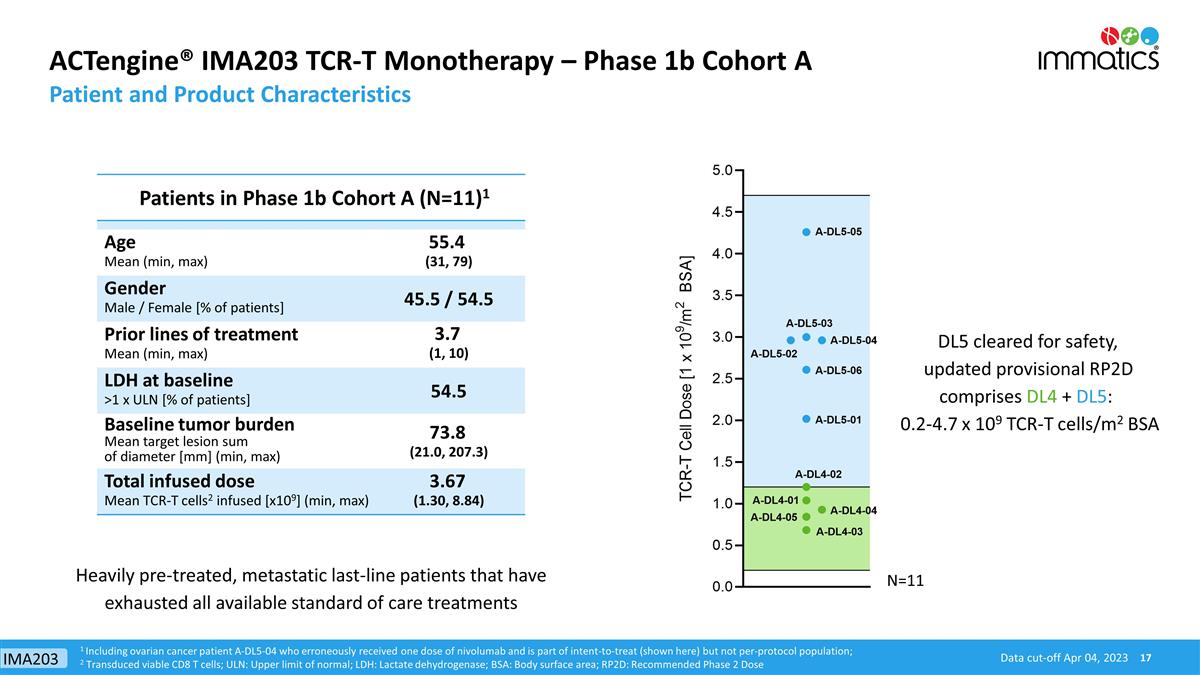
N=11 ACTengine® IMA203 TCR-T
Monotherapy – Phase 1b Cohort A Patient and Product Characteristics Data cut-off Apr 04, 2023 Heavily pre-treated, metastatic last-line patients that have exhausted all available standard of care treatments 1 Including ovarian cancer patient
A-DL5-04 who erroneously received one dose of nivolumab and is part of intent-to-treat (shown here) but not per-protocol population; 2 Transduced viable CD8 T cells; ULN: Upper limit of normal; LDH: Lactate dehydrogenase; BSA: Body surface area;
RP2D: Recommended Phase 2 Dose DL5 cleared for safety, updated provisional RP2D comprises DL4 + DL5:: 0.2-4.7 x 109 TCR-T cells/m2 BSA Patients in Phase 1b Cohort A (N=11)1 (N=11) Age Mean (min, max) 55.4 (31, 79) Gender Male
/ Female [% of patients] 45.5 / 54.5 Prior lines of treatment Mean (min, max) 3.7 (1, 10) LDH at baseline >1 x ULN [% of patients] 54.5 Baseline tumor burden Mean target lesion sum of diameter [mm] (min, max) 73.8 (21.0, 207.3)
Total infused dose Mean TCR-T cells2 infused [x109] (min, max) 3.67 (1.30, 8.84) IMA203

Most Frequent Adverse Events
– Phase 1b Cohort A (N=11) Manageable Treatment-emergent Adverse Events (TEAEs) Expected cytopenia (Grade 1-4) associated with lymphodepletion in all patients Low-moderate cytokine release syndrome (CRS) in 91% (10/11) of patients 45% (5/11)
of patients had Grade 1 CRS (3 in DL4, 2 in DL5) 45% (5/11) of patients had Grade 2 CRS (2 in DL4, 3 in DL5) No dose-dependent increase of CRS No ICANS1 No Dose-limiting toxicity For IMA203 TCR-T monotherapy tolerability profile including Phase 1a
dose escalation, see appendix CRS and ICANS graded by CARTOX criteria (Neelapu et al., 2018); 1 ICANS: Immune Effector Cell-Associated Neurotoxicity Syndrome IMA203 TCR-T monotherapy shows manageable tolerability at total doses as high as ~9x109
TCR-T cells Data cut-off Apr 04, 2023 IMA203
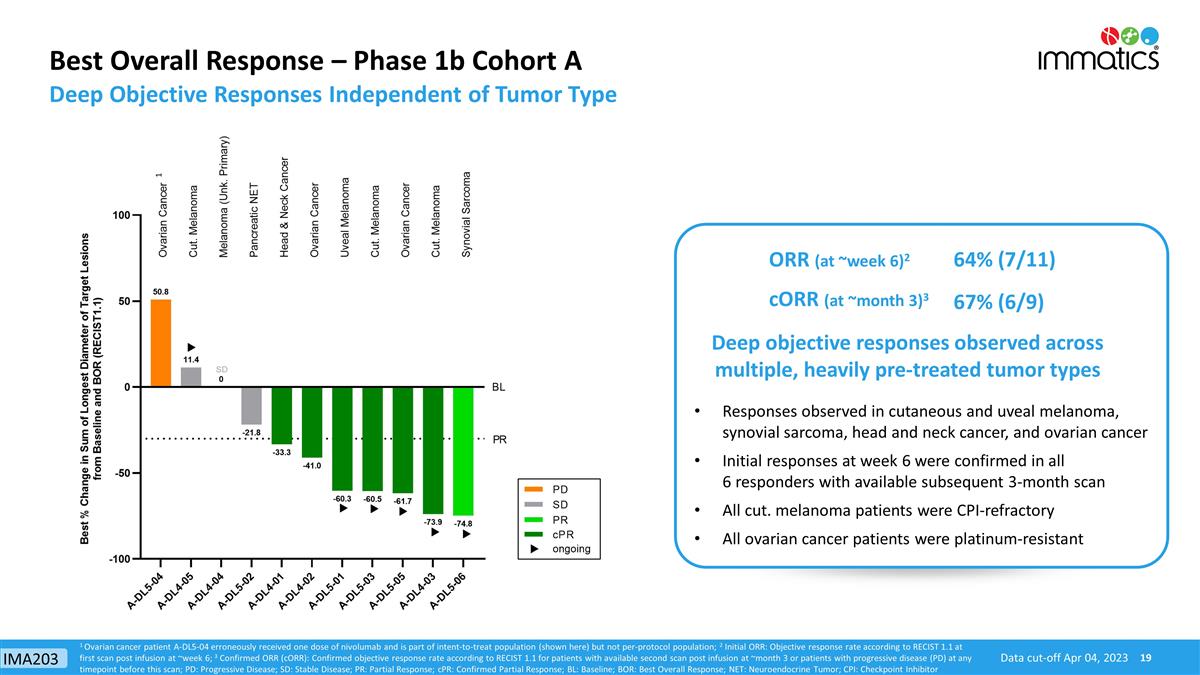
Best Overall Response – Phase
1b Cohort A Deep Objective Responses Independent of Tumor Type 1 Ovarian cancer patient A-DL5-04 erroneously received one dose of nivolumab and is part of intent-to-treat population (shown here) but not per-protocol population; 2 Initial ORR:
Objective response rate according to RECIST 1.1 at first scan post infusion at ~week 6; 3 Confirmed ORR (cORR): Confirmed objective response rate according to RECIST 1.1 for patients with available second scan post infusion at ~month 3 or patients
with progressive disease (PD) at any timepoint before this scan; PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR: Confirmed Partial Response; BL: Baseline; BOR: Best Overall Response; NET: Neuroendocrine Tumor; CPI: Checkpoint
Inhibitor Responses observed in cutaneous and uveal melanoma, synovial sarcoma, head and neck cancer, and ovarian cancer Initial responses at week 6 were confirmed in all 6 responders with available subsequent 3-month scan All cut. melanoma patients
were CPI-refractory All ovarian cancer patients were platinum-resistant ORR (at ~week 6)2 64% (7/11) cORR (at ~month 3)3 67% (6/9) Deep objective responses observed across multiple, heavily pre-treated tumor types Data cut-off Apr 04, 2023 1
IMA203
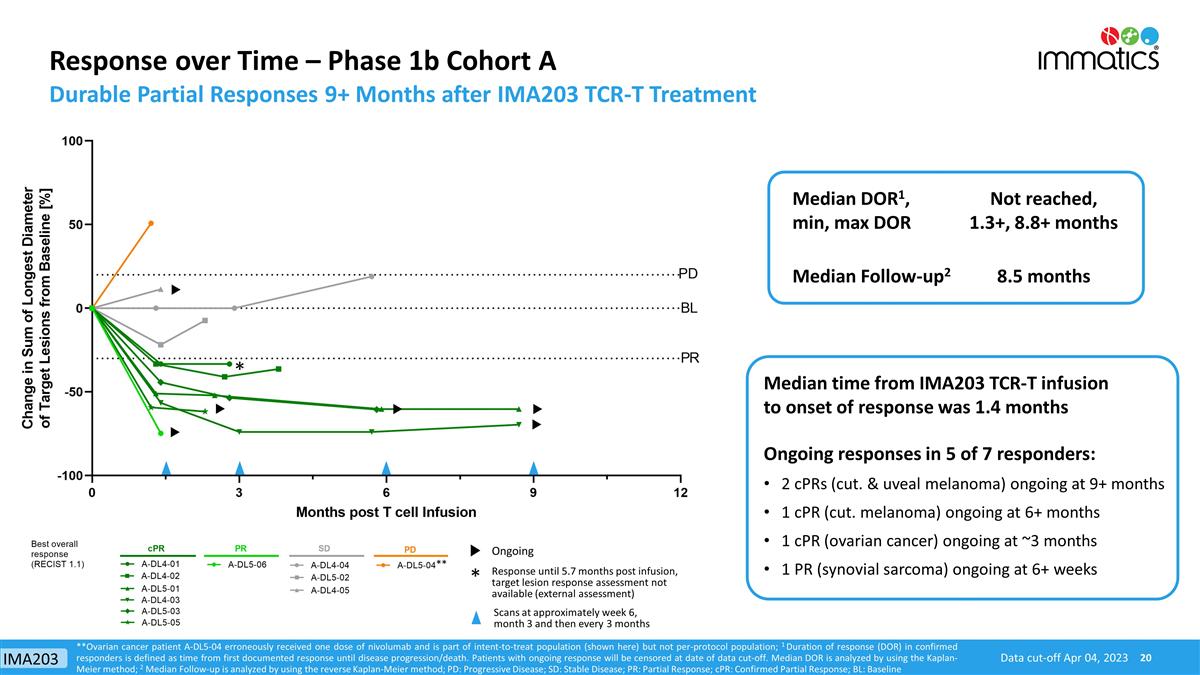
Response over Time – Phase 1b
Cohort A Durable Partial Responses 9+ Months after IMA203 TCR-T Treatment **Ovarian cancer patient A-DL5-04 erroneously received one dose of nivolumab and is part of intent-to-treat population (shown here) but not per-protocol population; 1 Duration
of response (DOR) in confirmed responders is defined as time from first documented response until disease progression/death. Patients with ongoing response will be censored at date of data cut-off. Median DOR is analyzed by using the Kaplan-Meier
method; 2 Median Follow-up is analyzed by using the reverse Kaplan-Meier method; PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR: Confirmed Partial Response; BL: Baseline Median time from IMA203 TCR-T infusion to onset
of response was 1.4 months Ongoing responses in 5 of 7 responders: 2 cPRs (cut. & uveal melanoma) ongoing at 9+ months 1 cPR (cut. melanoma) ongoing at 6+ months 1 cPR (ovarian cancer) ongoing at ~3 months 1 PR (synovial sarcoma) ongoing at 6+
weeks Median DOR1, min, max DOR Not reached, 1.3+, 8.8+ months Median Follow-up2 8.5 months Scans at approximately week 6, month 3 and then every 3 months Ongoing * ** * Response until 5.7 months post infusion, target lesion response assessment not
available (external assessment) Data cut-off Apr 04, 2023 IMA203
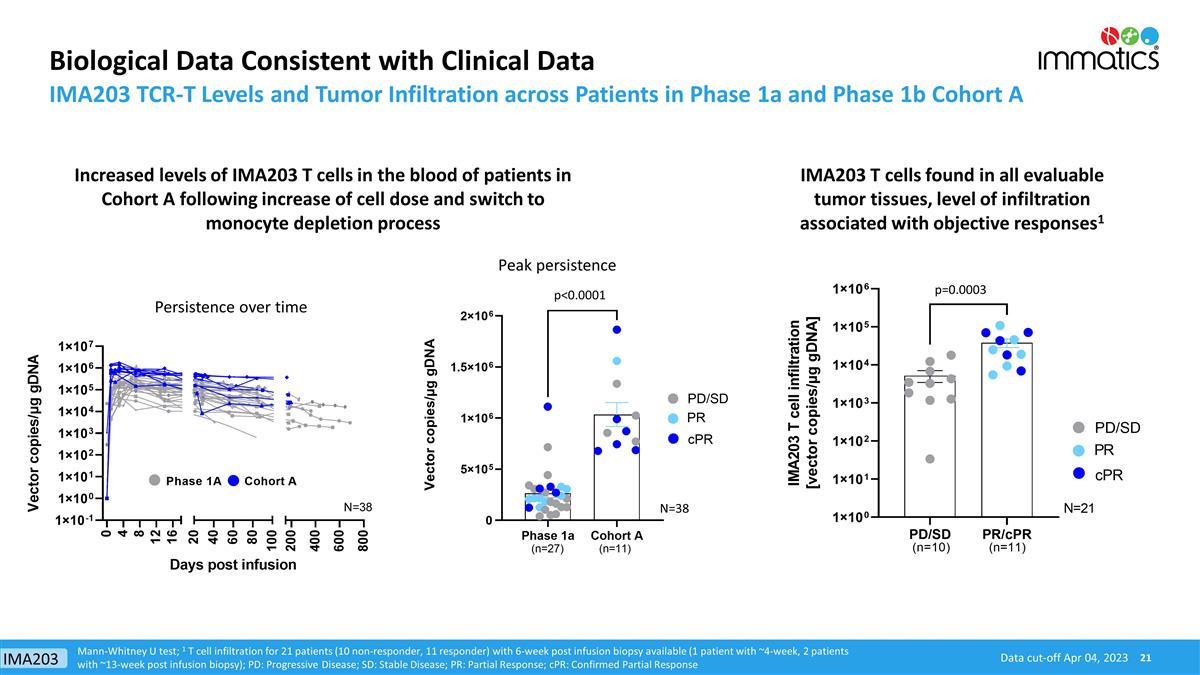
Biological Data Consistent with
Clinical Data IMA203 TCR-T Levels and Tumor Infiltration across Patients in Phase 1a and Phase 1b Cohort A IMA203 T cells found in all evaluable tumor tissues, level of infiltration associated with objective responses1 Increased levels of IMA203 T
cells in the blood of patients in Cohort A following increase of cell dose and switch to monocyte depletion process Data cut-off Apr 04, 2023 Mann-Whitney U test; 1 T cell infiltration for 21 patients (10 non-responder, 11 responder) with 6-week
post infusion biopsy available (1 patient with ~4-week, 2 patients with ~13-week post infusion biopsy); PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR: Confirmed Partial Response p=0.0003 Vector copies/µg gDNA
p<0.0001 Persistence over time Peak persistence N=38 IMA203
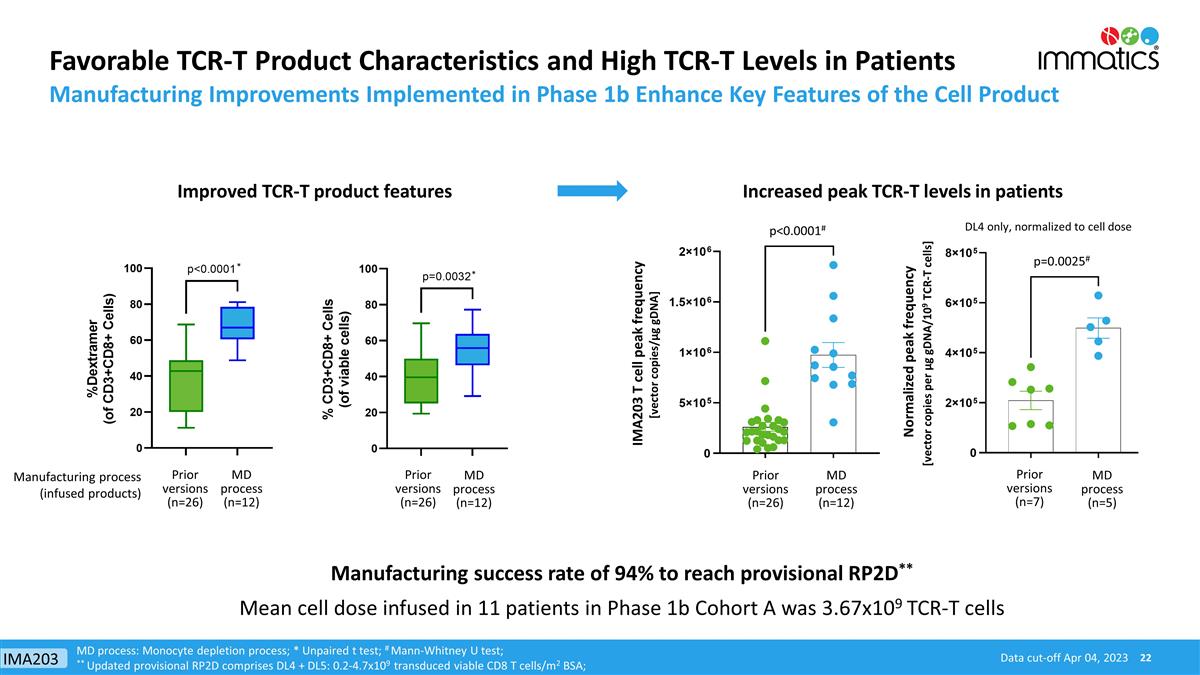
Favorable TCR-T Product
Characteristics and High TCR-T Levels in Patients Manufacturing Improvements Implemented in Phase 1b Enhance Key Features of the Cell Product Manufacturing success rate of 94% to reach provisional RP2D** Mean cell dose infused in 11 patients in
Phase 1b Cohort A was 3.67x109 TCR-T cells Prior versions (n=26) Manufacturing process (infused products) MD process (n=12) Prior versions (n=26) MD process (n=12) MD process: Monocyte depletion process; * Unpaired t test; # Mann-Whitney U
test; ** Updated provisional RP2D comprises DL4 + DL5: 0.2-4.7x109 transduced viable CD8 T cells/m2 BSA; Increased peak TCR-T levels in patients Improved TCR-T product features IMA203 T cell peak frequency [vector copies/µg
gDNA] MD process (n=12) Prior versions (n=26) MD process (n=5) Prior versions (n=7) p<0.0001# p=0.0025# DL4 only, normalized to cell dose Normalized peak frequency [vector copies per µg gDNA/109 TCR-T cells] Data cut-off Apr
04, 2023 * * IMA203
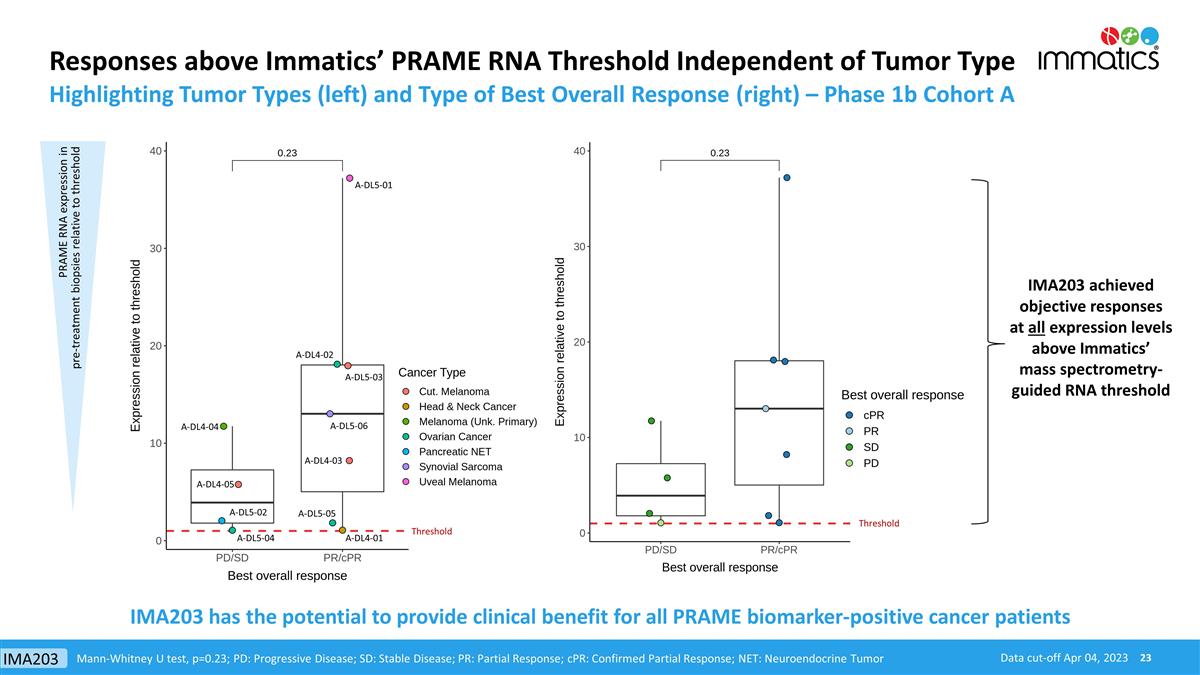
Responses above Immatics’
PRAME RNA Threshold Independent of Tumor Type Highlighting Tumor Types (left) and Type of Best Overall Response (right) – Phase 1b Cohort A Mann-Whitney U test, p=0.23; PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR:
Confirmed Partial Response; NET: Neuroendocrine Tumor PRAME RNA expression in pre-treatment biopsies relative to threshold IMA203 has the potential to provide clinical benefit for all PRAME biomarker-positive cancer patients IMA203 achieved
objective responses at all expression levels above Immatics’ mass spectrometry- guided RNA threshold A-DL5-01 A-DL4-04 A-DL4-05 A-DL5-02 A-DL5-04 A-DL4-03 A-DL5-05 A-DL4-01 A-DL5-03 A-DL4-02 A-DL5-06 Data cut-off Apr 04, 2023 Threshold
Threshold IMA203
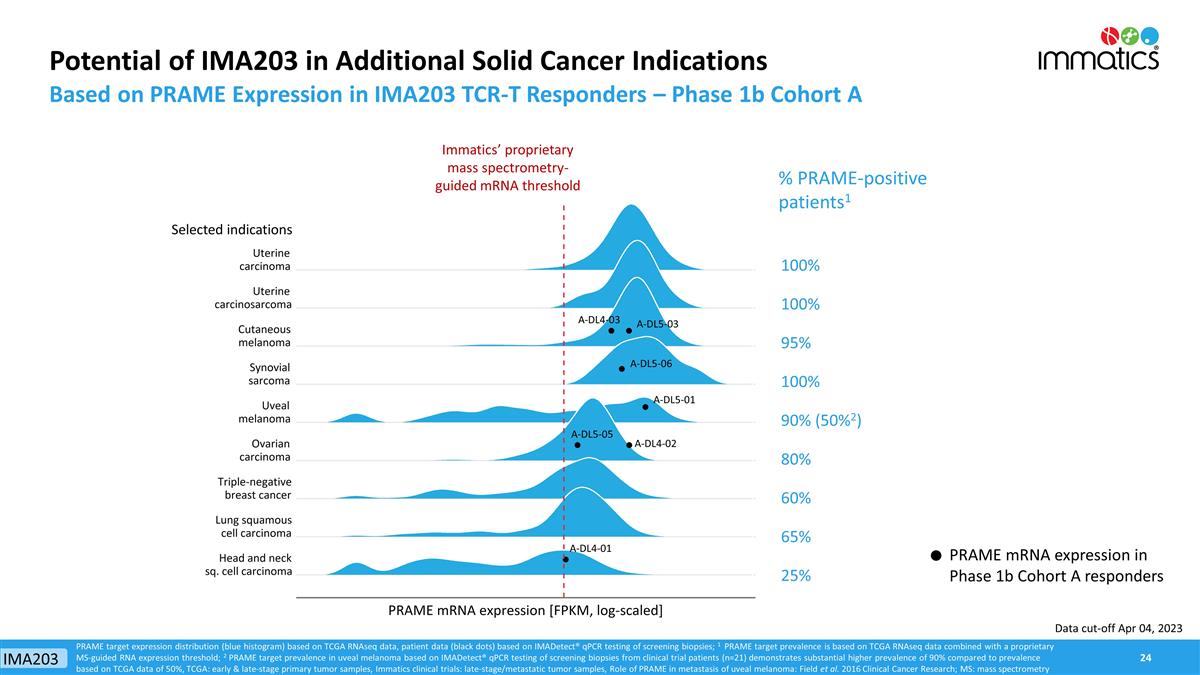
Potential of IMA203 in Additional
Solid Cancer Indications Based on PRAME Expression in IMA203 TCR-T Responders – Phase 1b Cohort A Immatics’ proprietary mass spectrometry-guided mRNA threshold 100% 100% 95% 100% 90% (50%2) 80% 60% 65% 25% % PRAME-positive patients1
PRAME target expression distribution (blue histogram) based on TCGA RNAseq data, patient data (black dots) based on IMADetect® qPCR testing of screening biopsies; 1 PRAME target prevalence is based on TCGA RNAseq data combined with a
proprietary MS-guided RNA expression threshold; 2 PRAME target prevalence in uveal melanoma based on IMADetect® qPCR testing of screening biopsies from clinical trial patients (n=21) demonstrates substantial higher prevalence of 90% compared to
prevalence based on TCGA data of 50%, TCGA: early & late-stage primary tumor samples, Immatics clinical trials: late-stage/metastatic tumor samples, Role of PRAME in metastasis of uveal melanoma: Field et al. 2016 Clinical Cancer Research; MS:
mass spectrometry PRAME mRNA expression in Phase 1b Cohort A responders Data cut-off Apr 04, 2023 Selected indications A-DL4-03 A-DL5-03 A-DL5-06 A-DL5-01 A-DL4-02 A-DL5-05 A-DL4-01 IMA203

ACTengine® IMA203 TCR-T
Monotherapy Targeting PRAME Summary of Phase 1b Cohort A Interim Data Update Manageable tolerability with no high-grade CRS, no ICANS in 11 patients in Cohort A1 Objective responses observed in heavily pre-treated last-line solid cancer patients
including checkpoint-refractory cutaneous melanoma, platinum-resistant ovarian cancer, uveal melanoma, head and neck cancer, synovial sarcoma High objective response rate (ORR): 64% (7/11) ORR (at ~week 6) 67% (6/9) cORR (at ~month 3) Ongoing
durable responses: Median duration of response not reached at a median follow-up time of 8.5 months Ongoing PRs 9+ months after IMA203 TCR-T treatment Objective responses independent of tumor type at low, medium and high PRAME levels above
threshold Manufacturing success rate of 94% to reach current RP2D, rapid 7-day manufacturing process (+7-day release testing) Increased confidence in the success and broad potential of targeting PRAME and our product candidate IMA203 TCR-T 1 For
IMA203 TCR-T monotherapy tolerability profile including Phase 1a dose escalation, see appendix; CRS: Cytokine Release Syndrome; ICANS: Immune effector cell-associated neurotoxicity syndrome; RP2D: provisional recommended Phase 2 dose Data cut-off
Apr 04, 2023 IMA203

Immatics’ ACTengine®
IMA203 TCR-T Development Strategy Two-Pillared Strategy Objective: Expand development to other cancer types Signal finding in other cancer types with a broad patient reach, such as ovarian cancer, uterine cancer, lung cancer, breast cancer, head and
neck cancer GO BROAD Next update on all three IMA203 Phase 1b cohorts including the projected clinical development path for PRAME-targeted TCR-T monotherapy towards registration-directed trials is planned for 4Q 2023 IMA203 Objective: Deliver
best-in-class therapy in 1-2 last-line solid cancer types as fast as possible Focus on cutaneous melanoma, uveal melanoma and potentially other tumor types with high PRAME prevalence where clinical proof-of-concept has been demonstrated Highly
modular and scalable manufacturing facility expected to be operational in 2024 to support efforts to maximize speed to market Planned start of a first Phase 2 trial in 1H 2024 – targeted to be already registration-directed FAST &
FOCUSED
ACTengine® IMA203 TCR-T
Product Manufacturing Enhancing Manufacturing Process and Capabilities Leukapheresis Rapid Manufacturing Process 1 week Expedited QC Release Testing 1 week Infusion-ready Manufacturing of ACTengine® IMA203 TCR-T & other future autologous
/allogeneic candidates Expected to be operational in 2024 Approx. 100,000 sq ft in Houston area, TX – modular and flexible design Early-stage and registration-directed clinical trials as well as initial commercial supply State-of-the-art
research & GMP manufacturing facility Short manufacturing turnaround time IMA203
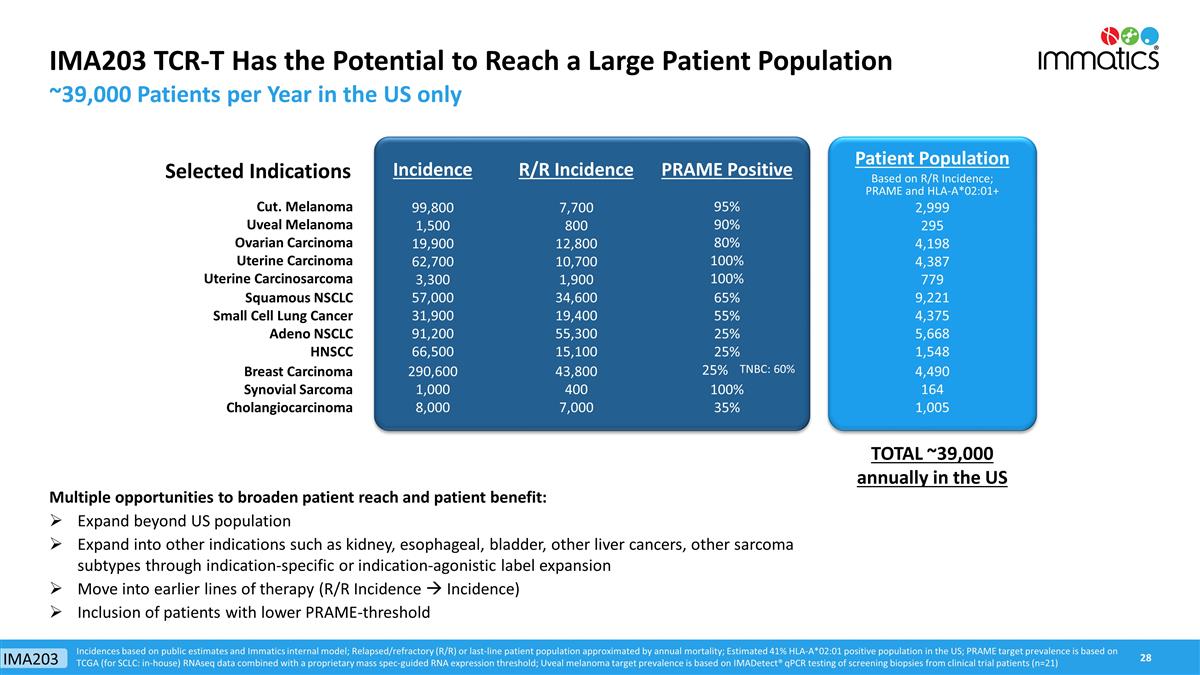
Selected Indications Incidence R/R
Incidence PRAME Positive Patient Population Based on R/R Incidence; PRAME and HLA-A*02:01+ Cut. Melanoma 99,800 7,700 95% 2,999 Uveal Melanoma 1,500 800 90% 295 Ovarian Carcinoma 19,900 12,800 80% 4,198 Uterine Carcinoma 62,700 10,700 100% 4,387
Uterine Carcinosarcoma 3,300 1,900 100% 779 Squamous NSCLC 57,000 34,600 65% 9,221 Small Cell Lung Cancer 31,900 19,400 55% 4,375 Adeno NSCLC 91,200 55,300 25% 5,668 HNSCC 66,500 15,100 25% 1,548 Breast Carcinoma 290,600 43,800 25% TNBC: 60% 4,490
Synovial Sarcoma 1,000 400 100% 164 Cholangiocarcinoma 8,000 7,000 35% 1,005 IMA203 TCR-T Has the Potential to Reach a Large Patient Population ~39,000 Patients per Year in the US only Incidences based on public estimates and Immatics internal
model; Relapsed/refractory (R/R) or last-line patient population approximated by annual mortality; Estimated 41% HLA-A*02:01 positive population in the US; PRAME target prevalence is based on TCGA (for SCLC: in-house) RNAseq data combined with a
proprietary mass spec-guided RNA expression threshold; Uveal melanoma target prevalence is based on IMADetect® qPCR testing of screening biopsies from clinical trial patients (n=21) Multiple opportunities to broaden patient reach and patient
benefit: Expand beyond US population Expand into other indications such as kidney, esophageal, bladder, other liver cancers, other sarcoma subtypes through indication-specific or indication-agonistic label expansion Move into earlier lines of
therapy (R/R Incidence à Incidence) Inclusion of patients with lower PRAME-threshold TOTAL ~39,000 annually in the US IMA203
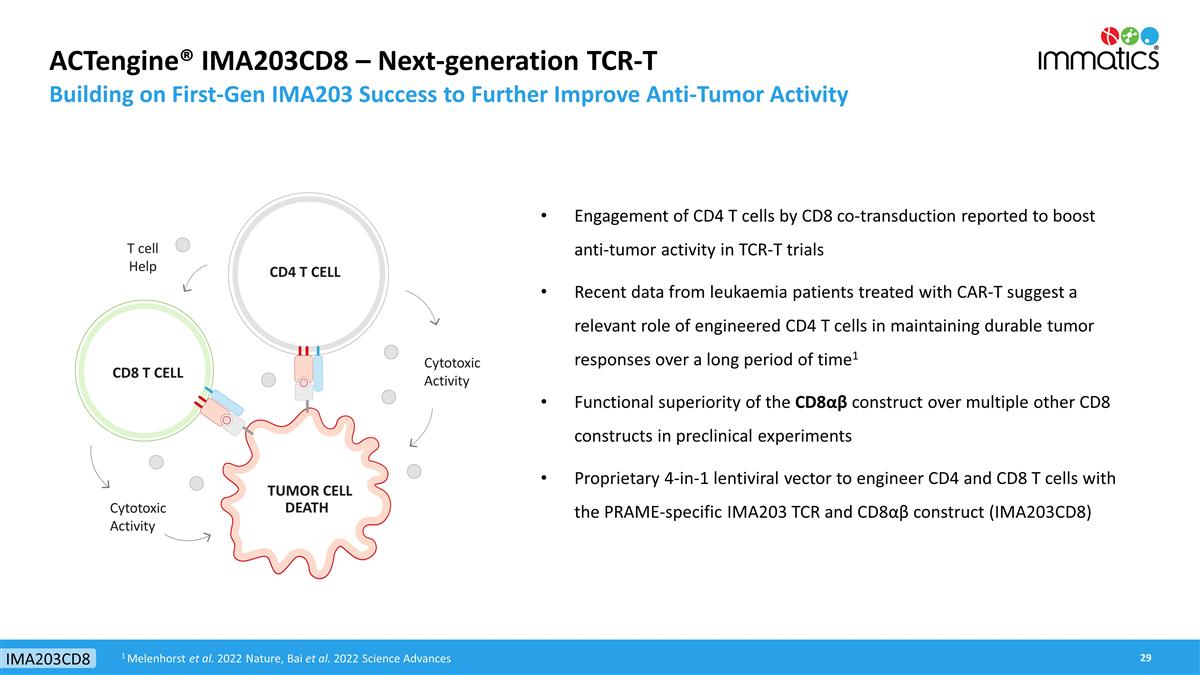
ACTengine® IMA203CD8 –
Next-generation TCR-T Building on First-Gen IMA203 Success to Further Improve Anti-Tumor Activity Engagement of CD4 T cells by CD8 co-transduction reported to boost anti-tumor activity in TCR-T trials Recent data from leukaemia patients treated with
CAR-T suggest a relevant role of engineered CD4 T cells in maintaining durable tumor responses over a long period of time1 Functional superiority of the CD8αβ construct over multiple other CD8 constructs in preclinical experiments
Proprietary 4-in-1 lentiviral vector to engineer CD4 and CD8 T cells with the PRAME-specific IMA203 TCR and CD8αβ construct (IMA203CD8) TUMOR CELL DEATH CD4 T CELL Cytotoxic Activity CD8 T CELL T cell Help Cytotoxic Activity 1 Melenhorst
et al. 2022 Nature, Bai et al. 2022 Science Advances IMA203CD8
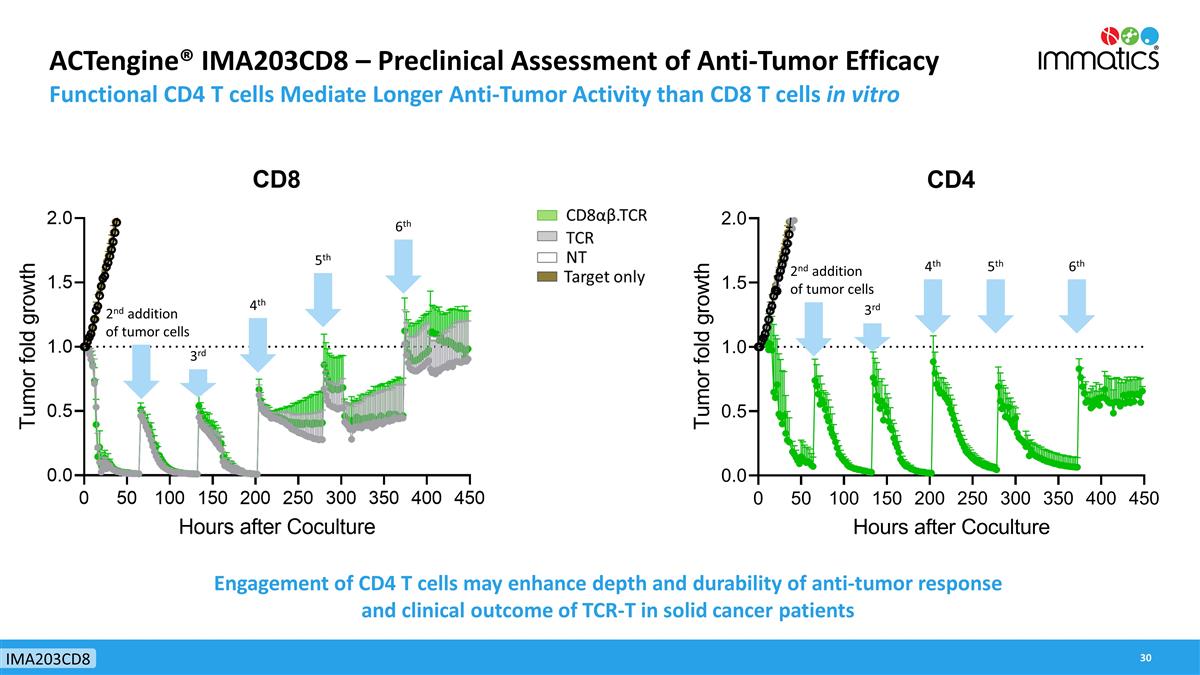
ACTengine® IMA203CD8 –
Preclinical Assessment of Anti-Tumor Efficacy Functional CD4 T cells Mediate Longer Anti-Tumor Activity than CD8 T cells in vitro 2nd addition of tumor cells 3rd 4th 5th 6th 2nd addition of tumor cells 3rd 4th 5th 6th IMA203CD8 Engagement of CD4 T
cells may enhance depth and durability of anti-tumor response and clinical outcome of TCR-T in solid cancer patients

ACTengine® IMA204 –
TCR-T Targeting COL6A3 Exon 6

ACTengine® IMA204
First-in-Class TCR-T Targeting Tumor Stroma Key Features HLA-A*02-presented peptide derived from COL6A3 exon 6 Naturally and specifically presented on tumors at high target density1: 100-700 copies/cell Novel tumor stroma target identified and
validated by XPRESIDENT® quant. mass spectrometry platform High-affinity, specific TCR targeting COL6A3 exon 6 Affinity-maturated, CD8-independent TCR High functional avidity2: ~0.01ng/ml Identified and characterized by XCEPTOR® TCR
discovery and engineering platform CD8-independent, next-generation TCR engages both, CD8 and CD4 T cells In vitro anti-tumor activity against target-positive cell lines in CD8 and CD4 T cells Complete tumor eradication in in vivo mouse models
Pancreatic Carcinoma – 80% Breast Carcinoma – 75% Stomach Carcinoma – 65% Sarcoma – 65% Esophageal Carcinoma – 60% Squamous NSCLC– 55% Adeno NSCLC– 55% HNSCC – 55% Uterine Carcinosarcoma – 55%
Colorectal Carcinoma – 45% Mesothelioma – 45% Cholangiocarcinoma – 40% Ovarian Carcinoma – 40% Melanoma – 35% Bladder Carcinoma – 35% 1 Target density: peptide copy number per tumor cell, approximate range
representing the majority of tumor samples analyzed; 2 Functional avidity: EC50 half maximal effective concentration; 3 Solid cancer indications with 20% or more target expression, Target prevalence for selected cancer indications based on mRNA
expression (TCGA and Immatics inhouse data) TARGET TCR PRECLINICAL DATA PATIENT POPULATION3 IMA204 provides a promising therapeutic opportunity for a broad patient population as monotherapy or in combination with TCR-T cells directed against tumor
targets IMA204
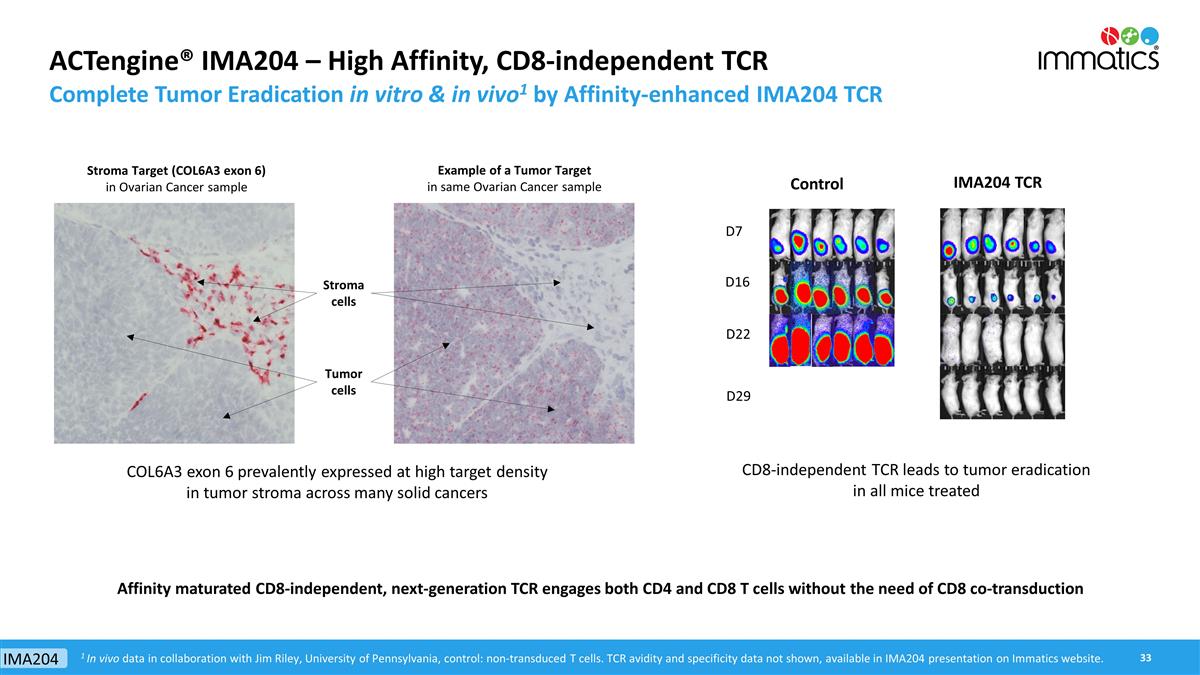
ACTengine® IMA204 – High
Affinity, CD8-independent TCR Complete Tumor Eradication in vitro & in vivo1 by Affinity-enhanced IMA204 TCR CD8-independent TCR leads to tumor eradication in all mice treated Control IMA204 TCR D7 D16 D22 D29 Affinity maturated CD8-independent,
next-generation TCR engages both CD4 and CD8 T cells without the need of CD8 co-transduction Stroma cells Tumor cells Stroma Target (COL6A3 exon 6) in Ovarian Cancer sample Example of a Tumor Target in same Ovarian Cancer sample 1 In vivo data in
collaboration with Jim Riley, University of Pennsylvania, control: non-transduced T cells. TCR avidity and specificity data not shown, available in IMA204 presentation on Immatics website. COL6A3 exon 6 prevalently expressed at high target density
in tumor stroma across many solid cancers IMA204

ACTallo® – Our
Next-generation Off-the-shelf TCR-T
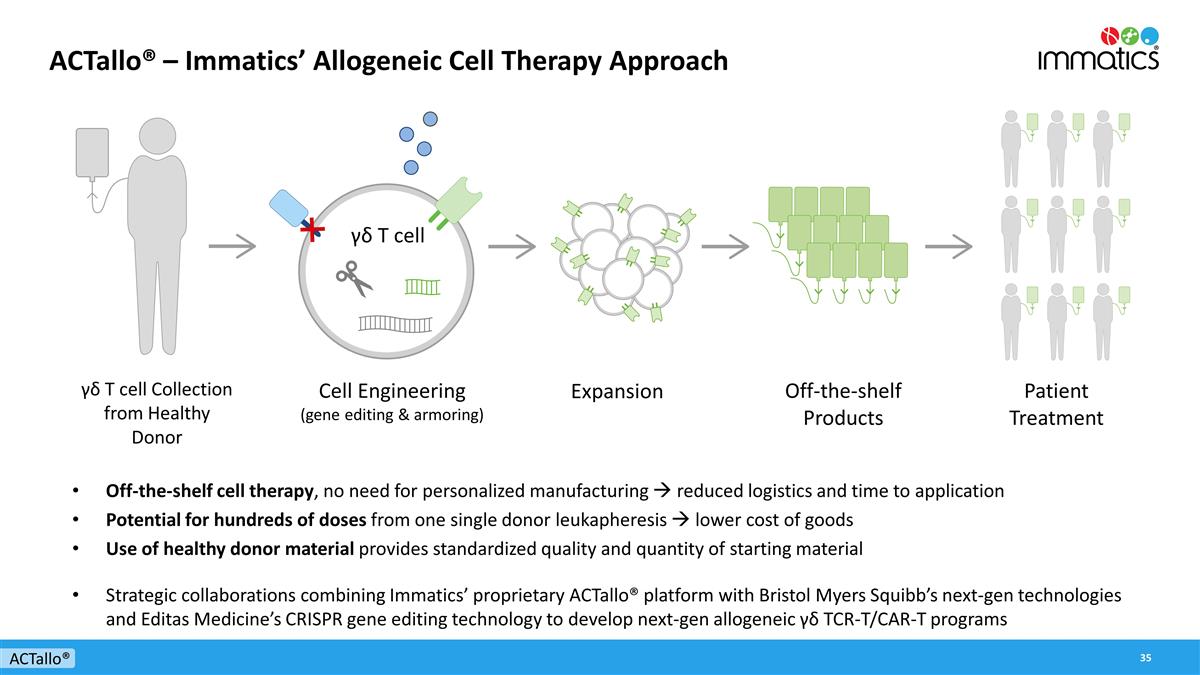
ACTallo® –
Immatics’ Allogeneic Cell Therapy Approach Off-the-shelf cell therapy, no need for personalized manufacturing à reduced logistics and time to application Potential for hundreds of doses from one single donor leukapheresis à lower
cost of goods Use of healthy donor material provides standardized quality and quantity of starting material Strategic collaborations combining Immatics’ proprietary ACTallo® platform with Bristol Myers Squibb’s next-gen technologies
and Editas Medicine’s CRISPR gene editing technology to develop next-gen allogeneic γδ TCR-T/CAR-T programs ACTallo® γδ T cell Cell Engineering (gene editing & armoring) γδ T cell Collection from Healthy
Donor Expansion Off-the-shelf Products Patient Treatment
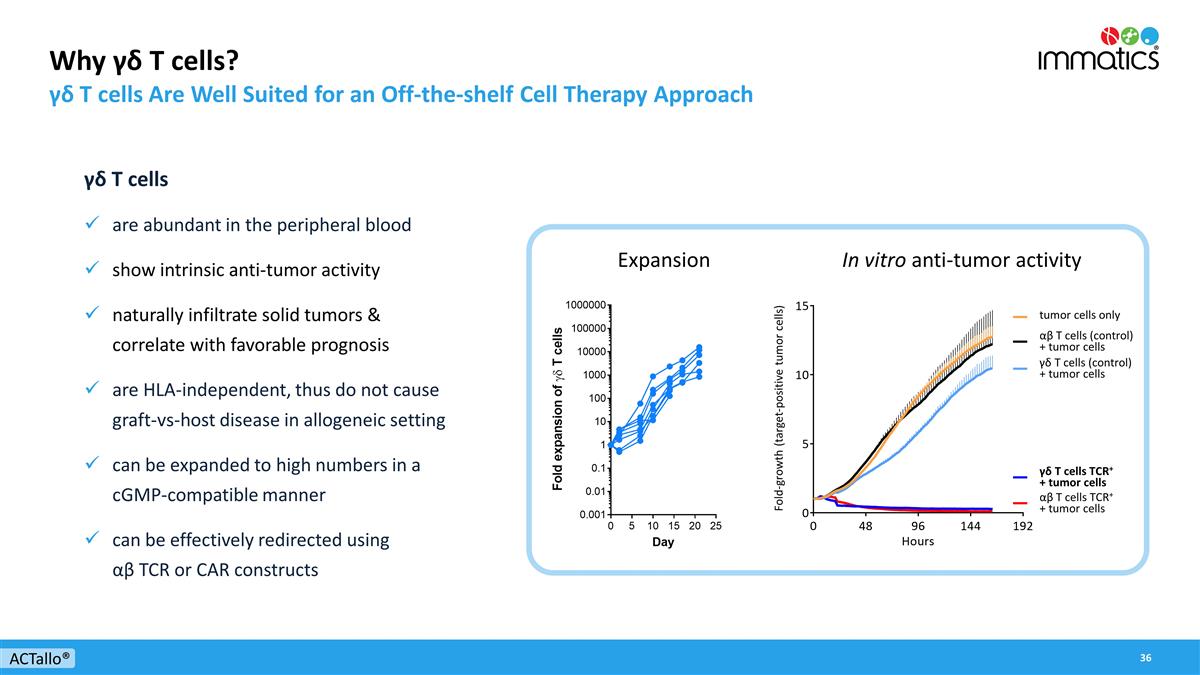
Why γδ T cells?
γδ T cells Are Well Suited for an Off-the-shelf Cell Therapy Approach γδ T cells are abundant in the peripheral blood show intrinsic anti-tumor activity naturally infiltrate solid tumors & correlate with favorable prognosis
are HLA-independent, thus do not cause graft-vs-host disease in allogeneic setting can be expanded to high numbers in a cGMP-compatible manner can be effectively redirected using αβ TCR or CAR constructs In vitro anti-tumor activity
γδ T cells (control) + tumor cells tumor cells only αβ T cells (control) + tumor cells γδ T cells TCR+ + tumor cells αβ T cells TCR+ + tumor cells ACTallo® Expansion Fold-growth (target-positive tumor
cells)

TCER® – TCR Bispecifics
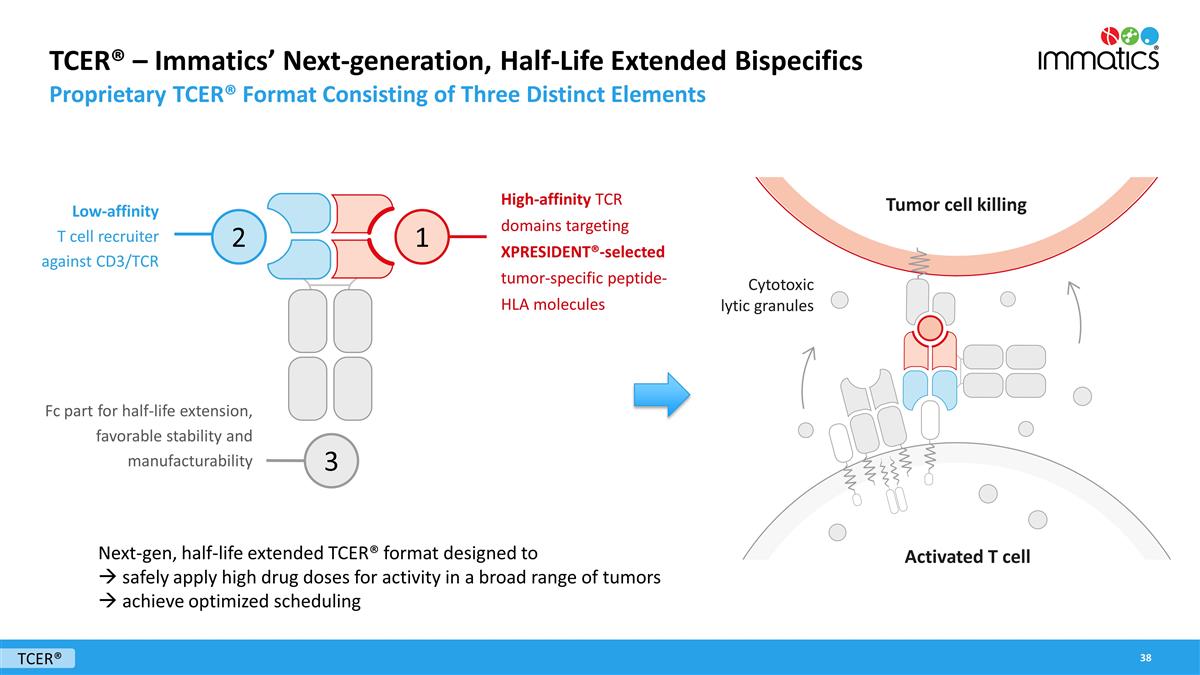
TCER® – Immatics’
Next-generation, Half-Life Extended Bispecifics Proprietary TCER® Format Consisting of Three Distinct Elements High-affinity TCR domains targeting XPRESIDENT®-selected tumor-specific peptide-HLA molecules Low-affinity T cell recruiter
against CD3/TCR Fc part for half-life extension, favorable stability and manufacturability Next-gen, half-life extended TCER® format designed to à safely apply high drug doses for activity in a broad range of tumors à achieve
optimized scheduling 2 1 3 Cytotoxic lytic granules Tumor cell killing Activated T cell TCER®
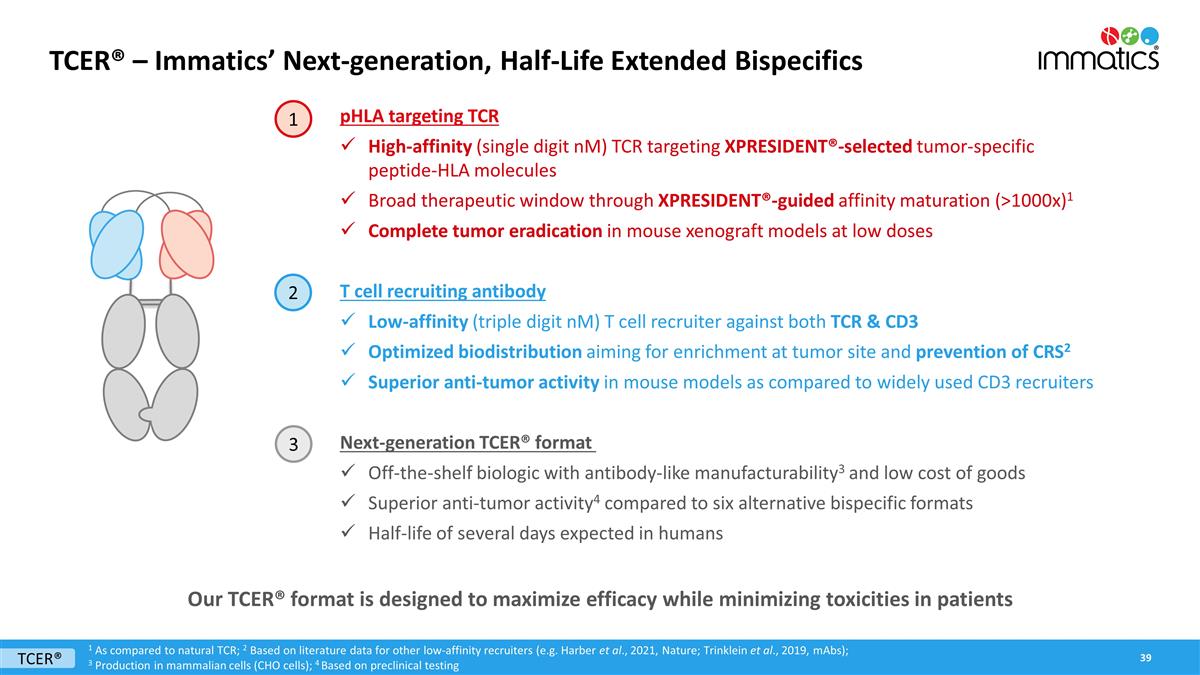
TCER® – Immatics’
Next-generation, Half-Life Extended Bispecifics pHLA targeting TCR High-affinity (single digit nM) TCR targeting XPRESIDENT®-selected tumor-specific peptide-HLA molecules Broad therapeutic window through XPRESIDENT®-guided affinity
maturation (>1000x)1 Complete tumor eradication in mouse xenograft models at low doses T cell recruiting antibody Low-affinity (triple digit nM) T cell recruiter against both TCR & CD3 Optimized biodistribution aiming for
enrichment at tumor site and prevention of CRS2 Superior anti-tumor activity in mouse models as compared to widely used CD3 recruiters Next-generation TCER® format Off-the-shelf biologic with antibody-like
manufacturability3 and low cost of goods Superior anti-tumor activity4 compared to six alternative bispecific formats Half-life of several days expected in humans Our TCER® format is designed to maximize efficacy while minimizing
toxicities in patients 1 As compared to natural TCR; 2 Based on literature data for other low-affinity recruiters (e.g. Harber et al., 2021, Nature; Trinklein et al., 2019, mAbs); 3 Production in mammalian cells (CHO cells); 4 Based on preclinical
testing TCER® 1 2 3
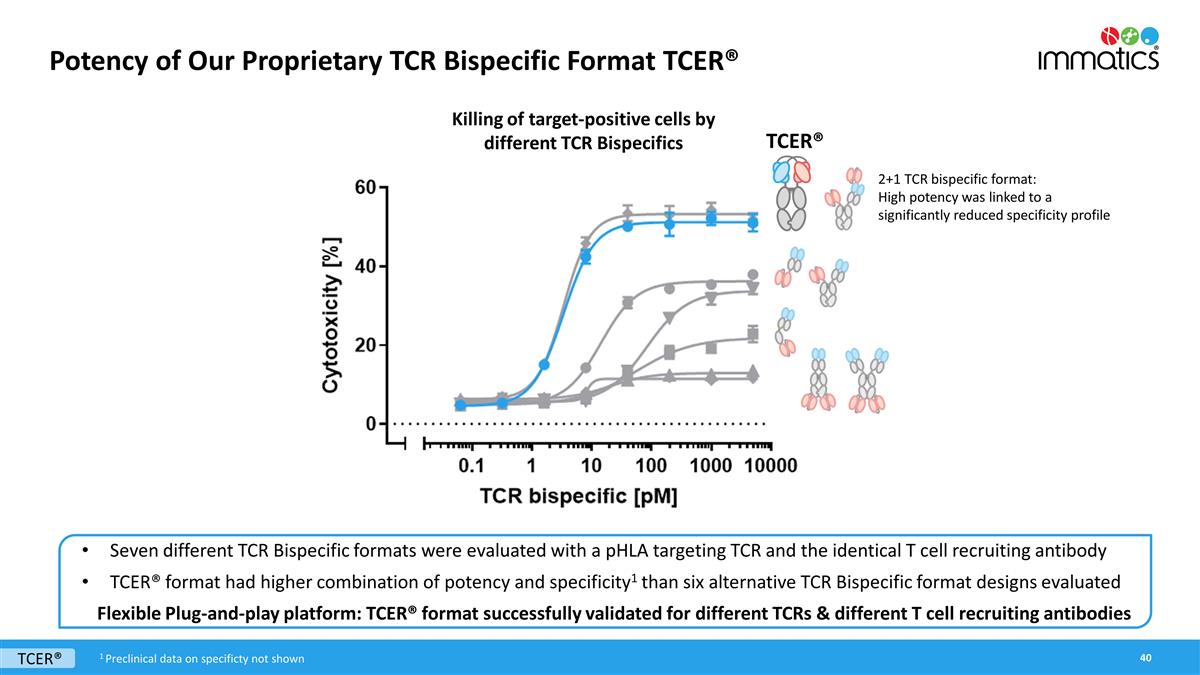
Potency of Our Proprietary TCR
Bispecific Format TCER® Seven different TCR Bispecific formats were evaluated with a pHLA targeting TCR and the identical T cell recruiting antibody TCER® format had higher combination of potency and specificity1 than six alternative TCR
Bispecific format designs evaluated Flexible Plug-and-play platform: TCER® format successfully validated for different TCRs & different T cell recruiting antibodies TCER® TCER® 2+1 TCR bispecific format: High potency was linked to
a significantly reduced specificity profile Killing of target-positive cells by different TCR Bispecifics 1 Preclinical data on specificty not shown
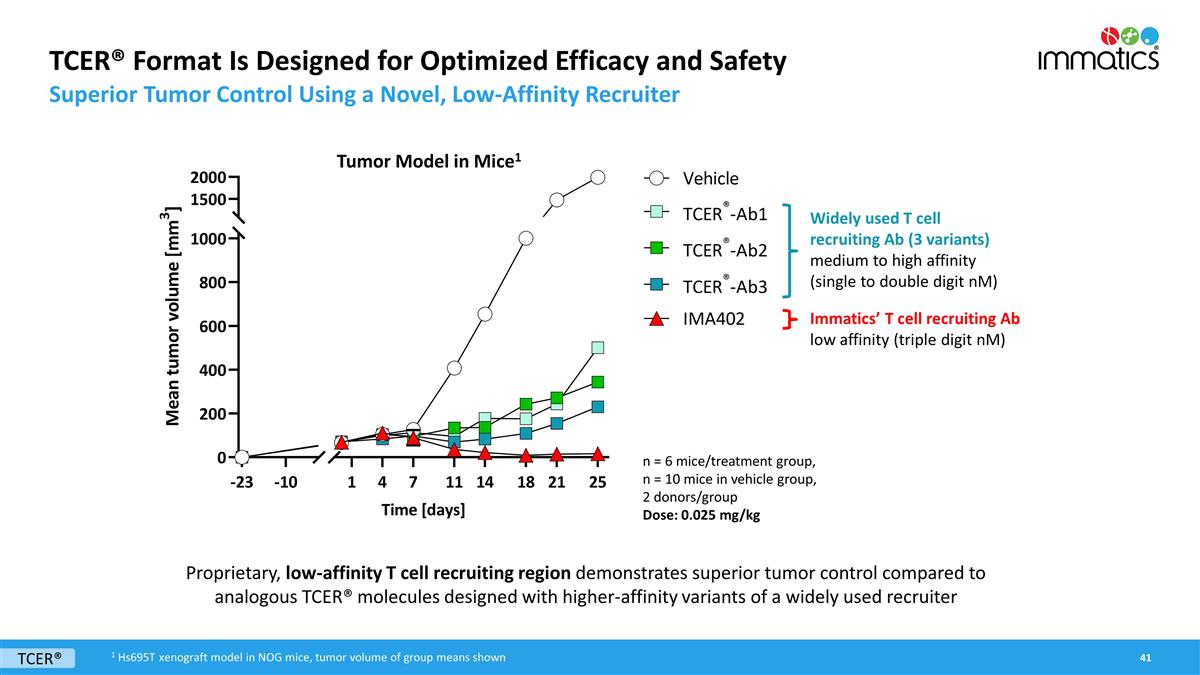
TCER® Format Is Designed for
Optimized Efficacy and Safety Superior Tumor Control Using a Novel, Low-Affinity Recruiter Widely used T cell recruiting Ab (3 variants) medium to high affinity (single to double digit nM) n = 6 mice/treatment group, n = 10 mice in vehicle group, 2
donors/group Dose: 0.025 mg/kg Proprietary, low-affinity T cell recruiting region demonstrates superior tumor control compared to analogous TCER® molecules designed with higher-affinity variants of a widely used recruiter Immatics’ T cell
recruiting Ab low affinity (triple digit nM) TCER® Tumor Model in Mice1 1 Hs695T xenograft model in NOG mice, tumor volume of group means shown
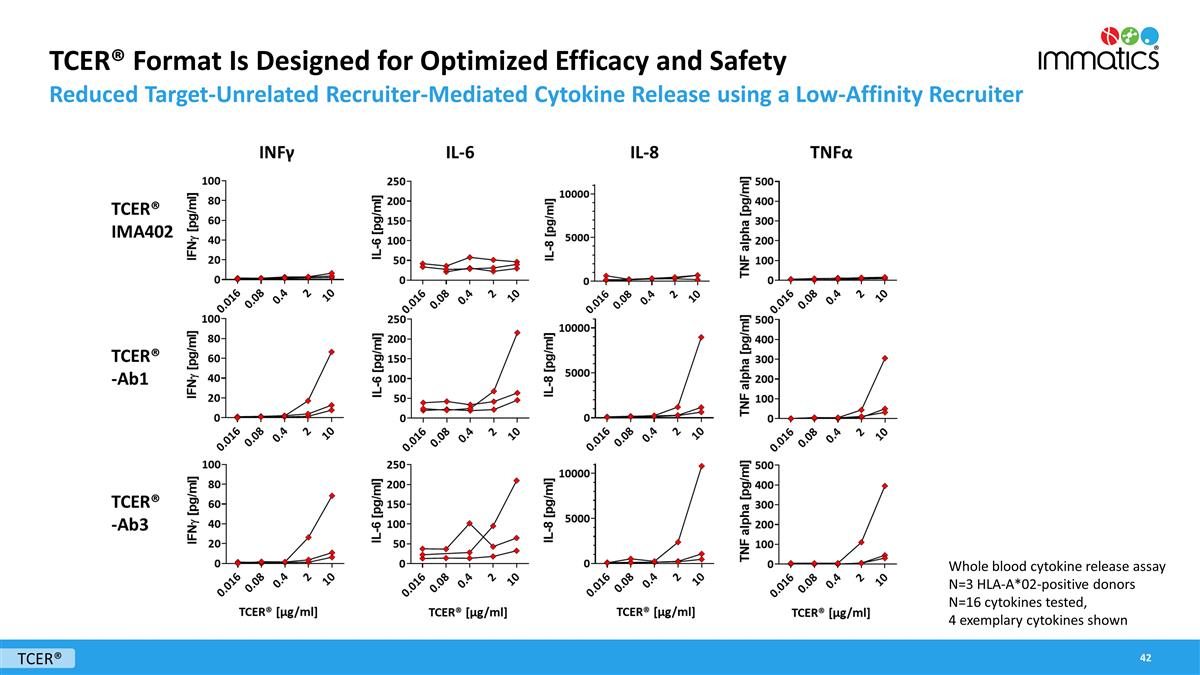
TCER® Format Is Designed for
Optimized Efficacy and Safety Reduced Target-Unrelated Recruiter-Mediated Cytokine Release using a Low-Affinity Recruiter TCER® Whole blood cytokine release assay N=3 HLA-A*02-positive donors N=16 cytokines tested, 4 exemplary cytokines
shown
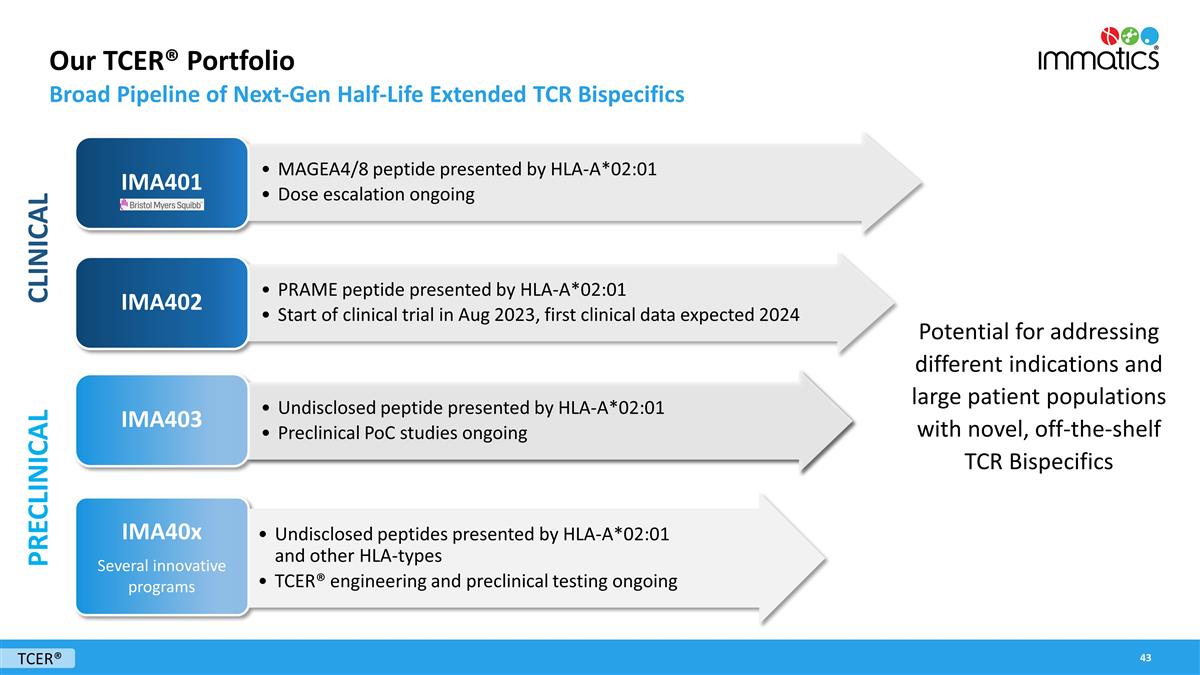
Our TCER® Portfolio Broad
Pipeline of Next-Gen Half-Life Extended TCR Bispecifics TCER® PRAME peptide presented by HLA-A*02:01 Start of clinical trial in Aug 2023, first clinical data expected 2024 IMA402 Undisclosed peptide presented by HLA-A*02:01 Preclinical PoC
studies ongoing IMA403 Potential for addressing different indications and large patient populations with novel, off-the-shelf TCR Bispecifics MAGEA4/8 peptide presented by HLA-A*02:01 Dose escalation ongoing IMA401 Undisclosed peptides presented by
HLA-A*02:01 and other HLA-types TCER® engineering and preclinical testing ongoing IMA40x Several innovative programs CLINICAL PRECLINICAL
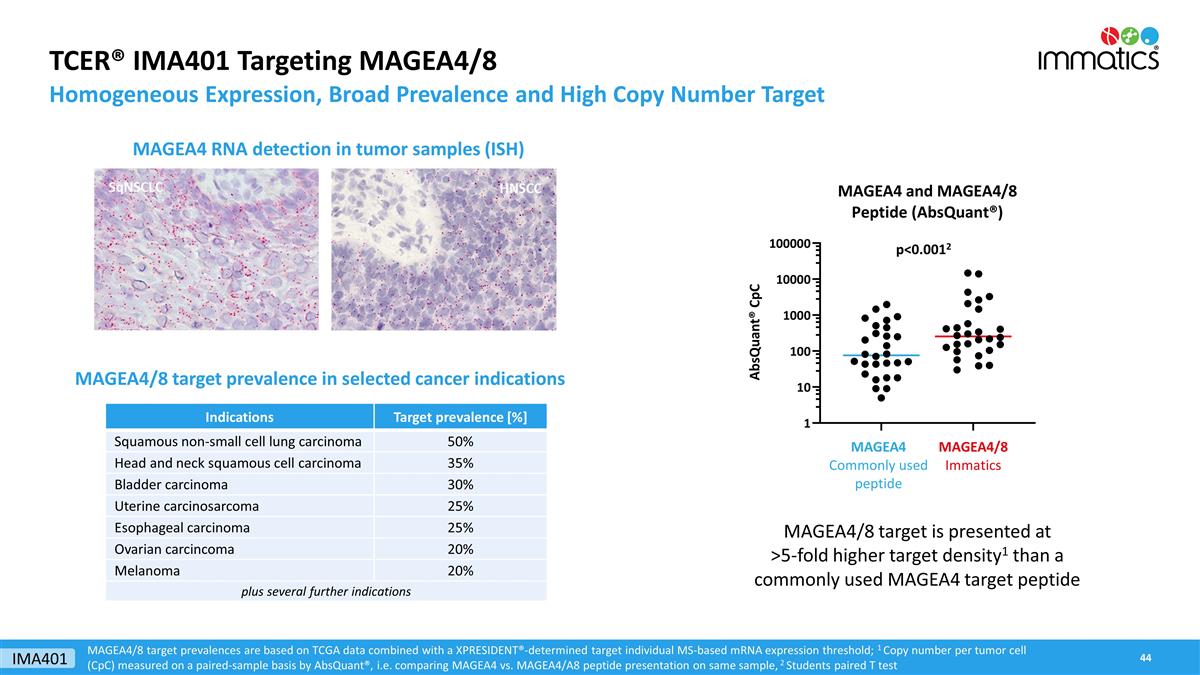
TCER® IMA401 Targeting
MAGEA4/8 Homogeneous Expression, Broad Prevalence and High Copy Number Target MAGEA4 RNA detection in tumor samples (ISH) Indications Target prevalence [%] Squamous non-small cell lung carcinoma 50% Head and neck squamous cell carcinoma 35% Bladder
carcinoma 30% Uterine carcinosarcoma 25% Esophageal carcinoma 25% Ovarian carcincoma 20% Melanoma 20% plus several further indications MAGEA4/8 target prevalence in selected cancer indications MAGEA4/8 target prevalences are based on TCGA data
combined with a XPRESIDENT®-determined target individual MS-based mRNA expression threshold; 1 Copy number per tumor cell (CpC) measured on a paired-sample basis by AbsQuant®, i.e. comparing MAGEA4 vs. MAGEA4/A8 peptide presentation
on same sample, 2 Students paired T test IMA401 p<0.0012 MAGEA4/8 target is presented at >5-fold higher target density1 than a commonly used MAGEA4 target peptide
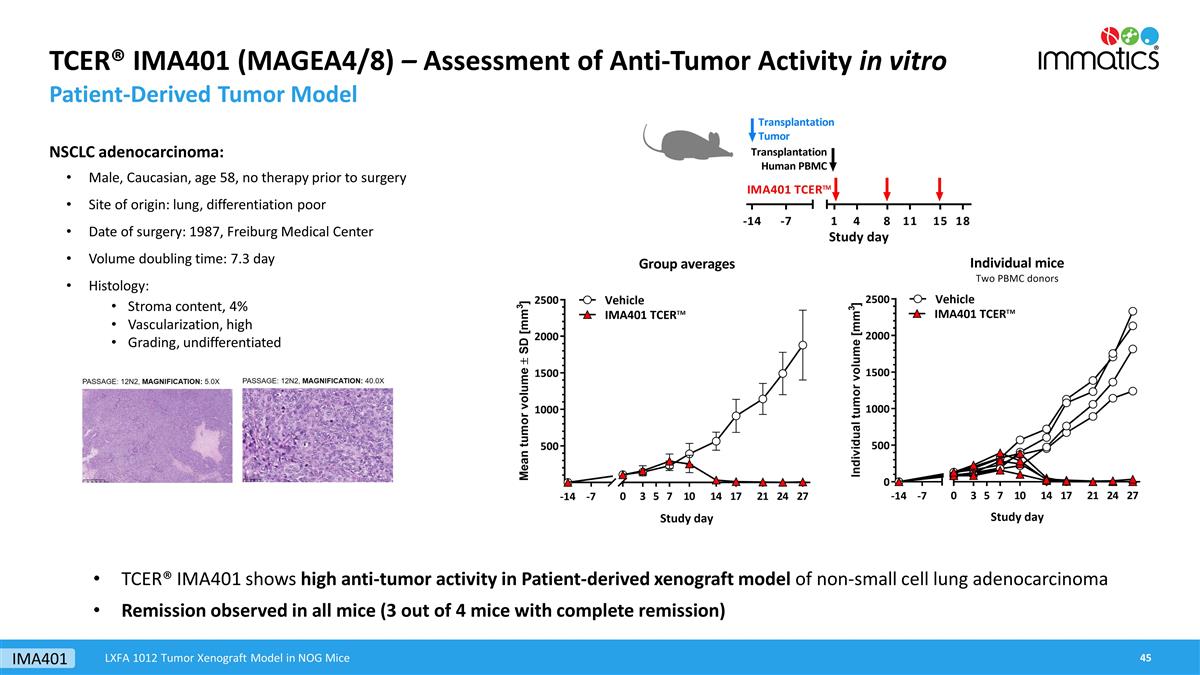
TCER® IMA401 (MAGEA4/8)
– Assessment of Anti-Tumor Activity in vitro Patient-Derived Tumor Model NSCLC adenocarcinoma: Male, Caucasian, age 58, no therapy prior to surgery Site of origin: lung, differentiation poor Date of surgery: 1987, Freiburg Medical Center
Volume doubling time: 7.3 day Histology: Stroma content, 4% Vascularization, high Grading, undifferentiated TCER® IMA401 shows high anti-tumor activity in Patient-derived xenograft model of non-small cell lung adenocarcinoma Remission
observed in all mice (3 out of 4 mice with complete remission) LXFA 1012 Tumor Xenograft Model in NOG Mice IMA401
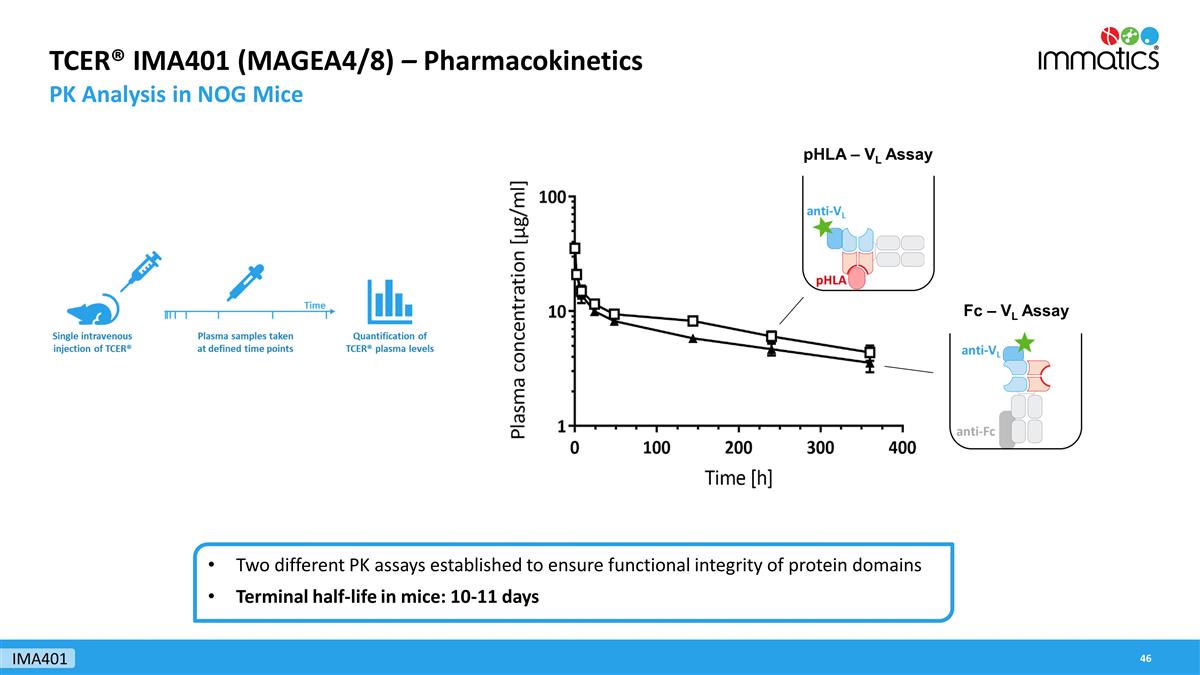
TCER® IMA401 (MAGEA4/8)
– Pharmacokinetics PK Analysis in NOG Mice Two different PK assays established to ensure functional integrity of protein domains Terminal half-life in mice: 10-11 days pHLA – VL Assay Fc – VL Assay IMA401
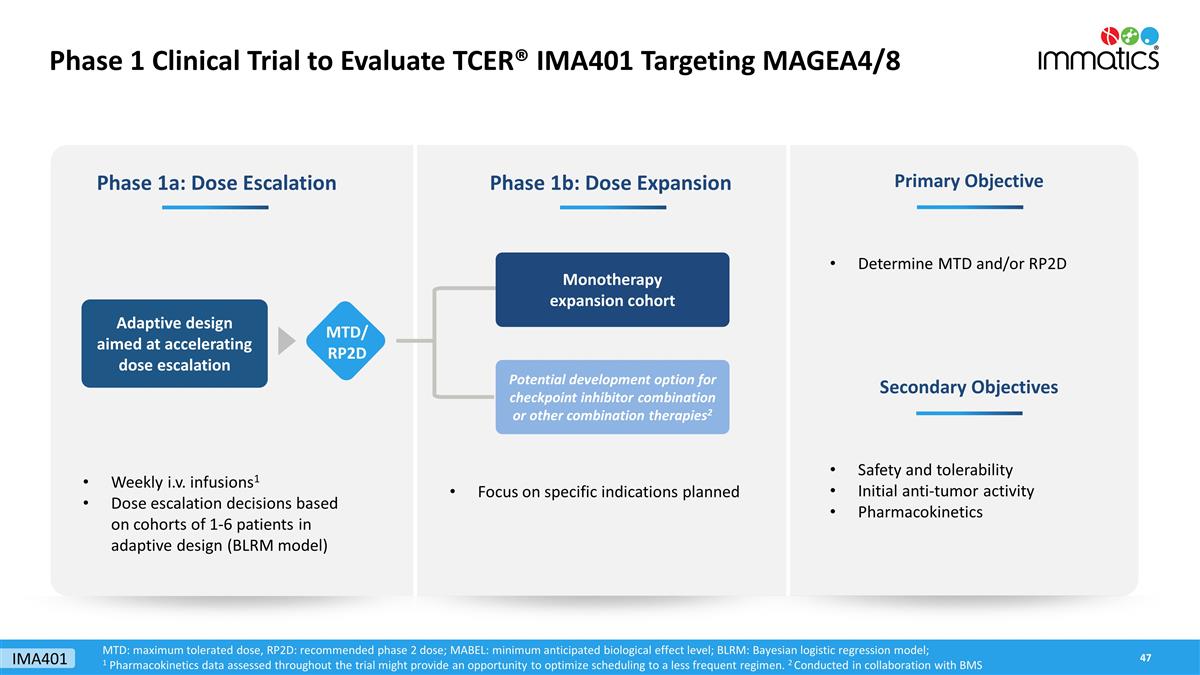
Phase 1 Clinical Trial to Evaluate
TCER® IMA401 Targeting MAGEA4/8 MTD: maximum tolerated dose, RP2D: recommended phase 2 dose; MABEL: minimum anticipated biological effect level; BLRM: Bayesian logistic regression model; 1 Pharmacokinetics data assessed throughout the trial
might provide an opportunity to optimize scheduling to a less frequent regimen. 2 Conducted in collaboration with BMS Phase 1a: Dose Escalation Phase 1b: Dose Expansion Weekly i.v. infusions1 Dose escalation decisions based on cohorts of 1-6
patients in adaptive design (BLRM model) MTD/ RP2D Adaptive design aimed at accelerating dose escalation Focus on specific indications planned Potential development option for checkpoint inhibitor combination or other combination therapies2
Monotherapy expansion cohort Primary Objective Determine MTD and/or RP2D Secondary Objectives Safety and tolerability Initial anti-tumor activity Pharmacokinetics IMA401
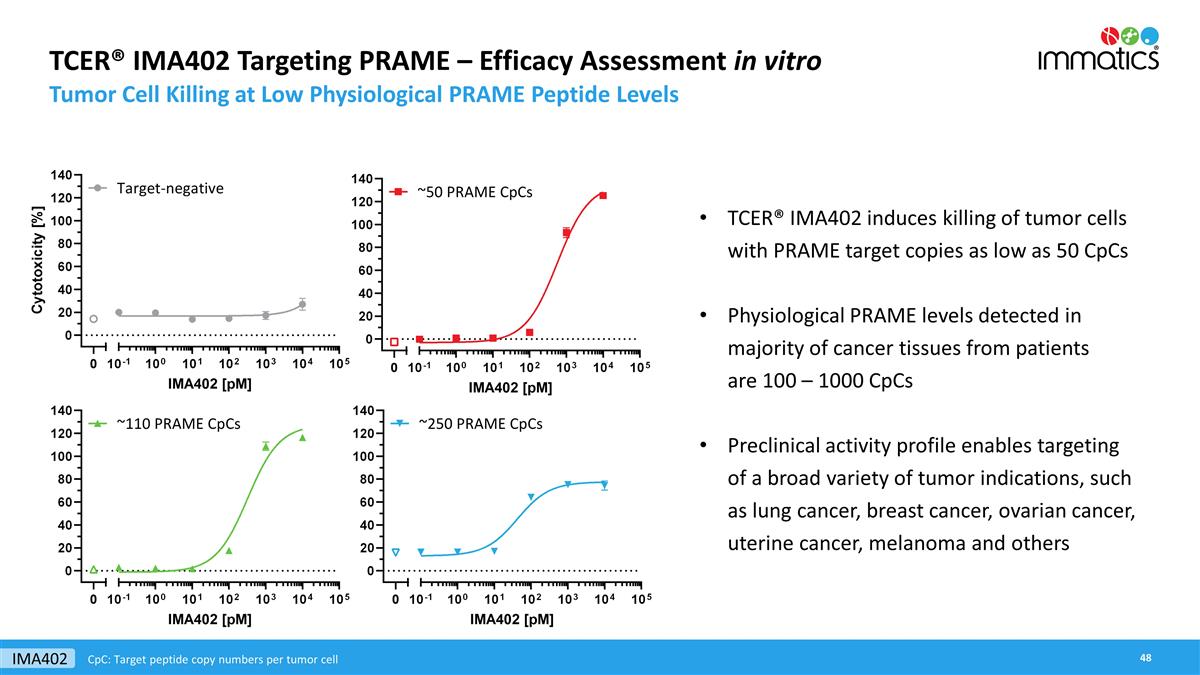
TCER® IMA402 Targeting PRAME
– Efficacy Assessment in vitro Tumor Cell Killing at Low Physiological PRAME Peptide Levels TCER® IMA402 induces killing of tumor cells with PRAME target copies as low as 50 CpCs Physiological PRAME levels detected in majority of cancer
tissues from patients are 100 – 1000 CpCs Preclinical activity profile enables targeting of a broad variety of tumor indications, such as lung cancer, breast cancer, ovarian cancer, uterine cancer, melanoma and others IMA402 CpC: Target
peptide copy numbers per tumor cell
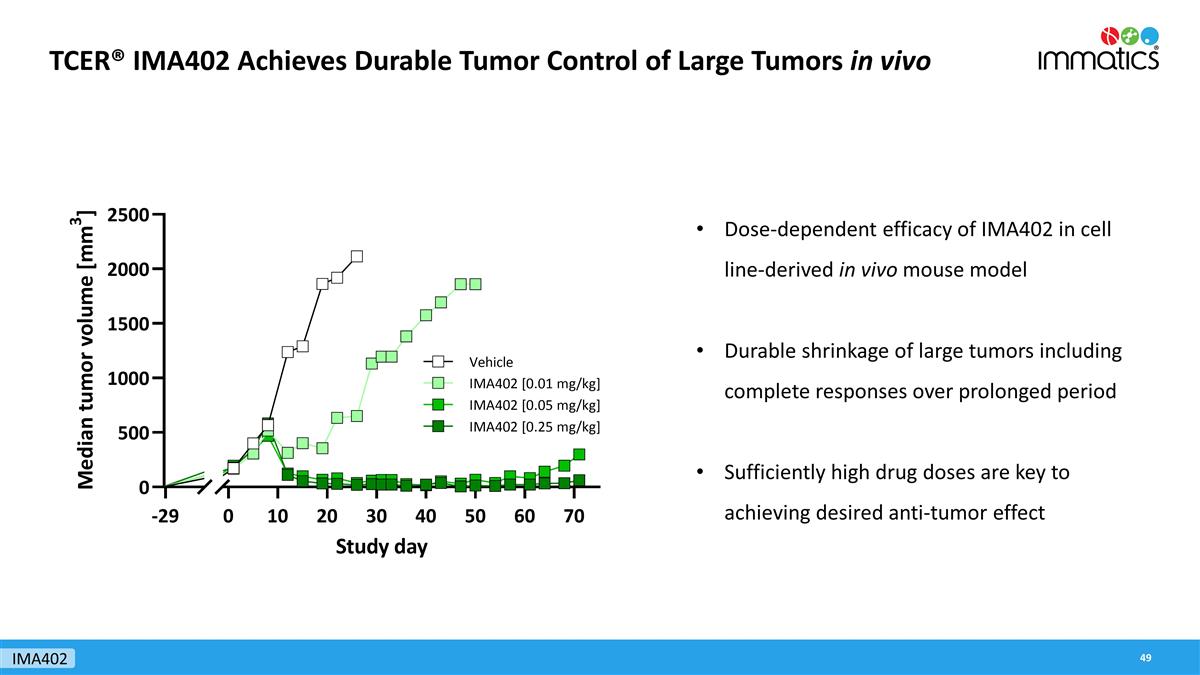
TCER® IMA402 Achieves Durable
Tumor Control of Large Tumors in vivo Dose-dependent efficacy of IMA402 in cell line-derived in vivo mouse model Durable shrinkage of large tumors including complete responses over prolonged period Sufficiently high drug doses are key to achieving
desired anti-tumor effect IMA402
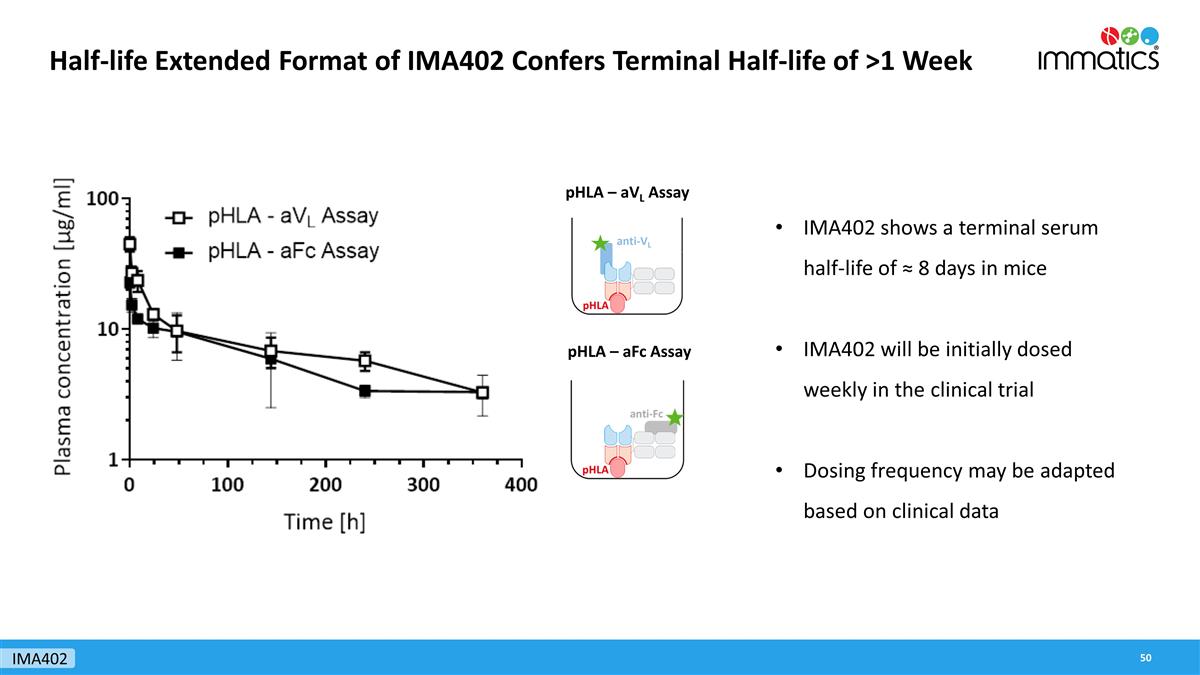
Half-life Extended Format of IMA402
Confers Terminal Half-life of >1 Week pHLA – aVL Assay pHLA – aFc Assay IMA402 shows a terminal serum half-life of ≈ 8 days in mice IMA402 will be initially dosed weekly in the clinical trial Dosing frequency may be adapted
based on clinical data IMA402
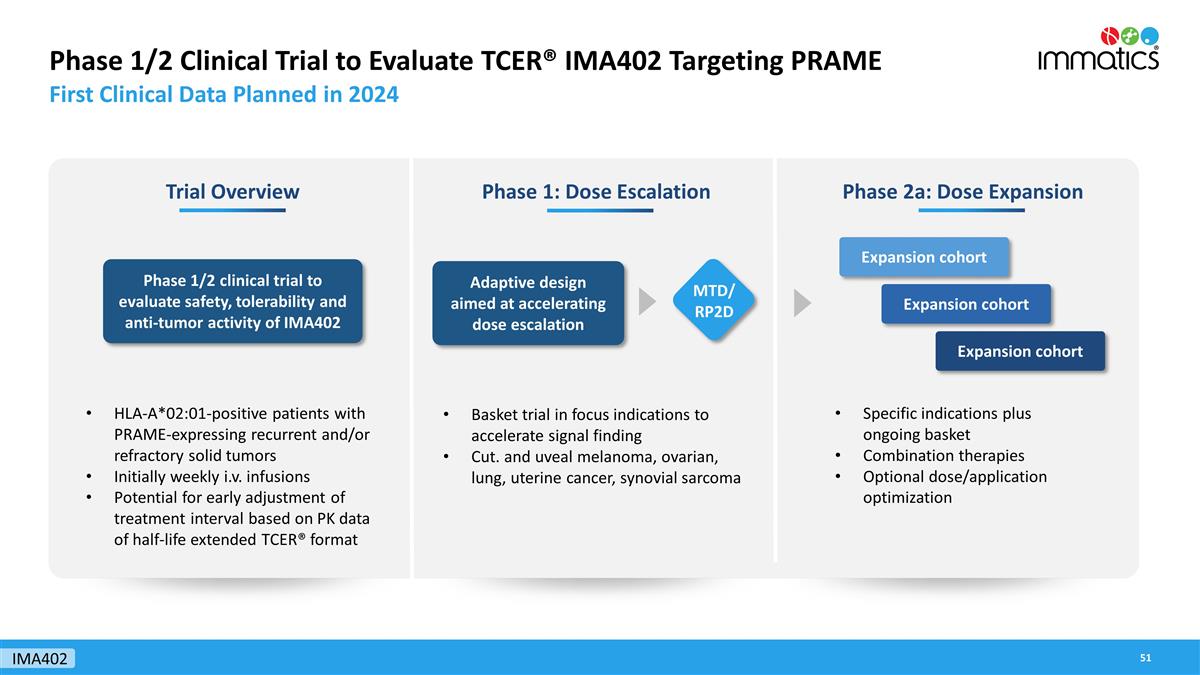
Phase 1/2 Clinical Trial to
Evaluate TCER® IMA402 Targeting PRAME First Clinical Data Planned in 2024 Phase 1: Dose Escalation Phase 2a: Dose Expansion Adaptive design aimed at accelerating dose escalation Specific indications plus ongoing basket Combination therapies
Optional dose/application optimization Expansion cohort Expansion cohort Expansion cohort Trial Overview Phase 1/2 clinical trial to evaluate safety, tolerability and anti-tumor activity of IMA402 HLA-A*02:01-positive patients with PRAME-expressing
recurrent and/or refractory solid tumors Initially weekly i.v. infusions Potential for early adjustment of treatment interval based on PK data of half-life extended TCER® format MTD/ RP2D IMA402 Basket trial in focus indications to accelerate
signal finding Cut. and uveal melanoma, ovarian, lung, uterine cancer, synovial sarcoma

Immatics’ Proprietary Target
and TCR Discovery Platforms
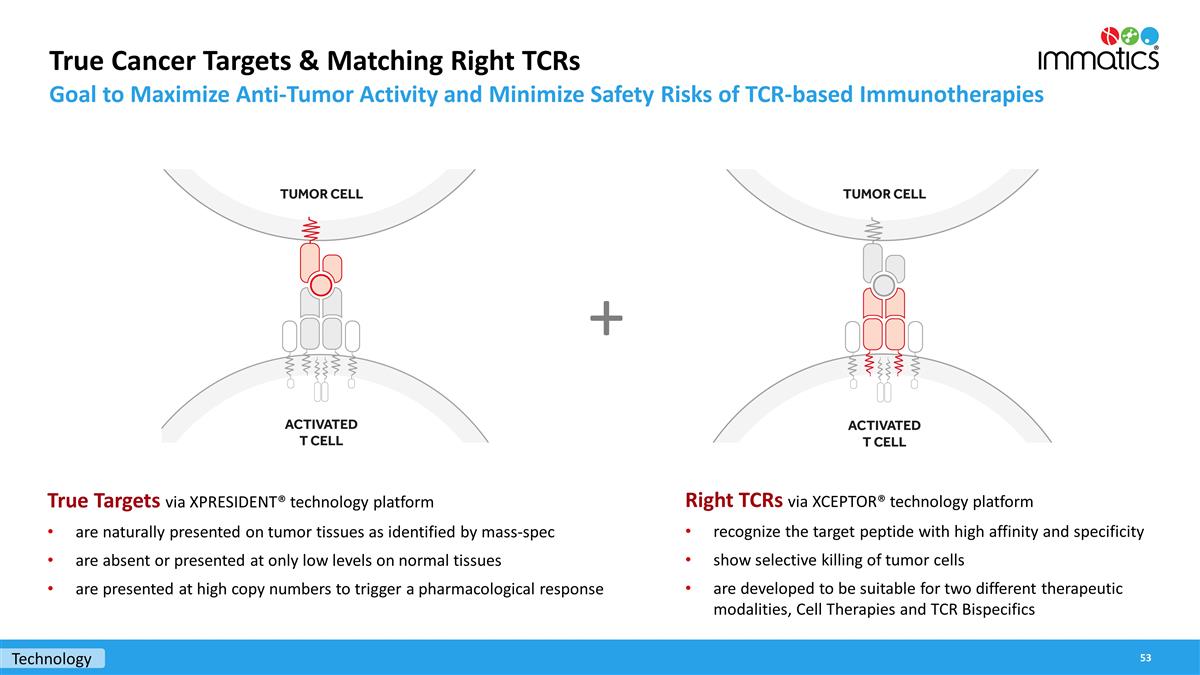
True Cancer Targets & Matching
Right TCRs Goal to Maximize Anti-Tumor Activity and Minimize Safety Risks of TCR-based Immunotherapies True Targets via XPRESIDENT® technology platform are naturally presented on tumor tissues as identified by mass-spec are absent or presented
at only low levels on normal tissues are presented at high copy numbers to trigger a pharmacological response + Technology Right TCRs via XCEPTOR® technology platform recognize the target peptide with high affinity and specificity show
selective killing of tumor cells are developed to be suitable for two different therapeutic modalities, Cell Therapies and TCR Bispecifics
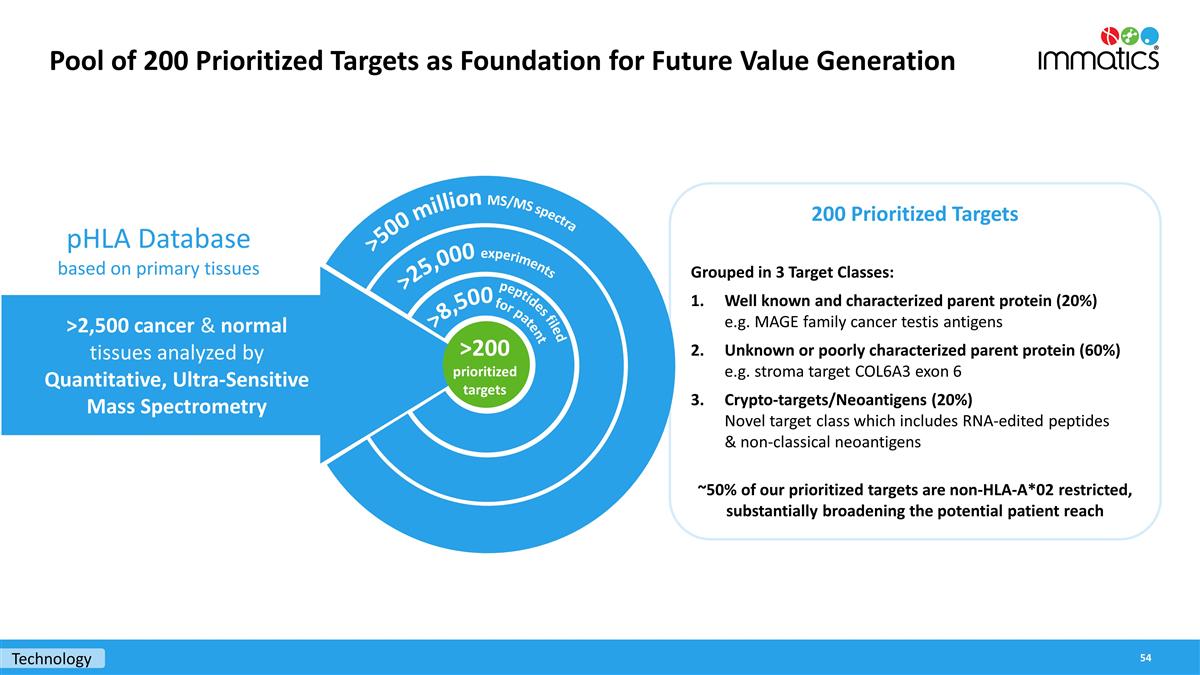
Technology Pool of 200 Prioritized
Targets as Foundation for Future Value Generation 200 Prioritized Targets Grouped in 3 Target Classes: Well known and characterized parent protein (20%) e.g. MAGE family cancer testis antigens Unknown or poorly characterized parent protein (60%)
e.g. stroma target COL6A3 exon 6 Crypto-targets/Neoantigens (20%) Novel target class which includes RNA-edited peptides & non-classical neoantigens ~50% of our prioritized targets are non-HLA-A*02 restricted, substantially broadening the
potential patient reach >500 million MS/MS spectra >25,000 experiments >8,500 peptides filed for patent >2,500 cancer & normal tissues analyzed by Quantitative, Ultra-Sensitive Mass Spectrometry pHLA Database based on primary tissues
>200 prioritized targets
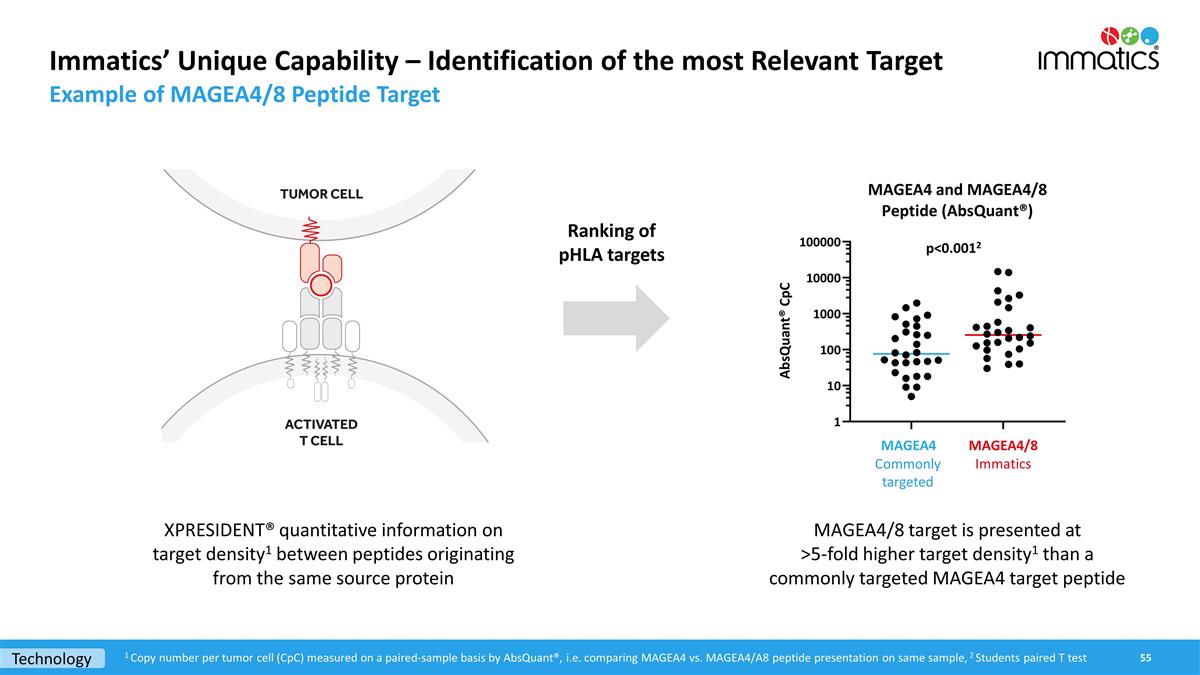
Immatics’ Unique Capability
– Identification of the most Relevant Target Example of MAGEA4/8 Peptide Target 1 Copy number per tumor cell (CpC) measured on a paired-sample basis by AbsQuant®, i.e. comparing MAGEA4 vs. MAGEA4/A8 peptide presentation on same sample, 2
Students paired T test p<0.0012 Technology MAGEA4/8 target is presented at >5-fold higher target density1 than a commonly targeted MAGEA4 target peptide XPRESIDENT® quantitative information on target density1 between peptides originating
from the same source protein Ranking of pHLA targets Commonly targeted
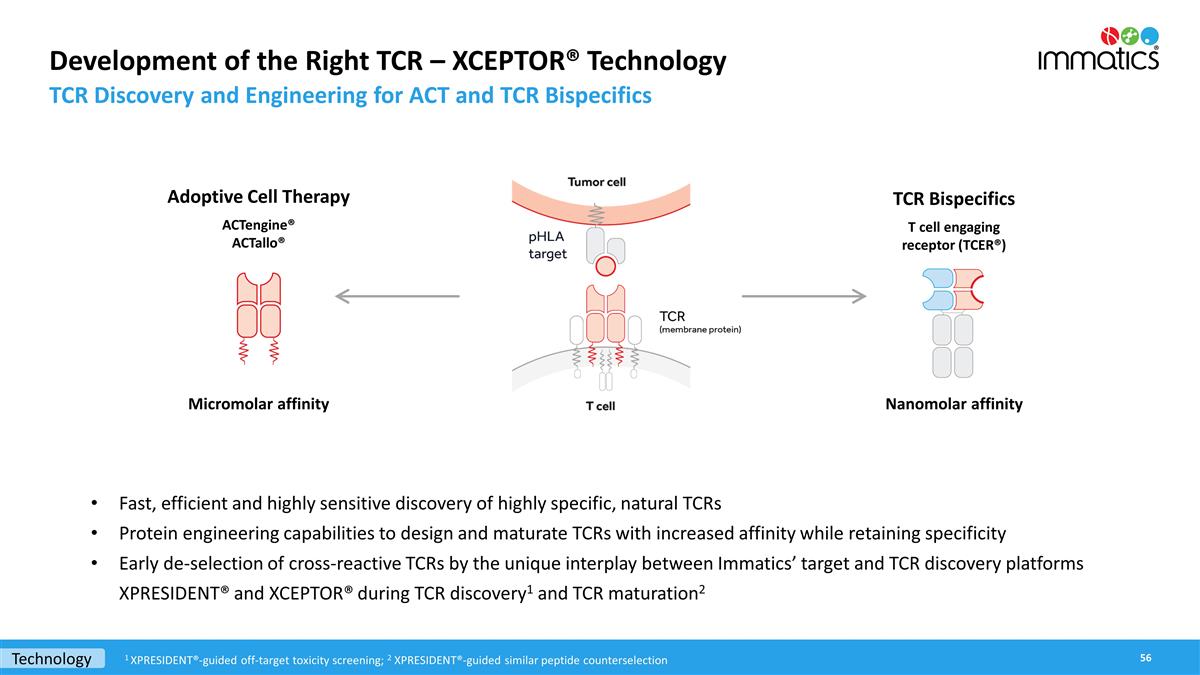
Development of the Right TCR
– XCEPTOR® Technology TCR Discovery and Engineering for ACT and TCR Bispecifics TCR Bispecifics T cell engaging receptor (TCER®) Adoptive Cell Therapy ACTengine® ACTallo® Fast, efficient and highly sensitive discovery of
highly specific, natural TCRs Protein engineering capabilities to design and maturate TCRs with increased affinity while retaining specificity Early de-selection of cross-reactive TCRs by the unique interplay between Immatics’ target and TCR
discovery platforms XPRESIDENT® and XCEPTOR® during TCR discovery1 and TCR maturation2 Micromolar affinity Nanomolar affinity Technology 1 XPRESIDENT®-guided off-target toxicity screening; 2 XPRESIDENT®-guided similar peptide
counterselection
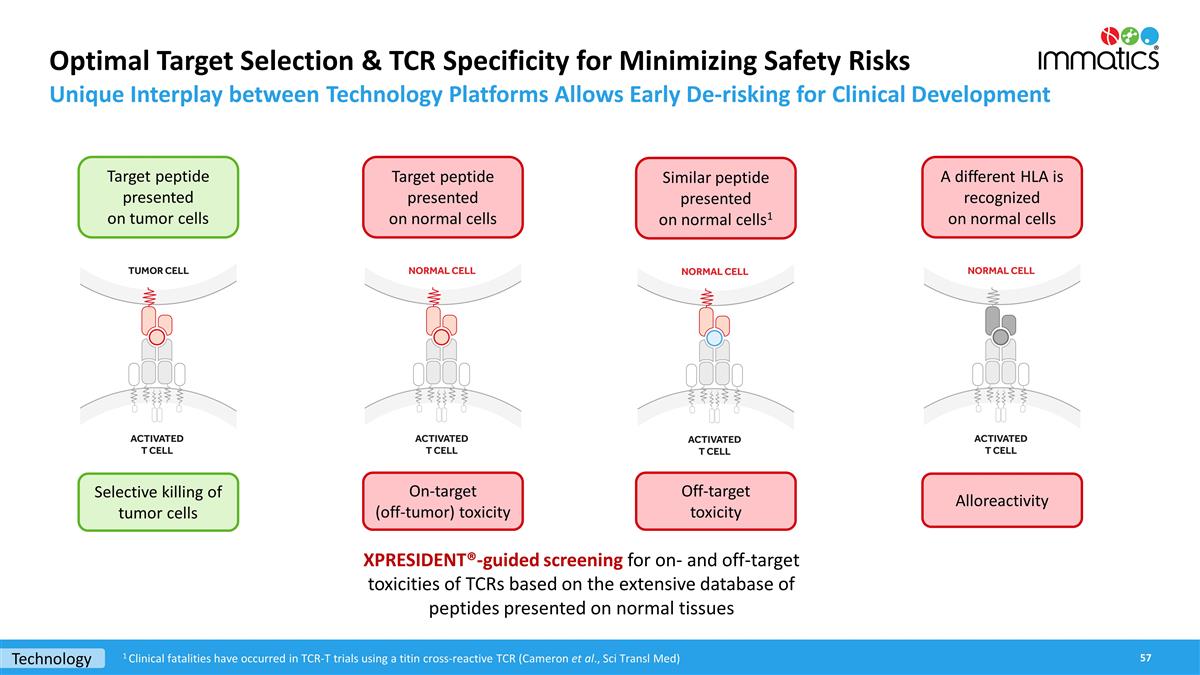
Optimal Target Selection & TCR
Specificity for Minimizing Safety Risks Unique Interplay between Technology Platforms Allows Early De-risking for Clinical Development Target peptide presented on tumor cells Selective killing of tumor cells Target peptide presented on normal cells
Off-target toxicity On-target (off-tumor) toxicity A different HLA is recognized on normal cells Alloreactivity Similar peptide presented on normal cells1 XPRESIDENT®-guided screening for on- and off-target toxicities of TCRs based on the
extensive database of peptides presented on normal tissues Technology 1 Clinical fatalities have occurred in TCR-T trials using a titin cross-reactive TCR (Cameron et al., Sci Transl Med)
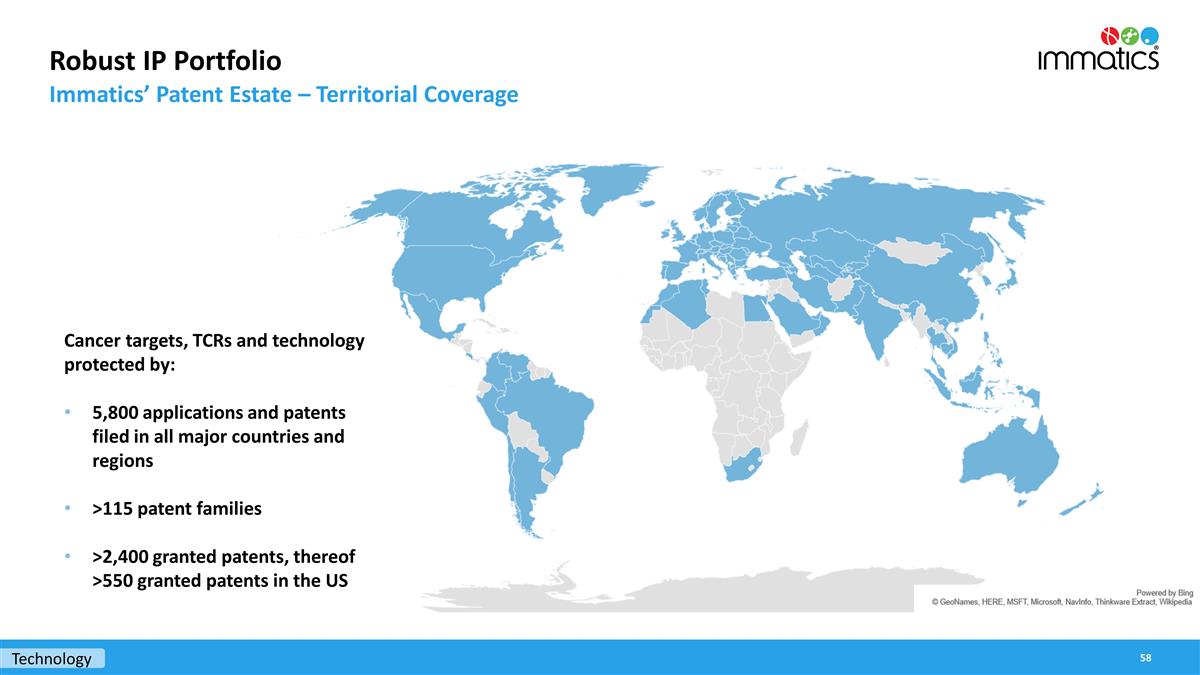
Robust IP Portfolio Immatics’
Patent Estate – Territorial Coverage Cancer targets, TCRs and technology protected by: 5,800 applications and patents filed in all major countries and regions >115 patent families >2,400 granted patents, thereof >550
granted patents in the US Technology

Corporate Information &
Milestones

David Leitner Schuldirektor David
Leitner Schuldirektor David Leitner Schuldirektor Harpreet Singh Chief Executive Officer Co-Founder >20 yrs biotech experience Arnd Christ Chief Financial Officer >20 yrs biotech experience (InflaRx, Medigene, NovImmune,
Probiodrug) Carsten Reinhardt Chief Development Officer >20 yrs pharma & biotech experience (Micromet, Roche, Fresenius) Cedrik Britten Chief Medical Officer 15 yrs pharma & biotech experience (GSK, BioNTech) Rainer
Kramer Chief Business Officer 25 yrs pharma & biotech experience (Amgen, MorphoSys, Jerini, Shire, Signature Dx) Steffen Walter Chief Operating Officer Co-Founder Immatics US >15 yrs biotech experience Edward Sturchio General Counsel >15
yrs pharma & biotech experience (Abeona Therapeutics, AAA, Novartis, Merck, Schering) ) Jordan Silverstein Head of Strategy >10 yrs biotech experience (InflaRx, AAA) Toni Weinschenk Chief Innovation Officer Co-Founder >15 yrs
biotech experience Experienced Global Leadership Team Across Europe and the US Corporate
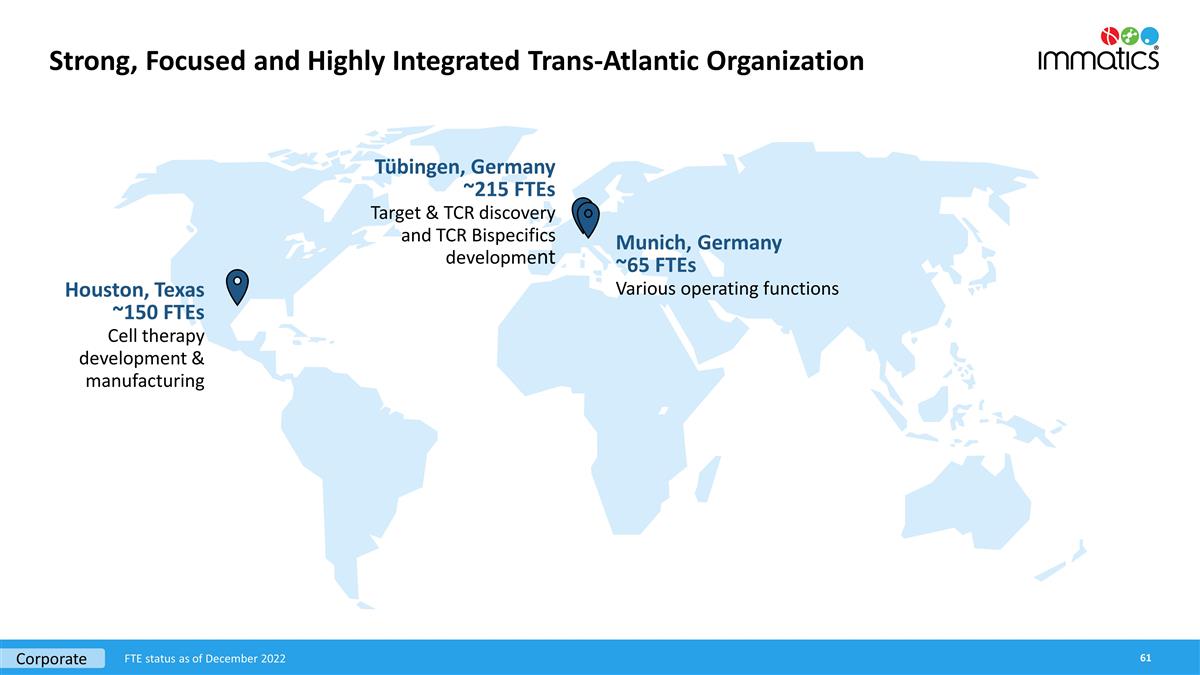
Strong, Focused and Highly
Integrated Trans-Atlantic Organization Houston, Texas ~150 FTEs Cell therapy development & manufacturing Munich, Germany ~65 FTEs Various operating functions Tübingen, Germany ~215 FTEs Target & TCR discovery and TCR Bispecifics
development Corporate FTE status as of December 2022

Appendix
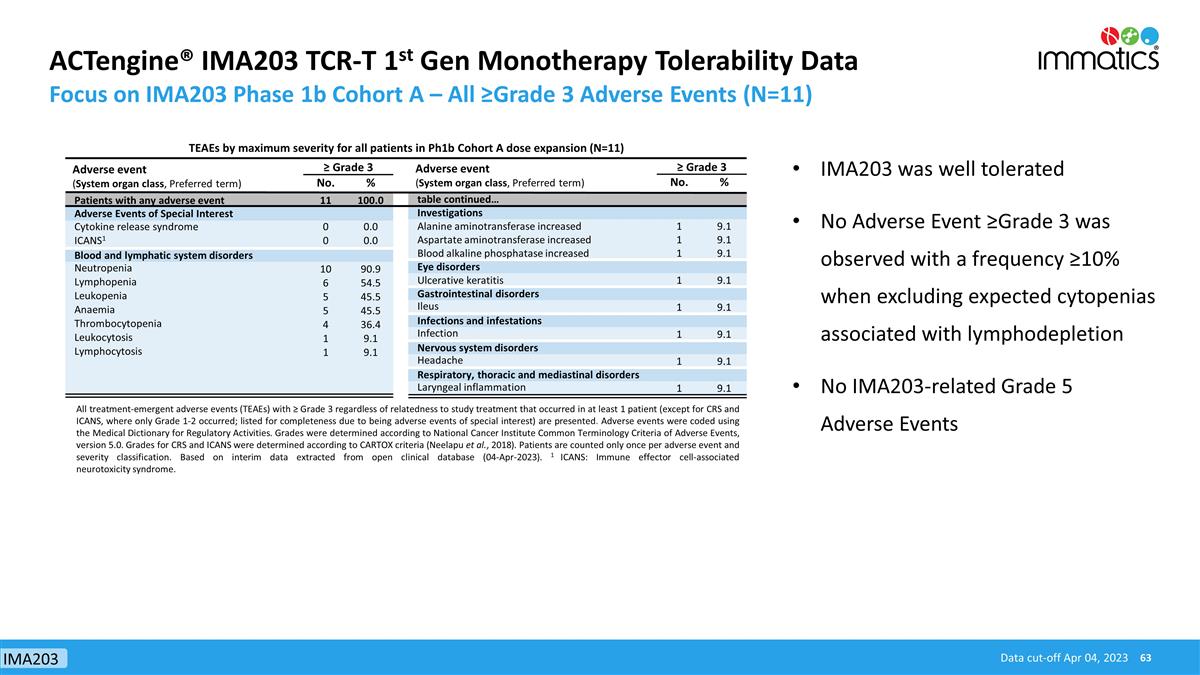
ACTengine® IMA203 TCR-T 1st
Gen Monotherapy Tolerability Data Focus on IMA203 Phase 1b Cohort A – All ≥Grade 3 Adverse Events (N=11) IMA203 was well tolerated No Adverse Event ≥Grade 3 was observed with a frequency ≥10% when excluding expected
cytopenias associated with lymphodepletion No IMA203-related Grade 5 Adverse Events All treatment-emergent adverse events (TEAEs) with ≥ Grade 3 regardless of relatedness to study treatment that occurred in at least 1 patient (except for CRS
and ICANS, where only Grade 1-2 occurred; listed for completeness due to being adverse events of special interest) are presented. Adverse events were coded using the Medical Dictionary for Regulatory Activities. Grades were determined according
to National Cancer Institute Common Terminology Criteria of Adverse Events, version 5.0. Grades for CRS and ICANS were determined according to CARTOX criteria (Neelapu et al., 2018). Patients are counted only once per adverse event and
severity classification. Based on interim data extracted from open clinical database (04-Apr-2023). 1 ICANS: Immune effector cell-associated neurotoxicity syndrome. Data cut-off Apr 04, 2023 Adverse event (System organ class, Preferred
term) ≥ Grade 3 No. % Patients with any adverse event 11 100.0 Adverse Events of Special Interest Cytokine release syndrome 0 0.0 ICANS1 0 0.0 Blood and lymphatic system disorders Neutropenia 10 90.9 Lymphopenia 6 54.5 Leukopenia 5 45.5
Anaemia 5 45.5 Thrombocytopenia 4 36.4 Leukocytosis 1 9.1 Lymphocytosis 1 9.1 Adverse event (System organ class, Preferred term) ≥ Grade 3 No. % table continued… Investigations Alanine aminotransferase increased 1 9.1 Aspartate
aminotransferase increased 1 9.1 Blood alkaline phosphatase increased 1 9.1 Eye disorders Ulcerative keratitis 1 9.1 Gastrointestinal disorders Ileus 1 9.1 Infections and infestations Infection 1 9.1 Nervous system disorders Headache 1 9.1
Respiratory, thoracic and mediastinal disorders Laryngeal inflammation 1 9.1 TEAEs by maximum severity for all patients in Ph1b Cohort A dose expansion (N=11) IMA203
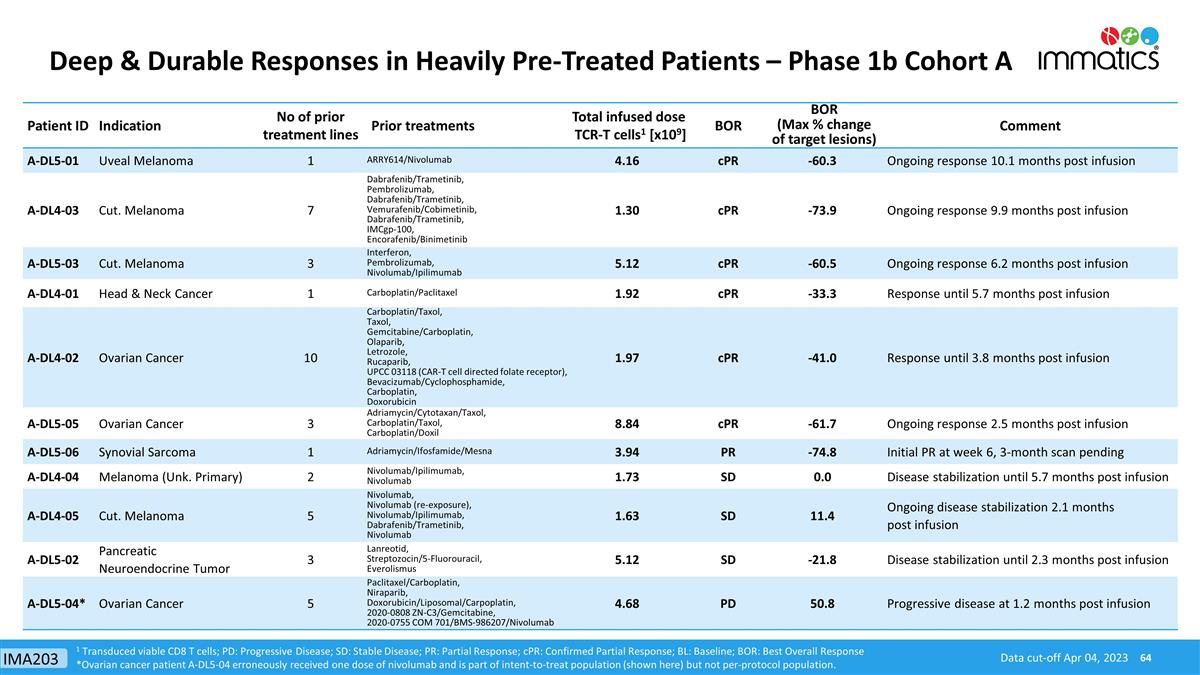
Deep & Durable Responses in
Heavily Pre-Treated Patients – Phase 1b Cohort A Patient ID Indication No of prior treatment lines Prior treatments Total infused dose TCR-T cells1 [x109] BOR BOR (Max % change of target lesions) Comment A-DL5-01 Uveal Melanoma 1
ARRY614/Nivolumab 4.16 cPR -60.3 Ongoing response 10.1 months post infusion A-DL4-03 Cut. Melanoma 7 Dabrafenib/Trametinib, Pembrolizumab, Dabrafenib/Trametinib, Vemurafenib/Cobimetinib, Dabrafenib/Trametinib, IMCgp-100,
Encorafenib/Binimetinib 1.30 cPR -73.9 Ongoing response 9.9 months post infusion A-DL5-03 Cut. Melanoma 3 Interferon, Pembrolizumab, Nivolumab/Ipilimumab 5.12 cPR -60.5 Ongoing response 6.2 months post infusion A-DL4-01
Head & Neck Cancer 1 Carboplatin/Paclitaxel 1.92 cPR -33.3 Response until 5.7 months post infusion A-DL4-02 Ovarian Cancer 10 Carboplatin/Taxol, Taxol, Gemcitabine/Carboplatin, Olaparib, Letrozole, Rucaparib, UPCC 03118 (CAR-T cell
directed folate receptor), Bevacizumab/Cyclophosphamide, Carboplatin, Doxorubicin 1.97 cPR -41.0 Response until 3.8 months post infusion A-DL5-05 Ovarian Cancer 3 Adriamycin/Cytotaxan/Taxol, Carboplatin/Taxol, Carboplatin/Doxil
8.84 cPR -61.7 Ongoing response 2.5 months post infusion A-DL5-06 Synovial Sarcoma 1 Adriamycin/Ifosfamide/Mesna 3.94 PR -74.8 Initial PR at week 6, 3-month scan pending A-DL4-04 Melanoma (Unk. Primary) 2
Nivolumab/Ipilimumab, Nivolumab 1.73 SD 0.0 Disease stabilization until 5.7 months post infusion A-DL4-05 Cut. Melanoma 5 Nivolumab, Nivolumab (re-exposure), Nivolumab/Ipilimumab, Dabrafenib/Trametinib, Nivolumab 1.63 SD 11.4
Ongoing disease stabilization 2.1 months post infusion A-DL5-02 Pancreatic Neuroendocrine Tumor 3 Lanreotid, Streptozocin/5-Fluorouracil, Everolismus 5.12 SD -21.8 Disease stabilization until 2.3 months post infusion A-DL5-04*
Ovarian Cancer 5 Paclitaxel/Carboplatin, Niraparib, Doxorubicin/Liposomal/Carpoplatin, 2020-0808 ZN-C3/Gemcitabine, 2020-0755 COM 701/BMS-986207/Nivolumab 4.68 PD 50.8 Progressive disease at 1.2 months post infusion 1
Transduced viable CD8 T cells; PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR: Confirmed Partial Response; BL: Baseline; BOR: Best Overall Response *Ovarian cancer patient A-DL5-04 erroneously received one dose of
nivolumab and is part of intent-to-treat population (shown here) but not per-protocol population. Data cut-off Apr 04, 2023 IMA203
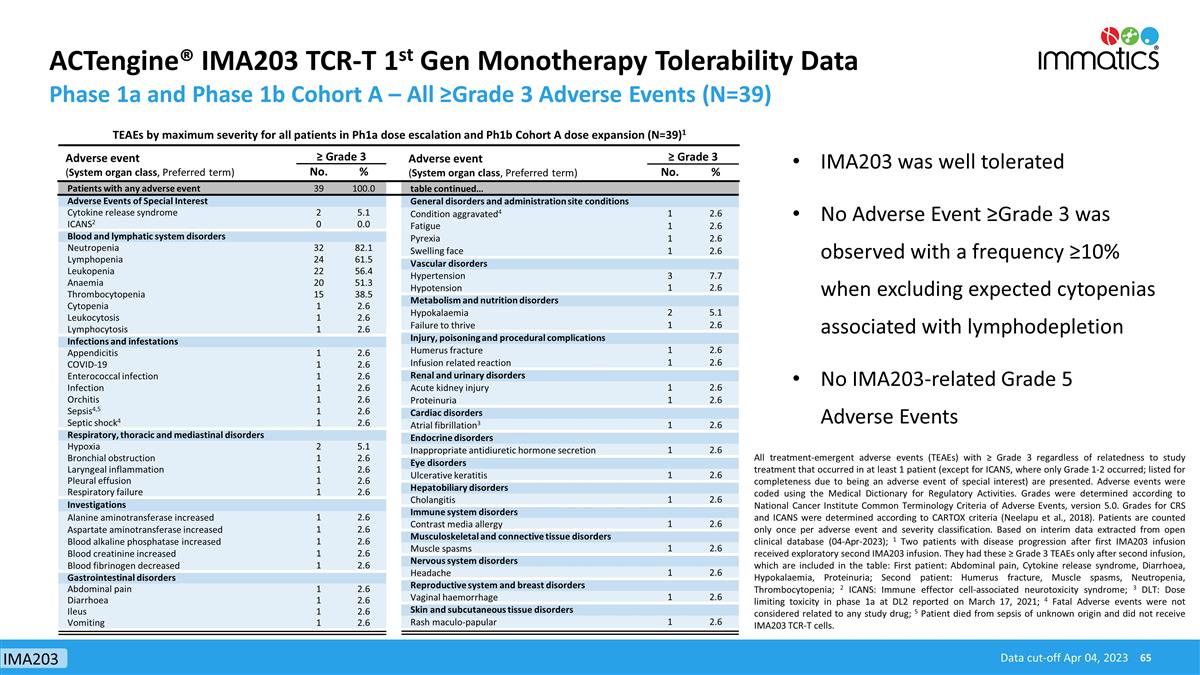
ACTengine® IMA203 TCR-T 1st
Gen Monotherapy Tolerability Data Phase 1a and Phase 1b Cohort A – All ≥Grade 3 Adverse Events (N=39) IMA203 was well tolerated No Adverse Event ≥Grade 3 was observed with a frequency ≥10% when excluding expected cytopenias
associated with lymphodepletion No IMA203-related Grade 5 Adverse Events All treatment-emergent adverse events (TEAEs) with ≥ Grade 3 regardless of relatedness to study treatment that occurred in at least 1 patient (except for ICANS, where
only Grade 1-2 occurred; listed for completeness due to being an adverse event of special interest) are presented. Adverse events were coded using the Medical Dictionary for Regulatory Activities. Grades were determined according to National Cancer
Institute Common Terminology Criteria of Adverse Events, version 5.0. Grades for CRS and ICANS were determined according to CARTOX criteria (Neelapu et al., 2018). Patients are counted only once per adverse event and severity
classification. Based on interim data extracted from open clinical database (04-Apr-2023); 1 Two patients with disease progression after first IMA203 infusion received exploratory second IMA203 infusion. They had these ≥ Grade 3 TEAEs only
after second infusion, which are included in the table: First patient: Abdominal pain, Cytokine release syndrome, Diarrhoea, Hypokalaemia, Proteinuria; Second patient: Humerus fracture, Muscle spasms, Neutropenia, Thrombocytopenia; 2 ICANS:
Immune effector cell-associated neurotoxicity syndrome; 3 DLT: Dose limiting toxicity in phase 1a at DL2 reported on March 17, 2021; 4 Fatal Adverse events were not considered related to any study drug; 5 Patient died from sepsis of unknown
origin and did not receive IMA203 TCR-T cells. Data cut-off Apr 04, 2023 Adverse event (System organ class, Preferred term) ≥ Grade 3 No. % Patients with any adverse event 39 100.0 Adverse Events of Special Interest Cytokine release syndrome 2
5.1 ICANS2 0 0.0 Blood and lymphatic system disorders Neutropenia 32 82.1 Lymphopenia 24 61.5 Leukopenia 22 56.4 Anaemia 20 51.3 Thrombocytopenia 15 38.5 Cytopenia 1 2.6 Leukocytosis 1 2.6 Lymphocytosis 1 2.6 Infections and infestations Appendicitis
1 2.6 COVID-19 1 2.6 Enterococcal infection 1 2.6 Infection 1 2.6 Orchitis 1 2.6 Sepsis4,5 1 2.6 Septic shock4 1 2.6 Respiratory, thoracic and mediastinal disorders Hypoxia 2 5.1 Bronchial obstruction 1 2.6 Laryngeal inflammation 1 2.6 Pleural
effusion 1 2.6 Respiratory failure 1 2.6 Investigations Alanine aminotransferase increased 1 2.6 Aspartate aminotransferase increased 1 2.6 Blood alkaline phosphatase increased 1 2.6 Blood creatinine increased 1 2.6 Blood fibrinogen decreased 1 2.6
Gastrointestinal disorders Abdominal pain 1 2.6 Diarrhoea 1 2.6 Ileus 1 2.6 Vomiting 1 2.6 Adverse event (System organ class, Preferred term) ≥ Grade 3 No. % table continued… General disorders and administration site conditions
Condition aggravated4 1 2.6 Fatigue 1 2.6 Pyrexia 1 2.6 Swelling face 1 2.6 Vascular disorders Hypertension 3 7.7 Hypotension 1 2.6 Metabolism and nutrition disorders Hypokalaemia 2 5.1 Failure to thrive 1 2.6 Injury, poisoning and procedural
complications Humerus fracture 1 2.6 Infusion related reaction 1 2.6 Renal and urinary disorders Acute kidney injury 1 2.6 Proteinuria 1 2.6 Cardiac disorders Atrial fibrillation3 1 2.6 Endocrine disorders Inappropriate antidiuretic hormone
secretion 1 2.6 Eye disorders Ulcerative keratitis 1 2.6 Hepatobiliary disorders Cholangitis 1 2.6 Immune system disorders Contrast media allergy 1 2.6 Musculoskeletal and connective tissue disorders Muscle spasms 1 2.6 Nervous system disorders
Headache 1 2.6 Reproductive system and breast disorders Vaginal haemorrhage 1 2.6 Skin and subcutaneous tissue disorders Rash maculo-papular 1 2.6 TEAEs by maximum severity for all patients in Ph1a dose escalation and Ph1b Cohort A dose expansion
(N=39)1 IMA203
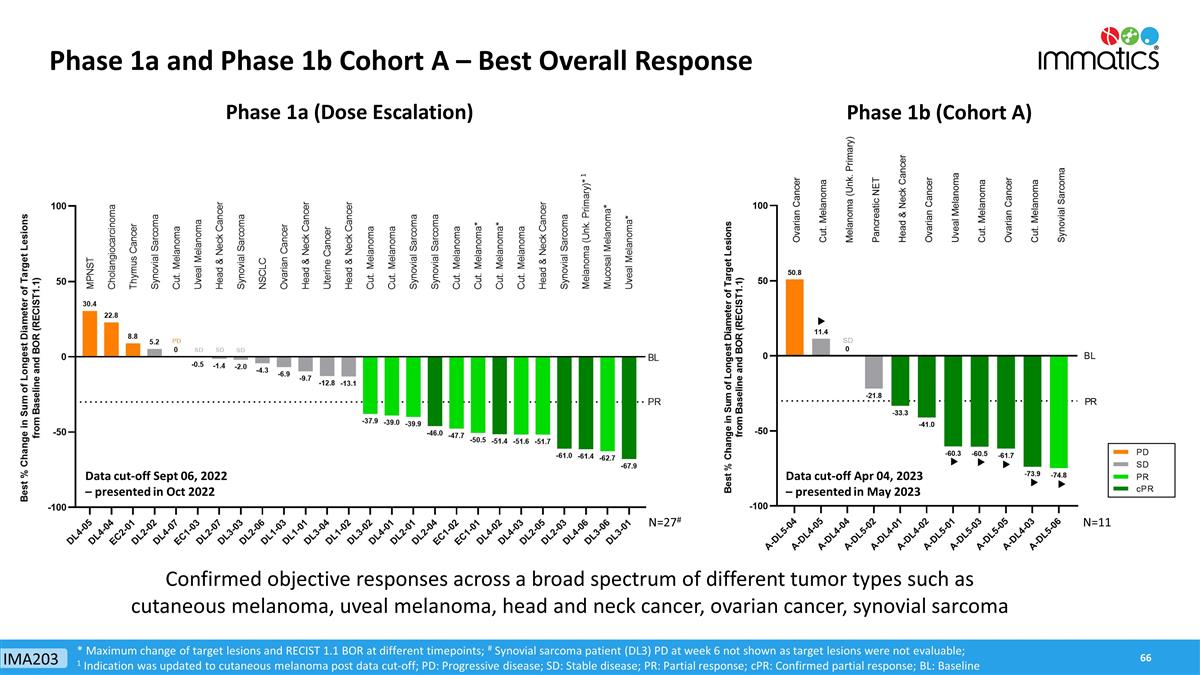
Phase 1a and Phase 1b Cohort A
– Best Overall Response Confirmed objective responses across a broad spectrum of different tumor types such as cutaneous melanoma, uveal melanoma, head and neck cancer, ovarian cancer, synovial sarcoma * Maximum change of target lesions and
RECIST 1.1 BOR at different timepoints; # Synovial sarcoma patient (DL3) PD at week 6 not shown as target lesions were not evaluable; 1 Indication was updated to cutaneous melanoma post data cut-off; PD: Progressive disease; SD: Stable disease; PR:
Partial response; cPR: Confirmed partial response; BL: Baseline Phase 1a (Dose Escalation) Phase 1b (Cohort A) IMA203 N=27# N=11 Data cut-off Sept 06, 2022 – presented in Oct 2022 Data cut-off Apr 04, 2023 – presented in May 2023 1
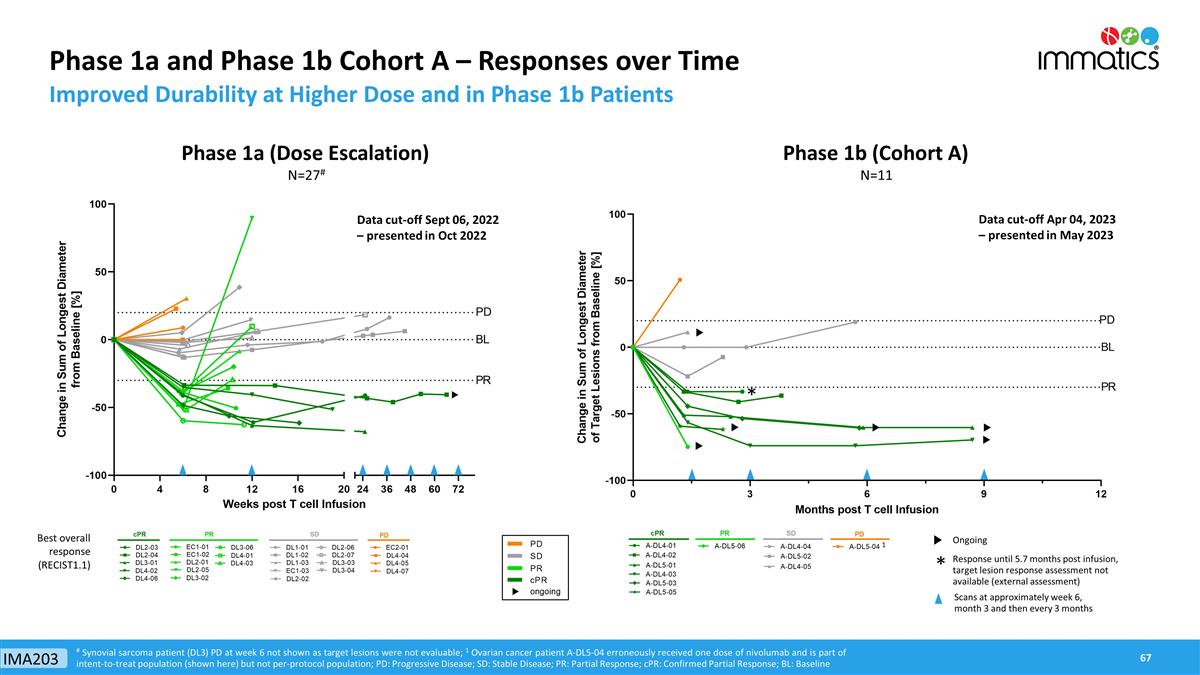
Phase 1a and Phase 1b Cohort A
– Responses over Time Improved Durability at Higher Dose and in Phase 1b Patients Phase 1a (Dose Escalation) N=27# Phase 1b (Cohort A) N=11 Best overall response (RECIST1.1) IMA203 # Synovial sarcoma patient (DL3) PD at week 6 not shown as
target lesions were not evaluable; 1 Ovarian cancer patient A-DL5-04 erroneously received one dose of nivolumab and is part of intent-to-treat population (shown here) but not per-protocol population; PD: Progressive Disease; SD: Stable Disease; PR:
Partial Response; cPR: Confirmed Partial Response; BL: Baseline Scans at approximately week 6, month 3 and then every 3 months Ongoing * 1 * Response until 5.7 months post infusion, target lesion response assessment not available (external
assessment) Data cut-off Sept 06, 2022 – presented in Oct 2022 Data cut-off Apr 04, 2023 – presented in May 2023
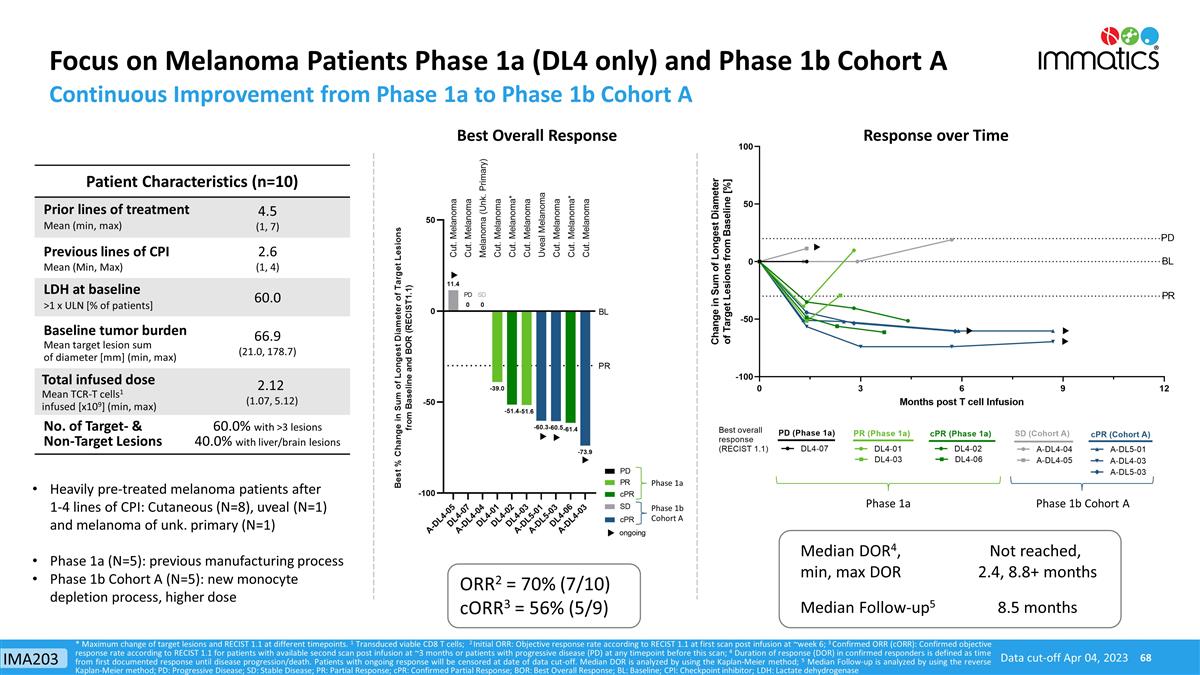
Focus on Melanoma Patients Phase 1a
(DL4 only) and Phase 1b Cohort A Continuous Improvement from Phase 1a to Phase 1b Cohort A Patient Characteristics (n=10) IMA203 melanoma patients (n=14) Prior lines of treatment Mean (min, max) 4.5 (1, 7) Previous lines of CPI Mean (Min, Max)
2.6 (1, 4) LDH at baseline >1 x ULN [% of patients] 60.0 Baseline tumor burden Mean target lesion sum of diameter [mm] (min, max) 66.9 (21.0, 178.7) Total infused dose Mean TCR-T cells1 infused [x109] (min, max) 2.12 (1.07,
5.12) No. of Target- & Non-Target Lesions 60.0% with >3 lesions 40.0% with liver/brain lesions ORR2 = 70% (7/10) cORR3 = 56% (5/9) Median DOR4, min, max DOR Not reached, 2.4, 8.8+ months Median Follow-up5 8.5 months * Maximum
change of target lesions and RECIST 1.1 at different timepoints. 1 Transduced viable CD8 T cells; 2 Initial ORR: Objective response rate according to RECIST 1.1 at first scan post infusion at ~week 6; 3 Confirmed ORR
(cORR): Confirmed objective response rate according to RECIST 1.1 for patients with available second scan post infusion at ~3 months or patients with progressive disease (PD) at any timepoint before this scan; 4 Duration of response (DOR) in
confirmed responders is defined as time from first documented response until disease progression/death. Patients with ongoing response will be censored at date of data cut-off. Median DOR is analyzed by using the Kaplan-Meier method; 5 Median
Follow-up is analyzed by using the reverse Kaplan-Meier method; PD: Progressive Disease; SD: Stable Disease; PR: Partial Response; cPR: Confirmed Partial Response; BOR: Best Overall Response; BL: Baseline; CPI: Checkpoint inhibitor; LDH: Lactate
dehydrogenase Data cut-off Apr 04, 2023 Heavily pre-treated melanoma patients after 1-4 lines of CPI: Cutaneous (N=8), uveal (N=1) and melanoma of unk. primary (N=1) Phase 1a (N=5): previous manufacturing process Phase 1b Cohort A (N=5): new
monocyte depletion process, higher dose Phase 1a Phase 1b Cohort A Best Overall Response Response over Time Phase 1a Phase 1b Cohort A IMA203

v3.23.2
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEnd date of current fiscal year in the format --MM-DD.
| Name: |
dei_CurrentFiscalYearEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gMonthDayItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFiscal period values are FY, Q1, Q2, and Q3. 1st, 2nd and 3rd quarter 10-Q or 10-QT statements have value Q1, Q2, and Q3 respectively, with 10-K, 10-KT or other fiscal year statements having FY.
| Name: |
dei_DocumentFiscalPeriodFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fiscalPeriodItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThis is focus fiscal year of the document report in YYYY format. For a 2006 annual report, which may also provide financial information from prior periods, fiscal 2006 should be given as the fiscal year focus. Example: 2006.
| Name: |
dei_DocumentFiscalYearFocus |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:gYearItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Condensed Consolidated Statement of Profit/(Loss) of Immatics N.V. - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Profit or loss [abstract] |
|
|
|
|
| Revenue from collaboration agreements |
€ 22,354
|
€ 17,215
|
€ 32,150
|
€ 120,123
|
| Research and development expenses |
(27,317)
|
(25,216)
|
(54,898)
|
(50,360)
|
| General and administrative expenses |
(9,358)
|
(8,683)
|
(18,944)
|
(17,961)
|
| Other income |
6
|
27
|
948
|
32
|
| Operating result |
(14,315)
|
(16,657)
|
(40,744)
|
51,834
|
| Change in fair value of liabilities for warrants |
(13,105)
|
(2,786)
|
(5,708)
|
13,743
|
| Other financial income |
3,954
|
7,015
|
6,748
|
8,774
|
| Other financial expenses |
(1,144)
|
(407)
|
(4,653)
|
(1,524)
|
| Financial result |
(10,295)
|
3,822
|
(3,613)
|
20,993
|
| Profit/(loss) before taxes |
(24,610)
|
(12,835)
|
(44,357)
|
72,827
|
| Taxes on income |
0
|
(1,145)
|
0
|
(1,145)
|
| Net profit/(loss) |
€ (24,610)
|
€ (13,980)
|
€ (44,357)
|
€ 71,682
|
| Net profit/(loss) per share: |
|
|
|
|
| Basic |
€ (0.32)
|
€ (0.22)
|
€ (0.58)
|
€ 1.12
|
| Diluted |
€ (0.32)
|
€ (0.22)
|
€ (0.58)
|
€ 1.11
|
| X |
- DefinitionThe amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator) divided by the weighted average number of ordinary shares outstanding during the period (the denominator). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 67
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_67&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_BasicEarningsLossPerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator), divided by the weighted average number of ordinary shares outstanding during the period (the denominator), both adjusted for the effects of all dilutive potential ordinary shares. [Refer: Ordinary shares [member]; Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 67
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_67&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DilutedEarningsLossPerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_EarningsPerShareAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of income or cost associated with interest and other financing activities of the entity. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 85
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_85&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinanceIncomeCost |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe gains (losses) on financial liabilities at fair value through profit or loss. [Refer: Financial liabilities at fair value through profit or loss] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_GainsLossesOnFinancialLiabilitiesAtFairValueThroughProfitOrLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of expense relating to general and administrative activities of the entity. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_GeneralAndAdministrativeExpense |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_IncomeStatementAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of finance costs that the entity does not separately disclose in the same statement or note. [Refer: Finance costs] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherFinanceCost |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of finance income that the entity does not separately disclose in the same statement or note. [Refer: Finance income] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherFinanceIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of operating income that the entity does not separately disclose in the same statement or note. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 26
-IssueDate 2022-03-24
-Paragraph 35
-Subparagraph b
-Clause iv
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=26&code=ifrs-tx-2022-en-r&anchor=para_35_b_iv&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 103
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_103&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 102
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_102&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 18
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_18_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Note Effective on first application of IFRS 9
-Name IFRS
-Number 4
-IssueDate 2022-03-24
-Paragraph 39L
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=4&code=ifrs-tx-2022-en-b&anchor=para_39L_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Note Effective 2023-01-01
-Name IFRS
-Number 17
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2022-en-r&anchor=para_113_b&doctype=Standard
-URIDate 2022-03-24
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_a&doctype=Standard
-URIDate 2022-03-24
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe profit (loss) before tax expense or income. [Refer: Profit (loss)] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 5
-IssueDate 2022-03-24
-Paragraph 33
-Subparagraph b
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=5&code=ifrs-tx-2022-en-r&anchor=para_33_b_i&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 103
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_103&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 102
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_102&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLossBeforeTax |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe profit (loss) from operating activities of the entity. [Refer: Profit (loss)] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 32
-IssueDate 2022-03-24
-Paragraph IE33
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=32&code=ifrs-tx-2022-en-r&anchor=para_IE33&doctype=Illustrative%20Examples
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 85
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_85&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLossFromOperatingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of expenditure directly attributable to research or development activities, recognised in profit or loss. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 38
-IssueDate 2022-03-24
-Paragraph 126
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=38&code=ifrs-tx-2022-en-r&anchor=para_126&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ResearchAndDevelopmentExpense |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionRevenue from collaboration agreements
| Name: |
imtx_RevenueFromCollaborationAgreements |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.23.2
Condensed Consolidated Statement of Comprehensive Income/(Loss) of Immatics N.V. - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Statement of comprehensive income [abstract] |
|
|
|
|
| Net profit/(loss) |
€ (24,610)
|
€ (13,980)
|
€ (44,357)
|
€ 71,682
|
| Items that may be reclassified subsequently to profit or loss |
|
|
|
|
| Currency translation differences from foreign operations |
(224)
|
778
|
340
|
1,338
|
| Total comprehensive income/(loss) for the year |
€ (24,834)
|
€ (13,202)
|
€ (44,017)
|
€ 73,020
|
| X |
- References
+ Details
| Name: |
ifrs-full_ComponentsOfOtherComprehensiveIncomeThatWillBeReclassifiedToProfitOrLossNetOfTaxAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of change in equity resulting from transactions and other events, other than those changes resulting from transactions with owners in their capacity as owners. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause ix
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_ix&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_c&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ComprehensiveIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe gains (losses) recognised in other comprehensive income on exchange differences on the translation of financial statements of foreign operations, net of tax, before reclassification adjustments. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 91
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_91_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_GainsLossesOnExchangeDifferencesOnTranslationNetOfTax |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 18
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_18_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Note Effective on first application of IFRS 9
-Name IFRS
-Number 4
-IssueDate 2022-03-24
-Paragraph 39L
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=4&code=ifrs-tx-2022-en-b&anchor=para_39L_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Note Effective 2023-01-01
-Name IFRS
-Number 17
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2022-en-r&anchor=para_113_b&doctype=Standard
-URIDate 2022-03-24
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_a&doctype=Standard
-URIDate 2022-03-24
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_StatementOfComprehensiveIncomeAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Condensed Consolidated Statement of Financial Position of Immatics N.V. - EUR (€)
€ in Thousands |
Jun. 30, 2023 |
Dec. 31, 2022 |
| Current assets |
|
|
| Cash and cash equivalents |
€ 130,405
|
€ 148,519
|
| Other financial assets |
217,222
|
213,686
|
| Accounts receivables |
330
|
1,111
|
| Other current assets |
16,668
|
13,838
|
| Total current assets |
364,625
|
377,154
|
| Non-current assets |
|
|
| Property, plant and equipment |
27,188
|
13,456
|
| Intangible assets |
1,655
|
1,632
|
| Right-of-use assets |
14,749
|
13,033
|
| Other non-current assets |
1,972
|
2,545
|
| Total non-current assets |
45,564
|
30,666
|
| Total assets |
410,189
|
407,820
|
| Current liabilities |
|
|
| Provisions |
3,117
|
0
|
| Accounts payables |
19,904
|
13,056
|
| Deferred revenue |
67,997
|
64,957
|
| Liabilities for warrants |
22,622
|
16,914
|
| Lease liabilities |
2,737
|
2,159
|
| Other current liabilities |
7,929
|
9,366
|
| Total current liabilities |
124,306
|
106,452
|
| Non-current liabilities |
|
|
| Deferred revenue |
53,559
|
75,759
|
| Lease liabilities |
14,085
|
12,403
|
| Other non-current liabilities |
26
|
42
|
| Total non-current liabilities |
67,670
|
88,204
|
| Shareholders' equity |
|
|
| Share capital |
804
|
767
|
| Share premium |
763,206
|
714,177
|
| Accumulated deficit |
(544,656)
|
(500,299)
|
| Other reserves |
(1,141)
|
(1,481)
|
| Total shareholders' equity |
218,213
|
213,164
|
| Total liabilities and shareholders' equity |
€ 410,189
|
€ 407,820
|
| X |
- DefinitionThe amount of a present economic resource controlled by the entity as a result of past events. Economic resource is a right that has the potential to produce economic benefits. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_a&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_c&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_e&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_Assets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of cash on hand and demand deposits, along with short-term, highly liquid investments that are readily convertible to known amounts of cash and that are subject to an insignificant risk of changes in value. [Refer: Cash; Cash equivalents] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 45
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_45&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B13
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B13_a&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashAndCashEquivalents |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of assets that the entity (a) expects to realise or intends to sell or consume in its normal operating cycle; (b) holds primarily for the purpose of trading; (c) expects to realise within twelve months after the reporting period; or (d) classifies as cash or cash equivalents (as defined in IAS 7) unless the asset is restricted from being exchanged or used to settle a liability for at least twelve months after the reporting period. [Refer: Assets] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_i&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_CurrentAssetsAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of current contract liabilities. [Refer: Contract liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 105
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_105&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentContractLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current lease liabilities. [Refer: Lease liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 16
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2022-en-r&anchor=para_47_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentLeaseLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionExpiry date 2023-01-01: The amount of liabilities that: (a) the entity expects to settle in its normal operating cycle; (b) the entity holds primarily for the purpose of trading; (c) are due to be settled within twelve months after the reporting period; or (d) the entity does not have an unconditional right to defer settlement for at least twelve months after the reporting period.
Effective 2023-01-01: The amount of liabilities that: (a) the entity expects to settle in its normal operating cycle; (b) the entity holds primarily for the purpose of trading; (c) are due to be settled within twelve months after the reporting period; or (d) the entity does not have the right at the end of the reporting period to defer settlement for at least twelve months after the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_iii&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 69
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_69&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_CurrentLiabilitiesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of current provisions, including provisions for employee benefits. [Refer: Provisions] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph l
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_l&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentProvisions |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current trade receivables. [Refer: Trade receivables] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 68
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_68&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentTradeReceivables |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current warrant liabilities. [Refer: Warrant liability] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentWarrantLiability |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of residual interest in the assets of the entity after deducting all its liabilities. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_a&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_i&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_a&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_Equity |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_EquityAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of the entity's equity and liabilities. [Refer: Equity; Liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_EquityAndLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of identifiable non-monetary assets without physical substance. This amount does not include goodwill. [Refer: Goodwill] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 38
-IssueDate 2022-03-24
-Paragraph 118
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=38&code=ifrs-tx-2022-en-r&anchor=para_118_e&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IntangibleAssetsOtherThanGoodwill |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe nominal value of capital issued. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IssuedCapital |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of assets that do not meet the definition of current assets. [Refer: Current assets] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_ii&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_NoncurrentAssetsAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of non-current contract liabilities. [Refer: Contract liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 105
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_105&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentContractLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of non-current lease liabilities. [Refer: Lease liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 16
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2022-en-r&anchor=para_47_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentLeaseLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of liabilities that do not meet the definition of current liabilities. [Refer: Current liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause iv
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_iv&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 69
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_69&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_NoncurrentLiabilitiesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of current assets that the entity does not separately disclose in the same statement or note. [Refer: Current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherCurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current financial assets that the entity does not separately disclose in the same statement or note. [Refer: Other financial assets; Current financial assets] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherCurrentFinancialAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current liabilities that the entity does not separately disclose in the same statement or note. [Refer: Current liabilities] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherCurrentLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of non-current assets that the entity does not separately disclose in the same statement or note. [Refer: Non-current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherNoncurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of non-current liabilities that the entity does not separately disclose in the same statement or note. [Refer: Non-current liabilities] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherNoncurrentLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionA component of equity representing reserves within equity, not including retained earnings. [Refer: Retained earnings] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherReserves |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of tangible assets that: (a) are held for use in the production or supply of goods or services, for rental to others, or for administrative purposes; and (b) are expected to be used during more than one period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 16
-IssueDate 2022-03-24
-Paragraph 73
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=16&code=ifrs-tx-2022-en-r&anchor=para_73_e&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_PropertyPlantAndEquipment |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionA component of equity representing the entity's cumulative undistributed earnings or deficit. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph IG6
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_IG6&doctype=Implementation%20Guidance
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_RetainedEarnings |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of assets that represent a lessee's right to use an underlying asset for the lease term. Underlying asset is an asset that is the subject of a lease, for which the right to use that asset has been provided by a lessor to a lessee. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 16
-IssueDate 2022-03-24
-Paragraph 53
-Subparagraph j
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2022-en-r&anchor=para_53_j&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_RightofuseAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount received or receivable from the issuance of the entity's shares in excess of nominal value. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_SharePremium |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe current amount of payment due to suppliers for goods and services used in entity's business. [Refer: Current liabilities; Trade payables] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 70
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_70&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_TradeAndOtherCurrentPayablesToTradeSuppliers |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.23.2
Condensed Consolidated Statement of Cash Flows of Immatics N.V. - EUR (€)
€ in Thousands |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Cash flows from operating activities |
|
|
| Net profit/(loss) |
€ (44,357)
|
€ 71,682
|
| Taxes on income |
0
|
1,145
|
| Profit/(loss) before tax |
(44,357)
|
72,827
|
| Adjustments for: |
|
|
| Interest income |
(4,999)
|
(23)
|
| Depreciation and amortization |
3,666
|
3,407
|
| Interest expenses |
401
|
538
|
| Equity settled share-based payment |
11,615
|
11,262
|
| Net foreign exchange differences and expected credit losses |
4,081
|
(7,834)
|
| Change in fair value of liabilities for warrants |
5,708
|
(13,743)
|
| Changes in: |
|
|
| Decrease/(increase) in accounts receivables |
781
|
(280)
|
| Decrease/(increase) in other assets |
765
|
(6,903)
|
| (Decrease)/increase in deferred revenue, accounts payables and other liabilities |
(9,889)
|
96,933
|
| Interest received |
2,051
|
23
|
| Interest paid |
(146)
|
(434)
|
| Income tax paid |
0
|
0
|
| Net cash (used in)/provided by operating activities |
(30,323)
|
155,773
|
| Cash flows from investing activities |
|
|
| Payments for property, plant and equipment |
(15,004)
|
(1,965)
|
| Payments for intangible assets |
(154)
|
(6)
|
| Proceeds from disposal of property, plant and equipment |
0
|
1
|
| Payments for investments classified in Other financial assets |
(170,326)
|
(59,253)
|
| Proceeds from maturity of investments classified in Other financial assets |
164,929
|
12,695
|
| Net cash (used in)/provided by investing activities |
(20,555)
|
(48,528)
|
| Cash flows from financing activities |
|
|
| Proceeds from issuance of shares to equity holders |
38,608
|
17,112
|
| Transaction costs deducted from equity |
(1,157)
|
(515)
|
| Repayment of lease liabilities |
(1,866)
|
(1,394)
|
| Net cash provided by/(used in) financing activities |
35,585
|
15,203
|
| Net (decrease)/increase in cash and cash equivalents |
(15,293)
|
122,448
|
| Cash and cash equivalents at beginning |
148,519
|
132,994
|
| Effects of exchange rate changes and expected credit losses on cash and cash equivalents |
(2,821)
|
9,683
|
| Cash and cash equivalents at end |
€ 130,405
|
€ 265,125
|
| X |
- DefinitionAdjustments for decrease (increase) in other assets to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Other assets; Profit (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustmentsForDecreaseIncreaseInOtherAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for decrease (increase) in trade accounts receivable to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Profit (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustmentsForDecreaseIncreaseInTradeAccountReceivable |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for depreciation and amortisation expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Depreciation and amortisation expense; Profit (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustmentsForDepreciationAndAmortisationExpense |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for interest expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Interest expense; Profit (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustmentsForInterestExpense |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for interest income expense to reconcile profit (loss) to net cash flow from (used in) operating activities. [Refer: Interest income; Profit (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustmentsForInterestIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_AdjustmentsForReconcileProfitLossAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of cash on hand and demand deposits, along with short-term, highly liquid investments that are readily convertible to known amounts of cash and that are subject to an insignificant risk of changes in value. [Refer: Cash; Cash equivalents] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 45
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_45&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B13
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B13_a&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashAndCashEquivalents |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash flows from (used in) financing activities, which are activities that result in changes in the size and composition of the contributed equity and borrowings of the entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInFinancingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_CashFlowsFromUsedInFinancingActivitiesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash flows from (used in) investing activities, which are the acquisition and disposal of long-term assets and other investments not included in cash equivalents. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInInvestingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_CashFlowsFromUsedInInvestingActivitiesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash flows from (used in) operating activities, which are the principal revenue-producing activities of the entity and other activities that are not investing or financing activities. [Refer: Revenue] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInOperatingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_CashFlowsFromUsedInOperatingActivitiesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe gains (losses) on financial liabilities at fair value through profit or loss. [Refer: Financial liabilities at fair value through profit or loss] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 20
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_20_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_GainsLossesOnFinancialLiabilitiesAtFairValueThroughProfitOrLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) in cash and cash equivalents before the effect of exchange rate changes on cash and cash equivalents held in foreign currencies. [Refer: Cash and cash equivalents; Effect of exchange rate changes on cash and cash equivalents] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 45
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_45&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IncreaseDecreaseInCashAndCashEquivalentsBeforeEffectOfExchangeRateChanges |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for interest paid, classified as operating activities. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 31
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_31&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_InterestPaidClassifiedAsOperatingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from interest received, classified as operating activities. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 31
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_31&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_InterestReceivedClassifiedAsOperatingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for payment of lease liabilities, classified as financing activities. [Refer: Lease liabilities] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 17
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_17_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_PaymentsOfLeaseLiabilitiesClassifiedAsFinancingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from issuing shares. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 17
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_17_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProceedsFromIssuingShares |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from sales of investments other than investments accounted for using the equity method. [Refer: Investments accounted for using equity method; Investments other than investments accounted for using equity method] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 16
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_16&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProceedsFromSalesOfInvestmentsOtherThanInvestmentsAccountedForUsingEquityMethod |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash inflow from sales of property, plant and equipment, classified as investing activities. [Refer: Property, plant and equipment] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 16
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_16_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProceedsFromSalesOfPropertyPlantAndEquipmentClassifiedAsInvestingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 18
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_18_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Note Effective on first application of IFRS 9
-Name IFRS
-Number 4
-IssueDate 2022-03-24
-Paragraph 39L
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=4&code=ifrs-tx-2022-en-b&anchor=para_39L_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Note Effective 2023-01-01
-Name IFRS
-Number 17
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2022-en-r&anchor=para_113_b&doctype=Standard
-URIDate 2022-03-24
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_a&doctype=Standard
-URIDate 2022-03-24
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe profit (loss) before tax expense or income. [Refer: Profit (loss)] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 5
-IssueDate 2022-03-24
-Paragraph 33
-Subparagraph b
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=5&code=ifrs-tx-2022-en-r&anchor=para_33_b_i&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 103
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_103&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 102
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_102&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLossBeforeTax |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for the purchases of intangible assets, classified as investing activities. [Refer: Intangible assets other than goodwill] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 16
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_16_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_PurchaseOfIntangibleAssetsClassifiedAsInvestingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for the purchase of investments other than investments accounted for using equity method. [Refer: Investments accounted for using equity method; Investments other than investments accounted for using equity method] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 16
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_16&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_PurchaseOfInvestmentsOtherThanInvestmentsAccountedForUsingEquityMethod |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash outflow for the purchases of property, plant and equipment, classified as investing activities. [Refer: Property, plant and equipment] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 16
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_16_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_PurchaseOfPropertyPlantAndEquipmentClassifiedAsInvestingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustment for equity settled share-based payment.
| Name: |
imtx_AdjustmentForEquitySettledSharebasedPayment |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for increase decrease in deferred revenue trade and other payables.
| Name: |
imtx_AdjustmentsForIncreaseDecreaseInDeferredRevenueTradeAndOtherPayables |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for unrealised foreign exchange and expected credit loss.
| Name: |
imtx_AdjustmentsForUnrealisedForeignExchangeAndExpectedCreditLoss |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionChanges in working capital [Abstract].
| Name: |
imtx_ChangesInWorkingCapitalAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionEffects of exchange rate changes and expected credit losses on cash and cash equivalents.
| Name: |
imtx_EffectsOfExchangeRateChangesAndExpectedCreditLossesOnCashAndCashEquivalents |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionTransaction costs deducted from equity.
| Name: |
imtx_TransactionCostsDeductedFromEquity |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.23.2
Condensed Consolidated Statement of Changes in Shareholders' equity of Immatics N.V. - EUR (€)
€ in Thousands |
Total |
Share capital [member] |
Share premium [member] |
Accumulated deficit [member] |
Other reserves [member] |
| Beginning Balance at Dec. 31, 2021 |
€ 24,063
|
€ 629
|
€ 565,192
|
€ (537,813)
|
€ (3,945)
|
| Other comprehensive income |
1,338
|
|
|
|
1,338
|
| Net profit/(loss) |
71,682
|
|
|
71,682
|
|
| Comprehensive income/(loss) for the year |
73,020
|
|
|
71,682
|
1,338
|
| Equity-settled share-based compensation |
11,262
|
|
11,262
|
|
|
| Share options exercised |
1
|
|
1
|
|
|
| Issue of share capital – net of transaction costs |
16,595
|
24
|
16,571
|
|
|
| Ending Balance at Jun. 30, 2022 |
124,941
|
653
|
593,026
|
(466,131)
|
(2,607)
|
| Beginning Balance at Dec. 31, 2022 |
213,164
|
767
|
714,177
|
(500,299)
|
(1,481)
|
| Other comprehensive income |
340
|
|
|
|
340
|
| Net profit/(loss) |
(44,357)
|
|
|
(44,357)
|
|
| Comprehensive income/(loss) for the year |
(44,017)
|
|
|
(44,357)
|
340
|
| Equity-settled share-based compensation |
11,615
|
|
11,615
|
|
|
| Share options exercised |
40
|
|
40
|
|
|
| Issue of share capital – net of transaction costs |
37,411
|
37
|
37,374
|
|
|
| Ending Balance at Jun. 30, 2023 |
€ 218,213
|
€ 804
|
€ 763,206
|
€ (544,656)
|
€ (1,141)
|
| X |
- DefinitionThe amount of change in equity resulting from transactions and other events, other than those changes resulting from transactions with owners in their capacity as owners. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause ix
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_ix&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_c&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ComprehensiveIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of residual interest in the assets of the entity after deducting all its liabilities. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_a&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_i&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_a&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 13
-IssueDate 2022-03-24
-Paragraph 93
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=13&code=ifrs-tx-2022-en-r&anchor=para_93_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_Equity |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe increase (decrease) in equity resulting from the exercise of options. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IncreaseDecreaseThroughExerciseOfOptions |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) in equity resulting from share-based payment transactions. [Refer: Equity] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_iii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IncreaseDecreaseThroughSharebasedPaymentTransactions |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of income and expense (including reclassification adjustments) that is not recognised in profit or loss as required or permitted by IFRSs. [Refer: IFRSs [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B12
-Subparagraph b
-Clause viii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B12_b_viii&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_b&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 91
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_91_a&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_ii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherComprehensiveIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 18
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_18_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Note Effective on first application of IFRS 9
-Name IFRS
-Number 4
-IssueDate 2022-03-24
-Paragraph 39L
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=4&code=ifrs-tx-2022-en-b&anchor=para_39L_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Note Effective 2023-01-01
-Name IFRS
-Number 17
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2022-en-r&anchor=para_113_b&doctype=Standard
-URIDate 2022-03-24
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_a&doctype=Standard
-URIDate 2022-03-24
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIssue of share capital – net of transaction costs.
| Name: |
imtx_IssueOfShareCapitalNetOfTransactionCosts |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
v3.23.2
Group information
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Group information |
Immatics N.V., together with its German subsidiary Immatics Biotechnologies GmbH and its U.S. subsidiary, Immatics US Inc., (“Immatics” or “the Group”) is a biotechnology group that is primarily engaged in the research and development of T cell redirecting immunotherapies for the treatment of cancer. Immatics N.V., a Dutch public limited liability company, was converted on July 1, 2020 from Immatics B.V., a Dutch company with limited liability. Immatics Biotechnologies GmbH (“Immatics GmbH”) and Immatics US Inc. became wholly-owned subsidiaries of Immatics N.V. as part of the ARYA Merger on July 1, 2020. Immatics N.V. is registered with the commercial register at the Netherlands Chamber of Commerce under RSIN 861058926 with a corporate seat in Amsterdam and is located at Paul-Ehrlich Str. 15 in 72076 Tübingen, Germany. These interim condensed consolidated financial statements of the Group for the three and six months ended June 30, 2023, were authorized for issue by the Audit Committee of Immatics N.V. on August 17, 2023.
|
| X |
- DefinitionThe disclosure of the composition of the group (the parent and all its subsidiaries). [Refer: Subsidiaries [member]; Parent [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_10_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfCompositionOfGroupExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Significant events and changes in the current reporting period
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Significant events and changes in the current reporting period |
2. Significant events and changes in the current reporting period The following significant events or transactions occurred during the three and six months ended June 30, 2023. Opt-In of TCR-T Candidate from ongoing collaboration with BMS Immatics GmbH entered into a License agreement (the “BMS Opt-In agreement”) with Bristol-Myer-Squibb Company (“BMS”). The agreement became effective on April 28, 2023. Pursuant to the BMS Opt-In agreement, the Group received an option exercise fee in the amount of $15 million (€13.7 million). Under the 2019 agreement with BMS, Immatics granted BMS the option to enter into a pre-negotiated license agreement on a target-by-target basis. Immatics developed individual TCR-T products directed against targets under the terms of the 2019 agreement. Under the BMS Opt-In agreement signed on April 28, 2023, BMS exercised its first option and entered into an exclusive license agreement for one target. The BMS Opt-In agreement created the right for BMS to receive the exclusive license and the right for Immatics to receive the Opt-In exercise fee as well as potential future milestones and royalties. Immatics promised an additional distinct performance obligation under the Opt-in agreement which is to issue the license to BMS. The price of the contract increases by an amount of consideration that reflects the entity’s stand-alone selling price of the license. The license grants BMS a right to use the license as no further work from Immatics is required under the agreement. The potential milestone and royalty payments are accounted for under the most likely method. No variable payment is considered likely, therefore, no variable payments are considered as part of the transaction price. For the three and six months ended June 30, 2023, the Group recognized €13.7 million of revenue related to the BMS Opt-In agreement. Macroeconomic environment Currently, multiple global uncertainties are existing. The conflict between Russia and Ukraine has resulted, and is expected to further result, in significant disruption, instability and volatility in global markets, as well as higher energy and other commodity prices. Since the Company is not currently conducting any business or receiving any material services from vendors located in Russia or Ukraine, it does not expect that the ongoing war will have a direct impact on its operations in the near term. However, the Company may be indirectly affected by price increases or certain policy changes, such as new tax legislation, economic sanctions and comparable measures. In addition, other geopolitical instabilities might impact the Group in the future. During the six months ended June 30, 2023, Silicon Valley Bank and Credit Suisse, two large banks, as well as other smaller banks, were subject to liquidity problems. The Group does not hold deposits or securities with any of the affected banks. While the banking system remained stable overall, we will continue to closely monitor the situation. While there is currently no material direct risk identified for the Group from COVID-19, Immatics will continue to closely monitor the effects of the pandemic as well. |
| X |
- DefinitionThe entire disclosure for changes in accounting policies, accounting estimates and errors. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 8
-IssueDate 2022-03-24
-Section Accounting policies
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=8&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IAS08_g7-31__IAS08_g7-31_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfChangesInAccountingPoliciesAccountingEstimatesAndErrorsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Significant accounting policies
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Significant accounting policies |
3. Significant accounting policies The interim condensed consolidated financial statements of the Group as of June 30, 2023 and for the three and six months ended June 30, 2023 and 2022 have been prepared on a going concern basis in accordance with International Accounting Standard 34 (“Interim Financial Reporting”), as issued by the International Accounting Standards Board (“IASB”) and have not been audited or reviewed by a statutory auditor. In accordance with IAS 34, the interim condensed consolidated financial statements do not include all the information and disclosures required in the annual financial statements and should be read in conjunction with the Group’s annual financial statements for the year ended December 31, 2022, which have been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the IASB, taking into account the recommendations of the International Financial Reporting Standards Interpretations Committee (“IFRS IC”). In these condensed notes to the consolidated financial statements, information is provided primarily on the items for which there have been significant changes compared with the consolidated financial statements of the Group for fiscal year 2022. The interim condensed consolidated financial statements are presented in Euros. Amounts are stated in thousands of Euros, unless otherwise indicated. For technical reasons, the information provided in these financial statements may contain rounding differences of +/- one unit. The accounting policies adopted in the preparation of the interim condensed consolidated financial statements are consistent with those followed in the preparation of the Group’s annual consolidated financial statements for the year ended December 31, 2022. The new and amended standards and interpretations applicable for the first time as of January 1, 2023, as disclosed in the notes to the consolidated financial statements for the year ended December 31, 2022, had no impact on the interim condensed consolidated financial statements of the Group for the three and six months ended June 30, 2023. Estimates and assumptions have to be made in the interim consolidated financial statements as of June 30, 2023. These have an impact on the amount and disclosure of the recognized assets and liabilities, income and expenses, and contingent liabilities. The estimates and judgments are basically unchanged from the circumstances described in the consolidated financial statements of the Group for the fiscal year 2022. Developments deviating from this may result in the amounts that arise deviating from the original estimates. These possible developments are outside the sphere of influence of the management. Revision of previously issued financial statements During the preparation of the unaudited interim consolidated financial statements for the three and nine months ended September 30, 2022, the Group identified an error in the presentation of ‘Net foreign exchange differences’ and ‘Effects of exchange rate changes on cash and cash equivalents’ in the statement of cash flows. The error resulted in a presentation of effects from exchange rate changes on non-functional currency denominated cash and cash equivalents in Immatics N.V. and Immatics GmbH as operating cash flow instead of the presentation as non-cash items in effects of exchange rate changes on cash and cash equivalents. This error had no impact on the Company’s consolidated statements of financial position, of profit/(loss), of comprehensive income/(loss) and of consolidated statements of changes in equity. The Company assessed the materiality of these errors on the previously issued consolidated financial statements and concluded that the errors were not material to any period presented. The impact of the revision of the previously issued financial statements is as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, 2022 |
|
| |
|
|
|
|
|
|
|
|
|
Net foreign exchange differences and expected credit losses |
|
|
115 |
|
|
|
(7,949 |
) |
|
|
(7,834 |
) |
Net cash provided by/(used in) operating activities |
|
|
163,722 |
|
|
|
(7,949 |
) |
|
|
155,773 |
|
Net cash (used in)/provided by investing activities |
|
|
(48,528 |
) |
|
|
— |
|
|
|
(48,528 |
) |
Net cash (used in)/provided by financing activities |
|
|
15,203 |
|
|
|
— |
|
|
|
15,203 |
|
Net increase/(decrease) in cash and cash equivalents |
|
|
130,397 |
|
|
|
(7,949 |
) |
|
|
122,448 |
|
Cash and cash equivalents at beginning of period |
|
|
132,994 |
|
|
|
— |
|
|
|
132,994 |
|
Effects of exchange rate changes on cash and cash equivalents |
|
|
1,734 |
|
|
|
7,949 |
|
|
|
9,683 |
|
Cash and cash equivalents at end of period |
|
|
265,125 |
|
|
|
— |
|
|
|
265,125 |
|
|
| X |
- DefinitionThe entire disclosure for significant accounting policies applied by the entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Note Expiry date 2023-01-01
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 117
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_117&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfSummaryOfSignificantAccountingPoliciesExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Segment information
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Segment information |
The Group manages its operations as a single segment for the purpose of assessing performance and making operating decisions. The Group’s focus is on the research and development of T cell redirecting immunotherapies for the treatment of cancer. The Chief Executive Officer is the chief operating decision maker who regularly reviews the consolidated operating results and makes decisions about the allocation of the Group’s resources.
|
| X |
- DefinitionThe entire disclosure for operating segments. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Section Disclosure
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IFRS08_g20-24__IFRS08_g20-24_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfEntitysReportableSegmentsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Revenue from collaboration agreements
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Revenue from collaboration agreements |
5. Revenue from collaboration agreements The Group currently earns revenue through strategic collaboration agreements with third party pharmaceutical and biotechnology companies. As of June 30, 2023, the Group had four strategic collaboration agreements in place after the collaboration with GSK plc (“GSK”) was terminated in 2022. Three of the four collaboration programs are still at pre-clinical development stage and IMA401, which is subject of a collaboration with Bristol Myers Squibb (“BMS”) is in clinical development. Revenue from collaboration agreements were realized with the following partners:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
| Genmab, Denmark |
|
|
915 |
|
|
|
4,125 |
|
|
|
215 |
|
|
|
7,044 |
|
| BMS, United States |
|
|
21,439 |
|
|
|
12,107 |
|
|
|
31,935 |
|
|
|
110,532 |
|
| GSK, United Kingdom |
|
|
— |
|
|
|
983 |
|
|
|
— |
|
|
|
2,547 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| As of June 30, 2023, the Group has not recognized any milestone revenue under the collaboration agreements, due to the scientific uncertainty of achieving the milestones or the successful commercialization of a product. As of June 30, 2023, Immatics had not received any milestone or royalty payments in connection with the collaboration agreements. The Group expects to recognize the remaining deferred revenue balance as revenue as it performs the related performance obligations under each contract. The revenue for the three and six months ended June 30, 2023 from the collaboration agreement with BMS includes the Opt-in payment of €13.7 million. The remaining revenue for the three and six months ended June 30, 2023 from the collaboration with BMS is the revenue recognized over time on a basis. The revenue for the six months ended June 30, 2023 from the collaboration agreement with Genmab decreases in comparison to the revenue for the three months ended June 30, 2023, due to the negative revenue for the three months ended March 31, 2023, which is a result of changes to the inputs in the model resulting from an increase in the expected cost of the collaboration. The revenue from collaboration agreements with BMS includes the revenue regarding the license for IMA401 amounting to €91.3 million for the six months ended June 30, 2022. Deferred revenue related to the collaboration agreements consists of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Current |
|
|
67,997 |
|
|
|
64,957 |
|
| |
|
|
53,559 |
|
|
|
75,759 |
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
| Deferred revenues are contract liabilities within the scope of IFRS 15. The Group recognized expenses related to the amortization of capitalized cost of obtaining a contract of €0.1 million and €0.2 million for the three months ended June 30, 2023 and June 30, 2022. The Group recognized expenses related to the amortization of capitalized cost of obtaining a contract of €0.1 million and €0.3 million for the six months ended June 30, 2023 and June 30, 2022.
|
| X |
- DefinitionThe entire disclosure for revenue from contracts with customers. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Section Presentation
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IFRS15_g105-109__IFRS15_g105-109_TI
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Section Disclosure
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IFR15_g110-129_IFRS15_g110-129_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfRevenueFromContractsWithCustomersExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Financial result
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Financial result |
Other financial income and financial expenses consist of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Interest income |
|
|
2,744 |
|
|
|
69 |
|
|
|
4,999 |
|
|
|
75 |
|
| Foreign currency gains |
|
|
1,210 |
|
|
|
6,946 |
|
|
|
1,749 |
|
|
|
8,699 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Interest expenses |
|
|
(206 |
) |
|
|
(376 |
) |
|
|
(401 |
) |
|
|
(538 |
) |
| Foreign currency losses |
|
|
(805 |
) |
|
|
(31 |
) |
|
|
(4,119 |
) |
|
|
(986 |
) |
| Losses on financial instruments |
|
|
(133 |
) |
|
|
— |
|
|
|
(133 |
) |
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Change in fair value of liabilities for warrants |
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Interest income mainly results from short-term deposits as well as cash balances for the three and six months ended June 30, 2023. Interest expenses mainly results from leases. Foreign currency gains and losses mainly consist of realized and unrealized gains and losses in connection with our USD holdings of cash and cash equivalents, short-term deposits as well as bonds. The fair value of the warrants decreased from €2.35 ($2.51) per warrant as of December 31, 2022 to €1.32 ($1.44) as of March 31, 2023 and increased to €3.15 ($3.42) as of June 30, 2023. The result is an increase in fair value of warrant liabilities of €13.1 million for the three months ended June 30, 2023 and an increase in fair value of warrant liabilities of €5.7 million for the six months ended June 30, 2023. The fair value of the warrants decreased from €3.88 ($4.39) per warrant as of December 31, 2021 to €1.58 ($1.75) as of March 31, 2022 and increased to €1.96 ($2.04) as of June 30, 2022. The result is an increase in fair value of warrant liabilities of €2.8 million for the three months ended June 30, 2022 and a decrease in fair value of warrant liabilities of €13.7 million for the six months ended June 30, 2022.
|
| X |
- DefinitionThe disclosure of finance income (cost). [Refer: Finance income (cost)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfFinanceIncomeExpenseExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Income Tax
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Income Tax |
During the three and six months ended June 30, 2023, the Group generated a net loss. The Group correspondingly recognized no income tax expense and no equivalent current tax liability for the three and six months ended June 30, 2023. During the three months ended March 31, 2022, the Group generated a net income due to the recognition of revenue in connection with the license component of the BMS collaboration agreement on IMA401. This one-time revenue is not accounted for under German GAAP and consequently under German tax accounting. Instead, the Group recognizes revenue for the BMS agreement over the period of the clinical trial service. The deferred tax liability arising from the temporary difference related to delayed revenue recognition under German tax accounting is offset by deferred tax assets on tax losses carried forward that were previously not capitalized due to the Groups expectation of generating taxable losses in the foreseeable future. During the three and six months ended June 30, 2023 and 2022, the Group’s German operations were subject to a statutory tax rate of 28.5% and the Group´s U.S. operations were subject to a federal corporate income tax rate of 21%. For Immatics GmbH, the Group recognized an income tax expense and an equivalent current tax liability in the amount of €1.2 million for the three and six months ended June 30, 2022. The income tax expense is calculated based on taxable income of Immatics GmbH for the three and six months ended June 30, 2022 and does not take into account potential income or loss of the following quarters. The Group applied the estimated effective tax rate for the financial year 2022 to the taxable income for the three and six months ended June 30, 2022. Since no deferred tax assets have been recognized as of December 31, 2021, the Group took into account the tax losses carried forward that can be used to offset the taxable income generated in the three and six months ended June 30, 2022. In accordance with §10d para 2 EStG (German income tax code), 60% of an income of a given year can be offset with tax losses carried forward. Accordingly, 40% of the income before tax of Immatics GmbH are subject to income tax. As the profit generated in the three and six months ended June 30, 2022, is considered a one-time profit, no deferred tax assets exceeding the deferred tax liability for temporary differences have been recognized in respect of tax losses carried forward. The current assessment regarding the usability of deferred tax assets may change, depending on the Group’s taxable income in future years, which could result in the recognition of deferred tax assets. The Group continued to generate losses for all entities within the Group during the three and six months ended June 30, 2023 as well as for all entities apart from Immatics GmbH during the three and six months ended June 30, 2022. Due to changes in ownership in prior periods, there are certain limitations on tax losses carried forward for net operating losses incurred by Immatics US, Inc., under Section 382 of the U.S. Internal Revenue Code.
|
v3.23.2
Share-based payments
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Share-based payments |
Immatics N.V. has two share-based payment plans. In June 2020, Immatics N.V. established an initial equity incentive plan (“2020 Equity Plan”). At the Annual General Meeting on June 13, 2022, Immatics´s shareholders approved the Company’s 2022 stock option and incentive plan (“2022 Equity Plan”). The 2022 Equity Plan allows the company to grant additional options, other than that, it does not materially differ from the 2020 Equity Plan. Immatics GmbH previously issued share-based awards to employees under two different plans. Under the GmbH Stock Appreciation Program 2010 (the “2010 Plan”), the Company issued stock appreciation rights (“SARs”), which the Group accounted for as cash-settled awards. Under the Immatics Biotechnologies 2016 Equity Incentive Plan (“2016 Plan”), the Company issued tandem awards, which contained the possibility to function as either a SAR or a stock option. The Group accounted for awards issued under the 2016 Plan, which were redeemable in either cash or equity shares at the Group’s discretion, as equity settled. As part of the ARYA Merger, all outstanding awards under the 2010 Plan and 2016 Plan were replaced by a combination of cash payments and share-based awards under the 2020 Equity Plan in Immatics N.V. Under the 2020 Plan, management and employees have been granted different types of options, all of which are equity-settled transactions. As part of the replacement, active employees and management members received stock options (“Matching Stock Options”) to acquire shares in Immatics N.V. The Matching Stock Options have an exercise price of $10.00 and vested fully on July 31, 2021. The awards have a ten-year contract life. Matching Stock Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
Weighted average
exercise price in USD |
|
|
|
|
| Matching Stock Options outstanding on January 1, |
|
|
10.00 |
|
|
|
1,348,004 |
|
| Matching Stock Options forfeited |
|
|
— |
|
|
|
— |
|
| Matching Stock Options exercised |
|
|
10.00 |
|
|
|
720 |
|
| Matching Stock Options expired |
|
|
10.00 |
|
|
|
4,280 |
|
| Matching Stock Options outstanding on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Matching Stock Options exercisable on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Weighted average remaining contract life (years) |
|
|
7.01 |
|
|
|
|
| For any outstanding 2016 Plan and 2010 Plan awards scheduled to vest on or after January 1, 2021, employees received replacement stock options (“Converted Options”) to acquire shares in Immatics N.V. The Converted Options have comparable terms to previous awards, with revised exercise prices reflecting the reorganized capital structure of Immatics. The options granted under the 2020 Equity Plan that gives employees the right to acquire shares in Immatics N.V., are accounted for as a modification under IFRS 2, with the incremental fair value expensed over the remaining vesting period. The incremental fair value is the difference between the fair value of the options to purchase ordinary shares under the 2020 Equity Plan to acquire shares in Immatics N.V., and the fair value of the exchanged unvested SAR (both measured at the date on which the replacement award is issued). Converted Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Converted Options outstanding on January 1, |
|
|
2.74 |
|
|
|
525,181 |
|
| Converted Options forfeited |
|
|
1.14 |
|
|
|
909 |
|
| Converted Options exercised |
|
|
1.28 |
|
|
|
11,341 |
|
| Converted Options expired |
|
|
1.17 |
|
|
|
8 |
|
| Converted Options outstanding on June 30, |
|
|
2.78 |
|
|
|
512,923 |
|
| Converted Options exercisable on June 30, |
|
|
2.78 |
|
|
|
469,161 |
|
| Weighted average remaining contract life (years) |
|
|
4.51 |
|
|
|
|
| Under the 2020 Plan and the 2022 Plan, Immatics also issues employee stock options with a service requirement (“Service Options”), to acquire shares of Immatics N.V. The service-based options for employees including management will vest on a four-year time-based vesting schedule. Under the 2022 Plan, annual service options for members of the Board of Directors will vest entirely after one year. Service Options are granted on a recurring basis. The Company granted Service Options on April 3, 2023 and on June 27, 2023, which were accounted for using the respective grant date fair value. Immatics applied a Black Scholes pricing model to estimate the fair value of the Service Options, with a weighted average fair value of $6.97 for Service Options granted during the six months ended June 30, 2023.
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Exercise price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Underlying share price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Volatility |
|
|
84.84 |
% |
|
|
86.76 |
% |
| Time period (years) |
|
|
6.11 |
|
|
|
5.74 |
|
| Risk free rate |
|
|
3.47 |
% |
|
|
3.94 |
% |
| Dividend yield |
|
|
0.00 |
% |
|
|
0.00 |
% | Service Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Service Options outstanding on January 1, |
|
|
10.07 |
|
|
|
6,129,160 |
|
| Service Options granted in 2023 |
|
|
9.48 |
|
|
|
506,163 |
|
| Service Options forfeited |
|
|
9.80 |
|
|
|
145,494 |
|
| Service Options exercised |
|
|
9.51 |
|
|
|
2,707 |
|
| Service Options expired |
|
|
10.75 |
|
|
|
17,825 |
|
| Service Options outstanding on June 30, |
|
|
10.02 |
|
|
|
6,469,297 |
|
| Service Options exercisable on June 30, |
|
|
10.07 |
|
|
|
2,135,617 |
|
| Weighted average remaining contract life (years) |
|
|
8.57 |
|
|
|
|
| In addition, after the closing of the ARYA Merger certain executive officers and key personnel of the Group received under the 2020 Equity Plan performance-based options (“PSUs”), vesting based on both the achievement of market capitalization milestones and satisfaction of a four-year time-based vesting schedule. The PSUs are split into three equal tranches. The performance criteria for each of the three respective tranches requires Immatics to achieve a market capitalization of at least $1.5 billion, $2 billion and $3 billion, respectively. A Monte-Carlo simulation model has been used to measure the fair value at grant date of the PSUs. This model incorporates the impact of the performance criteria regarding market capitalization in the calculation of the award’s fair value at grant date. PSUs outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| PSUs outstanding on January 1, |
|
|
10.08 |
|
|
|
3,666,000 |
|
| PSUs granted in 2023 |
|
|
— |
|
|
|
— |
|
| PSUs forfeited |
|
|
10.00 |
|
|
|
12,000 |
|
| PSUs outstanding on June 30, |
|
|
10.08 |
|
|
|
3,654,000 |
|
| PSUs exercisable on June 30, |
|
|
— |
|
|
|
— |
|
| Weighted average remaining contract life (years) |
|
|
7.05 |
|
|
|
|
| The Group recognized total employee-related share-based compensation expenses from all plans, during the three and six months ended June 30, 2023 and 2022 as set out below:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Research and development expenses |
|
|
(3,282 |
) |
|
|
(3,107 |
) |
|
|
(6,815 |
) |
|
|
(6,375 |
) |
| General and administrative expenses |
|
|
(2,231 |
) |
|
|
(2,453 |
) |
|
|
(4,800 |
) |
|
|
(4,887 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| X |
- DefinitionThe entire disclosure for share-based payment arrangements. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 44
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_44&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfSharebasedPaymentArrangementsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Other current and non-current assets
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Other current and non-current assets |
9. Other current and non-current assets Other current assets consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Prepaid expenses |
|
|
9,546 |
|
|
|
10,450 |
|
| Value added tax receivables |
|
|
1,605 |
|
|
|
1,031 |
|
| Other assets |
|
|
5,517 |
|
|
|
2,357 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Prepaid expenses include expenses for licenses and software of €6.7 million as of June 30, 2023 and €7.4 million as of December 31, 2022 and prepaid insurance expenses of €0.2 million as of June 30, 2023 and €1.2 million as of December 31, 2022. The Group accrued €0.3 million as of June 30, 2023 and €0.4 million as of December 31, 2022 of incremental cost for the successful arrangement of the BMS collaboration signed in 2019 and the Genmab collaboration agreement. Additionally, prepaid expenses include expenses for maintenance of €0.7 million as of June 30, 2023 and €0.7 million as of December 31, 2022. The remaining amount is mainly related to prepaid expenses for contract research organizations and travel expenses. Other assets include receivables from lease incentive, capital gains tax and prepaid deposit expenses. Other non-current assets consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Prepaid expenses |
|
|
1,365 |
|
|
|
1,906 |
|
| Other assets |
|
|
607 |
|
|
|
639 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Prepaid expenses include the non-current portion of prepayments for licensing agreements of €1.1 million as of June 30, 2023 and €1.5 million as of December 31, 2022 , prepaid maintenance expenses of €0.2 million as of June 30, 2023 and €0.3 million as of December 31, 2022 and accrued incremental cost of the BMS and Genmab collaboration agreement of €0.1 million as of June 30, 2023 and €0.1 million as of December 31, 2022. Other assets include the non-current portion for prepaid deposit expenses. |
| X |
- DefinitionThe disclosure of other assets. [Refer: Other assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfOtherAssetsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Property, plant and equipment, intangible assets and Right-of-use assets
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Property, plant and equipment, intangible assets and Right-of-use assets |
10. Property, plant and equipment, intangible assets and assets During the three months ended June 30, 2023 and June 30, 2022, the Group acquired property, plant and equ ip ment and intangible assets in the amount of €11.6 million and €1.1 million, respectively. During the six months ended June 30, 2023 and June 30, 2022, the Group acquired property, plant and equipment and intangible assets in the amount of €15.7 million and €2.2 million, respectively. The Group’s additions include leasehold improvements for the research and commercial GMP manufacturing facility construction in Houston, Texas of €9.8 million for the six months ended June 30, 2023. During the six months ended June 30, 2023, a new office space in Tübingen, Germany, lease term extensions and rent increases to existing lease agreements resulted in an addition in assets and corresponding lease liability in the amount of €3.8 million. |
| X |
- DefinitionDisclosure of Property, plant and equipment, intangible assets and Right-of-use assets explanatory.
| Name: |
imtx_DisclosureOfPropertyPlantAndEquipmentIntangibleAssetsAndRightofuseAssetsExplanatory |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Provisions
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Provisions |
Provisions consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Provision for bonuses |
|
|
3,117 |
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| These amounts include provisions for the Group’s annual employee bonuses.
|
| X |
- DefinitionDisclosure In Entirely Of Provisions.
| Name: |
imtx_DisclosureInEntirelyOfProvisionsTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Accounts payables
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Accounts payables |
Accounts payables consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Accounts payables |
|
|
5,473 |
|
|
|
4,025 |
|
| Accrued liabilities |
|
|
14,431 |
|
|
|
9,031 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| X |
- DefinitionThe disclosure of trade and other payables. [Refer: Trade and other payables] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfTradeAndOtherPayablesExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Other current liabilities
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Other current liabilities |
13. Other current liabilities Other current liabilities consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Income tax liability |
|
|
4,298 |
|
|
|
4,298 |
|
| Payroll tax |
|
|
1,840 |
|
|
|
3,426 |
|
| Accrual for vacation |
|
|
1,400 |
|
|
|
806 |
|
| Accrued bonuses |
|
|
— |
|
|
|
680 |
|
| Other liabilities |
|
|
391 |
|
|
|
156 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Other current liabilities are non-interest-bearing and are due within one year. The carrying amounts of other current liabilities represents fair values due to their short-term nature. |
| X |
- DefinitionThe disclosure of other current liabilities. [Refer: Other current liabilities] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfOtherCurrentLiabilitiesExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Shareholders' equity
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Shareholders' equity |
As of June 30, 2023 and December 31, 2022, the total number of ordinary shares of Immatics N.V. outstanding is 80,397,851 and 76,670,699 with a par value of €0.01, respectively. During the three months ended June 30, 2023, the Group issued 3.7 million shares under the ATM agreement with SVB Securities LLC and collected a gross amount of €38.6 million less transaction costs of €1.2 million, resulting in an increase in share capital of €37 thousand and share premium of €37.4 million. Additionally, the number of ordinary shares increased during the six months ended June 30, 2023, due to exercised share options from the Group’s equity incentive plan. Other reserves are related to accumulated foreign currency translation amounts associated with the Group’s U.S. operations.
|
| X |
- DefinitionThe entire disclosure for share capital, reserves and other equity interest. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 79
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_79&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfShareCapitalReservesAndOtherEquityInterestExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Related party disclosures
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Related party disclosures |
15. Related party disclosures During the three and six months ended June 30, 2023, the Group did not enter into any new related-party transactions with its key management personnel or with related entities other than the granting of a total of 175,000 Service options to its Board of Directors for the three and six months ended June 30, 2023. |
v3.23.2
Financial Instruments
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Financial Instruments |
16. Financial Instruments Set out below are the carrying amounts and fair values of the Group’s financial instruments that are carried in the interim condensed consolidated financial statements.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Carrying amount per measurement category |
|
| |
|
|
|
|
|
|
|
|
At fair value
through profit
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
130,405 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
217,222 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
330 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
5,158 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
19,212 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
58 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
22,622 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
16,822 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Carrying amount per measurement category |
|
|
|
|
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
148,519 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
154,930 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
58,756 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
1,111 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
2,402 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
11,735 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
54 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
16,914 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
14,563 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| * |
“Short-term deposits” and “Bonds” are classified within the balance sheet item “Other financial assets” | In all valuation categories with the exception of Bonds, the carrying amount represents a reasonable approximation of the fair value based on the short-term maturities of these instruments. Set out below are the carrying amounts and fair values of the Group’s Bonds as of June 30, 2023 and December 31, 2022. The fair values of the financial assets and liabilities are included at the amount at which the instrument could be exchanged in a current transaction between willing parties, other than in a forced or liquidation sale.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bonds |
|
|
— |
|
|
|
— |
|
|
|
58,756 |
|
|
|
58,300 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| The following methods and assumptions were used to estimate the fair values: All financial assets are categorized based on Level 1 inputs and are therefore valued using quoted (unadjusted) market prices. All financial liabilities are also categorized based on Level 1 inputs. The bonds’ contractual cash flows represent solely payments of principal and interest and Immatics intends to hold the bonds to collect the contractual cash flows. The Group therefore accounts for the bonds as a financial asset at amortized cost. Bonds are classified as Level 1 of the fair value hierarchy, as they are listed on publicly traded markets. Liabilities for warrants are comprised of the Immatics Warrants issued to investors with a cashless exercise mechanism as a current liability which the Company accounted for according to provisions of IAS 32. The Company measures the warrants at fair value by using the closing price of warrants at Nasdaq. The warrants are measured in each reporting period. Changes in the fair value are recognized in the Company’s Consolidated Statement of Profit/(Loss) as financial income or expenses, as appropriate. The warrants are classified as Level 1 of the fair value hierarchy. The maturity of the liabilities for warrants is dependent on the development of the share price as well as the decisions by the Immatics Warrants holders.
|
| X |
- DefinitionThe entire disclosure for financial instruments. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Section Scope
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IFRS07_g3-5A__IFRS07_g3-5A_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfFinancialInstrumentsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Earnings and Loss per Share
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Earnings and Loss per Share |
17. Earnings and Loss per Share The Group reported basic and diluted loss and earnings per share during the three and six months ended June 30, 2023 and 2022. Basic and diluted loss per share and basic earnings per share are calculated by dividing the net profit or loss by the weighted-average number of ordinary shares outstanding for the reporting period. Diluted earnings per share for the six months ended June 30, 2022, are calculated by adjusting the weighted-average number of ordinary shares outstanding for any dilutive effects resulting from equity awards granted to the Board and employees of the Group as well as from publicly traded Immatics Warrants. The Group’s equity awards and Immatics Warrants for which the exercise price is exceeding the Group’s weighted average share price for the six months ended June 30, 2022, are anti-dilutive instruments and are excluded in the calculation of diluted weighted average number of ordinary shares. The Group was loss-making during the three and six months ended June 30, 2023 and during the three months ended June 30, 2022, therefore all instruments are anti-dilutive instruments and are excluded in the calculation of diluted weighted average number of ordinary shares outstanding, including the outstanding equity awards and the 7,187,500 Immatics Warrants issued in 2020 and outstanding as of June 30, 2023.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
(Euros in thousands, except share and per share data) |
|
|
(Euros in thousands, except share and per share data) |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
| Diluted |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
Weighted average shares outstanding: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
63,932,449 |
|
| Diluted |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
64,477,256 |
|
|
| X |
- DefinitionThe entire disclosure for earnings per share. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Section Disclosure
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IAS33_g70-73A__IAS33_g70-73A_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfEarningsPerShareExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Events occurring after the reporting period
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Events occurring after the reporting period |
18. Events occurring after the reporting period The Group evaluated subsequent events for recognition or disclosure through August 17, 2023. On July 19, 2023, the Group closed a private placement transaction of 2,419,818 ordinary shares with a subscription price of $14.46 per ordinary share with BMS. The Group received gross proceeds of approximately $35 million, before deducting transaction expenses and intends to use the net proceeds to fund the continued research and development of the Group’s pipeline, the manufacturing and production of product candidates and for working capital. After the reporting period, the Group issued 1.8 million shares under the ATM agreement with SVB Securities LLC and collected a gross amount of €20.4 million ($22.2 million).
|
| X |
- DefinitionThe entire disclosure for events after the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 10
-IssueDate 2022-03-24
-Section Disclosure
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=10&code=ifrs-tx-2022-en-r&doctype=Standard&dita_xref=IAS10_g17-22_IAS10_g17-22_TI
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfEventsAfterReportingPeriodExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Significant accounting policies (Policies)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Basis of presentation |
The interim condensed consolidated financial statements of the Group as of June 30, 2023 and for the three and six months ended June 30, 2023 and 2022 have been prepared on a going concern basis in accordance with International Accounting Standard 34 (“Interim Financial Reporting”), as issued by the International Accounting Standards Board (“IASB”) and have not been audited or reviewed by a statutory auditor. In accordance with IAS 34, the interim condensed consolidated financial statements do not include all the information and disclosures required in the annual financial statements and should be read in conjunction with the Group’s annual financial statements for the year ended December 31, 2022, which have been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the IASB, taking into account the recommendations of the International Financial Reporting Standards Interpretations Committee (“IFRS IC”). In these condensed notes to the consolidated financial statements, information is provided primarily on the items for which there have been significant changes compared with the consolidated financial statements of the Group for fiscal year 2022. The interim condensed consolidated financial statements are presented in Euros. Amounts are stated in thousands of Euros, unless otherwise indicated. For technical reasons, the information provided in these financial statements may contain rounding differences of +/- one unit. The accounting policies adopted in the preparation of the interim condensed consolidated financial statements are consistent with those followed in the preparation of the Group’s annual consolidated financial statements for the year ended December 31, 2022. The new and amended standards and interpretations applicable for the first time as of January 1, 2023, as disclosed in the notes to the consolidated financial statements for the year ended December 31, 2022, had no impact on the interim condensed consolidated financial statements of the Group for the three and six months ended June 30, 2023. Estimates and assumptions have to be made in the interim consolidated financial statements as of June 30, 2023. These have an impact on the amount and disclosure of the recognized assets and liabilities, income and expenses, and contingent liabilities. The estimates and judgments are basically unchanged from the circumstances described in the consolidated financial statements of the Group for the fiscal year 2022. Developments deviating from this may result in the amounts that arise deviating from the original estimates. These possible developments are outside the sphere of influence of the management.
|
| Revision of previously issued financial statements |
Revision of previously issued financial statements During the preparation of the unaudited interim consolidated financial statements for the three and nine months ended September 30, 2022, the Group identified an error in the presentation of ‘Net foreign exchange differences’ and ‘Effects of exchange rate changes on cash and cash equivalents’ in the statement of cash flows. The error resulted in a presentation of effects from exchange rate changes on non-functional currency denominated cash and cash equivalents in Immatics N.V. and Immatics GmbH as operating cash flow instead of the presentation as non-cash items in effects of exchange rate changes on cash and cash equivalents. This error had no impact on the Company’s consolidated statements of financial position, of profit/(loss), of comprehensive income/(loss) and of consolidated statements of changes in equity. The Company assessed the materiality of these errors on the previously issued consolidated financial statements and concluded that the errors were not material to any period presented. The impact of the revision of the previously issued financial statements is as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, 2022 |
|
| |
|
|
|
|
|
|
|
|
|
Net foreign exchange differences and expected credit losses |
|
|
115 |
|
|
|
(7,949 |
) |
|
|
(7,834 |
) |
Net cash provided by/(used in) operating activities |
|
|
163,722 |
|
|
|
(7,949 |
) |
|
|
155,773 |
|
Net cash (used in)/provided by investing activities |
|
|
(48,528 |
) |
|
|
— |
|
|
|
(48,528 |
) |
Net cash (used in)/provided by financing activities |
|
|
15,203 |
|
|
|
— |
|
|
|
15,203 |
|
Net increase/(decrease) in cash and cash equivalents |
|
|
130,397 |
|
|
|
(7,949 |
) |
|
|
122,448 |
|
Cash and cash equivalents at beginning of period |
|
|
132,994 |
|
|
|
— |
|
|
|
132,994 |
|
Effects of exchange rate changes on cash and cash equivalents |
|
|
1,734 |
|
|
|
7,949 |
|
|
|
9,683 |
|
Cash and cash equivalents at end of period |
|
|
265,125 |
|
|
|
— |
|
|
|
265,125 |
|
|
| X |
- DefinitionDescription of accounting policy for basis of presentation explanatory.
| Name: |
imtx_DescriptionOfAccountingPolicyForBasisOfPresentationExplanatoryPolicyTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDescription Of accounting policy for revision of previously issued financial statements explanatory.
| Name: |
imtx_DescriptionOfAccountingPolicyForRevisionOfPreviouslyIssuedFinancialStatementsExplanatoryPolicyTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Significant accounting policies (Table)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Summary of impact revision to previously issued financial statements |
The impact of the revision of the previously issued financial statements is as follows:
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Six months ended June 30, 2022 |
|
| |
|
|
|
|
|
|
|
|
|
Net foreign exchange differences and expected credit losses |
|
|
115 |
|
|
|
(7,949 |
) |
|
|
(7,834 |
) |
Net cash provided by/(used in) operating activities |
|
|
163,722 |
|
|
|
(7,949 |
) |
|
|
155,773 |
|
Net cash (used in)/provided by investing activities |
|
|
(48,528 |
) |
|
|
— |
|
|
|
(48,528 |
) |
Net cash (used in)/provided by financing activities |
|
|
15,203 |
|
|
|
— |
|
|
|
15,203 |
|
Net increase/(decrease) in cash and cash equivalents |
|
|
130,397 |
|
|
|
(7,949 |
) |
|
|
122,448 |
|
Cash and cash equivalents at beginning of period |
|
|
132,994 |
|
|
|
— |
|
|
|
132,994 |
|
Effects of exchange rate changes on cash and cash equivalents |
|
|
1,734 |
|
|
|
7,949 |
|
|
|
9,683 |
|
Cash and cash equivalents at end of period |
|
|
265,125 |
|
|
|
— |
|
|
|
265,125 |
|
|
| X |
- DefinitionThe description of the nature of accounting errors in prior periods. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 8
-IssueDate 2022-03-24
-Paragraph 49
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=8&code=ifrs-tx-2022-en-r&anchor=para_49_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfNatureAmountAndCorrectionOfAccountingErrorsInPriorPeriodsEstimate |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Revenue from collaboration agreements (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Summary of Deferred revenue related to the collaboration agreements |
Deferred revenue related to the collaboration agreements consists of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Current |
|
|
67,997 |
|
|
|
64,957 |
|
| |
|
|
53,559 |
|
|
|
75,759 |
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
| Summary of revenue from collaboration agreements explanatory |
Revenue from collaboration agreements were realized with the following partners:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
| Genmab, Denmark |
|
|
915 |
|
|
|
4,125 |
|
|
|
215 |
|
|
|
7,044 |
|
| BMS, United States |
|
|
21,439 |
|
|
|
12,107 |
|
|
|
31,935 |
|
|
|
110,532 |
|
| GSK, United Kingdom |
|
|
— |
|
|
|
983 |
|
|
|
— |
|
|
|
2,547 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| X |
- DefinitionDisclosure of contract liabilities.
| Name: |
imtx_DisclosureOfContractLiabilitiesTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of revenue from collaboration agreements explanatory.
| Name: |
imtx_DisclosureOfRevenueFromCollaborationAgreementsExplanatory |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Financial result (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Summary of other financial income and financial expenses |
Other financial income and financial expenses consist of the following:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Interest income |
|
|
2,744 |
|
|
|
69 |
|
|
|
4,999 |
|
|
|
75 |
|
| Foreign currency gains |
|
|
1,210 |
|
|
|
6,946 |
|
|
|
1,749 |
|
|
|
8,699 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Interest expenses |
|
|
(206 |
) |
|
|
(376 |
) |
|
|
(401 |
) |
|
|
(538 |
) |
| Foreign currency losses |
|
|
(805 |
) |
|
|
(31 |
) |
|
|
(4,119 |
) |
|
|
(986 |
) |
| Losses on financial instruments |
|
|
(133 |
) |
|
|
— |
|
|
|
(133 |
) |
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Change in fair value of liabilities for warrants |
|
|
(13,105 |
) |
|
|
(2,786 |
) |
|
|
(5,708 |
) |
|
|
13,743 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
v3.23.2
Share-based payments (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Statements [Line Items] |
|
| Summary of Employee Related Share Based Compensation Expense |
The Group recognized total employee-related share-based compensation expenses from all plans, during the three and six months ended June 30, 2023 and 2022 as set out below:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Research and development expenses |
|
|
(3,282 |
) |
|
|
(3,107 |
) |
|
|
(6,815 |
) |
|
|
(6,375 |
) |
| General and administrative expenses |
|
|
(2,231 |
) |
|
|
(2,453 |
) |
|
|
(4,800 |
) |
|
|
(4,887 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Matching Stock Options [Member] |
|
| Statements [Line Items] |
|
| Summary of Employee Share Options |
Matching Stock Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
Weighted average
exercise price in USD |
|
|
|
|
| Matching Stock Options outstanding on January 1, |
|
|
10.00 |
|
|
|
1,348,004 |
|
| Matching Stock Options forfeited |
|
|
— |
|
|
|
— |
|
| Matching Stock Options exercised |
|
|
10.00 |
|
|
|
720 |
|
| Matching Stock Options expired |
|
|
10.00 |
|
|
|
4,280 |
|
| Matching Stock Options outstanding on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Matching Stock Options exercisable on June 30, |
|
|
10.00 |
|
|
|
1,343,004 |
|
| Weighted average remaining contract life (years) |
|
|
7.01 |
|
|
|
|
|
|
| Converted Options [Member] |
|
| Statements [Line Items] |
|
| Summary of Employee Share Options |
Converted Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Converted Options outstanding on January 1, |
|
|
2.74 |
|
|
|
525,181 |
|
| Converted Options forfeited |
|
|
1.14 |
|
|
|
909 |
|
| Converted Options exercised |
|
|
1.28 |
|
|
|
11,341 |
|
| Converted Options expired |
|
|
1.17 |
|
|
|
8 |
|
| Converted Options outstanding on June 30, |
|
|
2.78 |
|
|
|
512,923 |
|
| Converted Options exercisable on June 30, |
|
|
2.78 |
|
|
|
469,161 |
|
| Weighted average remaining contract life (years) |
|
|
4.51 |
|
|
|
|
|
|
| Additional Grants Under 2020 Plan [Member] |
|
| Statements [Line Items] |
|
| Summary of Options Granted Priced Using Black Scholes for SARs, Tandem Awards, Converted Options, Matching Stock Options and Monte Carlo Option Pricing Model |
Immatics applied a Black Scholes pricing model to estimate the fair value of the Service Options
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
| Exercise price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Underlying share price in USD |
|
$ |
6.89 |
|
|
$ |
11.41 |
|
| Volatility |
|
|
84.84 |
% |
|
|
86.76 |
% |
| Time period (years) |
|
|
6.11 |
|
|
|
5.74 |
|
| Risk free rate |
|
|
3.47 |
% |
|
|
3.94 |
% |
| Dividend yield |
|
|
0.00 |
% |
|
|
0.00 |
% |
|
| Summary of Employee Share Options |
Service Options outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| Service Options outstanding on January 1, |
|
|
10.07 |
|
|
|
6,129,160 |
|
| Service Options granted in 2023 |
|
|
9.48 |
|
|
|
506,163 |
|
| Service Options forfeited |
|
|
9.80 |
|
|
|
145,494 |
|
| Service Options exercised |
|
|
9.51 |
|
|
|
2,707 |
|
| Service Options expired |
|
|
10.75 |
|
|
|
17,825 |
|
| Service Options outstanding on June 30, |
|
|
10.02 |
|
|
|
6,469,297 |
|
| Service Options exercisable on June 30, |
|
|
10.07 |
|
|
|
2,135,617 |
|
| Weighted average remaining contract life (years) |
|
|
8.57 |
|
|
|
|
|
|
| Performance Based Options [Member] |
|
| Statements [Line Items] |
|
| Summary of Employee Share Options |
PSUs outstanding as of June 30, 2023:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| PSUs outstanding on January 1, |
|
|
10.08 |
|
|
|
3,666,000 |
|
| PSUs granted in 2023 |
|
|
— |
|
|
|
— |
|
| PSUs forfeited |
|
|
10.00 |
|
|
|
12,000 |
|
| PSUs outstanding on June 30, |
|
|
10.08 |
|
|
|
3,654,000 |
|
| PSUs exercisable on June 30, |
|
|
— |
|
|
|
— |
|
| Weighted average remaining contract life (years) |
|
|
7.05 |
|
|
|
|
|
|
| X |
- DefinitionThe disclosure of information about indirect, by reference to the fair value of the equity instruments granted, measurement of the fair value of goods or services received as consideration for the entity's share options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfIndirectMeasurementOfFairValueOfGoodsOrServicesReceivedShareOptionsGrantedDuringPeriodExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe disclosure of the range of exercise prices for outstanding share options. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfRangeOfExercisePricesOfOutstandingShareOptionsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of detailed information about share based compensation expense.
| Name: |
imtx_DisclosureOfDetailedInformationAboutShareBasedCompensationExpenseTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
imtx_StatementsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_MatchingStockOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_ConvertedOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_AdditionalGrantsUnderTwoThousandAndTwentyPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe disclosure of other current assets. [Refer: Other current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfOtherCurrentAssetsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe disclosure of other non-current assets. [Refer: Other non-current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfOtherNoncurrentAssetsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
| X |
- DefinitionThe disclosure of provisions. [Refer: Provisions] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 10
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_10_e&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DisclosureOfProvisionsExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
| X |
- DefinitionDisclosure of accounts payable explanatory.
| Name: |
imtx_DisclosureOfAccountsPayableExplanatoryTableTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Other current liabilities (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Summary of other current liabilities |
Other current liabilities consist of the following:
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
| |
|
|
|
| Income tax liability |
|
|
4,298 |
|
|
|
4,298 |
|
| Payroll tax |
|
|
1,840 |
|
|
|
3,426 |
|
| Accrual for vacation |
|
|
1,400 |
|
|
|
806 |
|
| Accrued bonuses |
|
|
— |
|
|
|
680 |
|
| Other liabilities |
|
|
391 |
|
|
|
156 |
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| X |
- DefinitionDisclosure of detailed information about other current liabilities abstract.
| Name: |
imtx_DisclosureOfDetailedInformationAboutOtherCurrentLiabilitiesTextBlock |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Financial Instruments (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Text Block [Abstract] |
|
| Summary of Carrying Amounts and Fair values of Group's Financial Instruments |
Set out below are the carrying amounts and fair values of the Group’s financial instruments that are carried in the interim condensed consolidated financial statements.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Carrying amount per measurement category |
|
| |
|
|
|
|
|
|
|
|
At fair value
through profit
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
130,405 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
217,222 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
330 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
5,158 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
19,212 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
58 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
22,622 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
16,822 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Carrying amount per measurement category |
|
|
|
|
|
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
At fair value
through profit
and loss |
|
|
|
|
|
|
|
Current/non-current assets |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents |
|
|
— |
|
|
|
148,519 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Short-term deposits* |
|
|
— |
|
|
|
154,930 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Bonds* |
|
|
— |
|
|
|
58,756 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Accounts receivables |
|
|
— |
|
|
|
1,111 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Other current/non-current assets |
|
|
— |
|
|
|
2,402 |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
Current/non-current liabilities |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts payable |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
11,735 |
|
|
|
|
|
Other current liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
54 |
|
|
|
|
|
Liabilities for warrants |
|
|
— |
|
|
|
— |
|
|
|
16,914 |
|
|
|
— |
|
|
|
|
|
Lease liabilities |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
14,563 |
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| * |
“Short-term deposits” and “Bonds” are classified within the balance sheet item “Other financial assets” | In all valuation categories with the exception of Bonds, the carrying amount represents a reasonable approximation of the fair value based on the short-term maturities of these instruments. Set out below are the carrying amounts and fair values of the Group’s Bonds as of June 30, 2023 and December 31, 2022. The fair values of the financial assets and liabilities are included at the amount at which the instrument could be exchanged in a current transaction between willing parties, other than in a forced or liquidation sale.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Bonds |
|
|
— |
|
|
|
— |
|
|
|
58,756 |
|
|
|
58,300 |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|
|
— |
|
|
|
|
|
|
|
|
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
v3.23.2
Earnings and Loss per Share (Tables)
|
6 Months Ended |
Jun. 30, 2023 |
|---|
| Earnings per share [abstract] |
|
| Summary of Earnings and loss per share |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
Three months ended June 30, |
|
|
Six months ended June 30, |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
| |
|
(Euros in thousands, except share and per share data) |
|
|
(Euros in thousands, except share and per share data) |
|
| |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.12 |
|
| Diluted |
|
|
(0.32 |
) |
|
|
(0.22 |
) |
|
|
(0.58 |
) |
|
|
1.11 |
|
Weighted average shares outstanding: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Basic |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
63,932,449 |
|
| Diluted |
|
|
77,311,053 |
|
|
|
64,915,600 |
|
|
|
76,994,713 |
|
|
|
64,477,256 |
|
|
| X |
- References
+ Details
| Name: |
ifrs-full_EarningsPerShareAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe disclosure of earnings per share. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_EarningsPerShareExplanatory |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:textBlockItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
| X |
- DefinitionThe date on which financial statements are authorised for issue. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 10
-IssueDate 2022-03-24
-Paragraph 17
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=10&code=ifrs-tx-2022-en-r&anchor=para_17&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DateOfAuthorisationForIssueOfFinancialStatements2013 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe date of the end of the reporting period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 51
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_51_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DateOfEndOfReportingPeriod2013 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfNonadjustingEventsAfterReportingPeriodLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_NonadjustingEventsAfterReportingPeriodAxis=imtx_AuthorizationOfFinancialStatementsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe amount of revenue from contracts with customers. A customer is a party that has contracted with an entity to obtain goods or services that are an output of the entity’s ordinary activities in exchange for consideration. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 114
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_114&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_113_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_RevenueFromContractsWithCustomers |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionDisclosure of changes in accounting policies accounting estimates and errors.
| Name: |
imtx_DisclosureOfChangesInAccountingPoliciesAccountingEstimatesAndErrorsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionProceeds from contractual option exercised.
| Name: |
imtx_ProceedsFromContractualOptionExercised |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_BMSOptInAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Significant accounting policies - Summary of impact revision to previously issued financial statements (Detail) - EUR (€)
€ in Thousands |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Disclosure of expected impact of initial application of new standards or interpretations [line items] |
|
|
| Net foreign exchange differences and expected credit losses |
€ 4,081
|
€ (7,834)
|
| Net cash provided by/(used in) operating activities |
(30,323)
|
155,773
|
| Net cash provided by/(used in) investing activities |
(20,555)
|
(48,528)
|
| Net cash (used in)/provided by financing activities |
35,585
|
15,203
|
| Net increase/(decrease) in cash and cash equivalents |
(15,293)
|
122,448
|
| Cash and cash equivalents at beginning |
148,519
|
132,994
|
| Effects of exchange rate changes on cash and cash equivalents |
(2,821)
|
9,683
|
| Cash and cash equivalents at end |
€ 130,405
|
265,125
|
| As reported |
|
|
| Disclosure of expected impact of initial application of new standards or interpretations [line items] |
|
|
| Net foreign exchange differences and expected credit losses |
|
115
|
| Net cash provided by/(used in) operating activities |
|
163,722
|
| Net cash provided by/(used in) investing activities |
|
(48,528)
|
| Net cash (used in)/provided by financing activities |
|
15,203
|
| Net increase/(decrease) in cash and cash equivalents |
|
130,397
|
| Cash and cash equivalents at beginning |
|
132,994
|
| Effects of exchange rate changes on cash and cash equivalents |
|
1,734
|
| Cash and cash equivalents at end |
|
265,125
|
| Adjustment |
|
|
| Disclosure of expected impact of initial application of new standards or interpretations [line items] |
|
|
| Net foreign exchange differences and expected credit losses |
|
(7,949)
|
| Net cash provided by/(used in) operating activities |
|
(7,949)
|
| Net cash provided by/(used in) investing activities |
|
0
|
| Net cash (used in)/provided by financing activities |
|
0
|
| Net increase/(decrease) in cash and cash equivalents |
|
(7,949)
|
| Cash and cash equivalents at beginning |
|
0
|
| Effects of exchange rate changes on cash and cash equivalents |
|
7,949
|
| Cash and cash equivalents at end |
|
0
|
| As revised |
|
|
| Disclosure of expected impact of initial application of new standards or interpretations [line items] |
|
|
| Net foreign exchange differences and expected credit losses |
|
(7,834)
|
| Net cash provided by/(used in) operating activities |
|
155,773
|
| Net cash provided by/(used in) investing activities |
|
(48,528)
|
| Net cash (used in)/provided by financing activities |
|
15,203
|
| Net increase/(decrease) in cash and cash equivalents |
|
122,448
|
| Cash and cash equivalents at beginning |
|
132,994
|
| Effects of exchange rate changes on cash and cash equivalents |
|
9,683
|
| Cash and cash equivalents at end |
|
€ 265,125
|
| X |
- DefinitionThe amount of cash on hand and demand deposits, along with short-term, highly liquid investments that are readily convertible to known amounts of cash and that are subject to an insignificant risk of changes in value. [Refer: Cash; Cash equivalents] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 45
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_45&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B13
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B13_a&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashAndCashEquivalents |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash flows from (used in) financing activities, which are activities that result in changes in the size and composition of the contributed equity and borrowings of the entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInFinancingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash flows from (used in) investing activities, which are the acquisition and disposal of long-term assets and other investments not included in cash equivalents. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInInvestingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe cash flows from (used in) operating activities, which are the principal revenue-producing activities of the entity and other activities that are not investing or financing activities. [Refer: Revenue] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 10
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_10&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 50
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_50_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CashFlowsFromUsedInOperatingActivities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DescriptionOfExpectedImpactOfInitialApplicationOfNewStandardsOrInterpretationsLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) in cash and cash equivalents before the effect of exchange rate changes on cash and cash equivalents held in foreign currencies. [Refer: Cash and cash equivalents; Effect of exchange rate changes on cash and cash equivalents] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 45
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_45&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IncreaseDecreaseInCashAndCashEquivalentsBeforeEffectOfExchangeRateChanges |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionAdjustments for unrealised foreign exchange and expected credit loss.
| Name: |
imtx_AdjustmentsForUnrealisedForeignExchangeAndExpectedCreditLoss |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionEffects of exchange rate changes and expected credit losses on cash and cash equivalents.
| Name: |
imtx_EffectsOfExchangeRateChangesAndExpectedCreditLossesOnCashAndCashEquivalents |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
srt_RestatementAxis=srt_ScenarioPreviouslyReportedMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RestatementAxis=srt_RevisionOfPriorPeriodErrorCorrectionAdjustmentMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
srt_RestatementAxis=imtx_CurrentlyReportedMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Revenue from collaboration agreements - Additional Information (Detail) - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Disclosure Of Revenue From Contract With Customers [Line Items] |
|
|
|
|
| Amortization of contract costs capitalized |
€ 100
|
€ 200
|
€ 100
|
€ 300
|
| Receipt of upfront of payment |
22,354
|
€ 17,215
|
32,150
|
€ 120,123
|
| BMS Collaboration Agreement [Member] |
|
|
|
|
| Disclosure Of Revenue From Contract With Customers [Line Items] |
|
|
|
|
| Proceeds from option exercise fee under collaborative arrangement |
13,700
|
|
13,700
|
|
| BMS Collaboration Agreement [Member] | IMA401 [Member] |
|
|
|
|
| Disclosure Of Revenue From Contract With Customers [Line Items] |
|
|
|
|
| Receipt of upfront of payment |
|
|
91,300
|
|
| Collaboration Agreement [Member] |
|
|
|
|
| Disclosure Of Revenue From Contract With Customers [Line Items] |
|
|
|
|
| Milestone or royalty payment receivable |
€ 0
|
|
€ 0
|
|
| X |
- DefinitionThe amount of amortisation for assets recognised from the costs incurred to obtain or fulfil contracts with customers. [Refer: Assets recognised from costs to obtain or fulfil contracts with customers; Amortisation expense] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 128
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_128_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AmortisationAssetsRecognisedFromCostsIncurredToObtainOrFulfilContractsWithCustomers |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
imtx_DisclosureOfRevenueFromContractWithCustomersLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionMilestone or royalty payment receivable.
| Name: |
imtx_MilestoneOrRoyaltyPaymentReceivable |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionProceeds from option exercise fee under collaborative arrangement.
| Name: |
imtx_ProceedsFromOptionExerciseFeeUnderCollaborativeArrangement |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionRevenue from collaboration agreements
| Name: |
imtx_RevenueFromCollaborationAgreements |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_BMSCollaborationAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ProductsAndServicesAxis=imtx_Ima401Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_CollaborationAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Revenue from collaboration agreements - Summary of revenue from collaboration agreements (Detail) - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Disclosure Of Revenue From Collaboration Agreements [Line Items] |
|
|
|
|
| Revenue from collaboration agreements |
€ 22,354
|
€ 17,215
|
€ 32,150
|
€ 120,123
|
| Total |
|
|
|
|
| Disclosure Of Revenue From Collaboration Agreements [Line Items] |
|
|
|
|
| Revenue from collaboration agreements |
22,354
|
17,215
|
32,150
|
120,123
|
| Denmark | Genmab |
|
|
|
|
| Disclosure Of Revenue From Collaboration Agreements [Line Items] |
|
|
|
|
| Revenue from collaboration agreements |
915
|
4,125
|
215
|
7,044
|
| United States | BMS |
|
|
|
|
| Disclosure Of Revenue From Collaboration Agreements [Line Items] |
|
|
|
|
| Revenue from collaboration agreements |
21,439
|
12,107
|
31,935
|
110,532
|
| United Kingdom | GSK |
|
|
|
|
| Disclosure Of Revenue From Collaboration Agreements [Line Items] |
|
|
|
|
| Revenue from collaboration agreements |
€ 0
|
€ 983
|
€ 0
|
€ 2,547
|
| X |
- DefinitionDisclosure of revenue from collaboration agreements.
| Name: |
imtx_DisclosureOfRevenueFromCollaborationAgreementsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionRevenue from collaboration agreements
| Name: |
imtx_RevenueFromCollaborationAgreements |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_CollaborationAgreementsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_GeographicalAreasAxis=country_DK |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_GenmabCollaborationAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_GeographicalAreasAxis=country_US |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_BMSCollaborationAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_GeographicalAreasAxis=country_GB |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_GSKCollaborationAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Revenue from collaboration agreements - Summary of Deferred revenue related to the collaboration agreements (Detail) - EUR (€)
€ in Thousands |
Jun. 30, 2023 |
Dec. 31, 2022 |
| Revenue [abstract] |
|
|
| Current |
€ 67,997
|
€ 64,957
|
| Non-current |
53,559
|
75,759
|
| Total |
€ 121,556
|
€ 140,716
|
| X |
- DefinitionThe amount of an entity’s obligation to transfer goods or services to a customer for which the entity has received consideration (or the amount is due) from the customer. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 105
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_105&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 116
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_116_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ContractLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current contract liabilities. [Refer: Contract liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 105
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_105&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentContractLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of non-current contract liabilities. [Refer: Contract liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 15
-IssueDate 2022-03-24
-Paragraph 105
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=15&code=ifrs-tx-2022-en-r&anchor=para_105&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentContractLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_RevenueAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Financial result - Summary of other financial income and financial expenses (Detail) - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Disclosure Of Detailed Information About Finance Income Expense [Abstract] |
|
|
|
|
| Interest income |
€ 2,744
|
€ 69
|
€ 4,999
|
€ 75
|
| Foreign currency gains |
1,210
|
6,946
|
1,749
|
8,699
|
| Other financial income |
3,954
|
7,015
|
6,748
|
8,774
|
| Interest expenses |
(206)
|
(376)
|
(401)
|
(538)
|
| Foreign currency losses |
(805)
|
(31)
|
(4,119)
|
(986)
|
| Losses on other financial instruments |
(133)
|
0
|
(133)
|
0
|
| Other financial expenses |
(1,144)
|
(407)
|
(4,653)
|
(1,524)
|
| Change in fair value of liabilities for warrants |
(13,105)
|
(2,786)
|
(5,708)
|
13,743
|
| Finance income (cost) |
€ (10,295)
|
€ 3,822
|
€ (3,613)
|
€ 20,993
|
| X |
- DefinitionThe amount of costs associated with financing activities of the entity. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 82
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_82_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinanceCosts |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of income associated with interest and other financing activities of the entity. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 85
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_85&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinanceIncome |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of income or cost associated with interest and other financing activities of the entity. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 85
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_85&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinanceIncomeCost |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of expense arising from interest. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_e&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23_d&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B13
-Subparagraph f
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B13_f&doctype=Appendix&subtype=B
-URIDate 2022-03-24
| Name: |
ifrs-full_InterestExpense |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of interest income on other financial assets. [Refer: Interest income; Other financial assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_InterestIncomeOnOtherFinancialAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe net gain arising from exchange differences recognised in profit or loss, excluding those arising on financial instruments measured at fair value through profit or loss in accordance with IFRS 9. [Refer: Foreign exchange gain (loss)] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 21
-IssueDate 2022-03-24
-Paragraph 52
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=21&code=ifrs-tx-2022-en-r&anchor=para_52_a&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 35
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_35&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NetForeignExchangeGain |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionChange in fair value of warrant liabilities.
| Name: |
imtx_ChangeInFairValueOfWarrantLiabilities |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionForeign currency losses net.
| Name: |
imtx_ForeignCurrencyLossesNet |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionLoss recognised on financial instruments whose fair value previously could not be reliably measured.
| Name: |
imtx_LossRecognisedOnFinancialInstrumentsWhoseFairValuePreviouslyCouldNotBeReliablyMeasured |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
v3.23.2
Financial result - Additional Information (Detail)
€ / shares in Units, € in Thousands |
3 Months Ended |
6 Months Ended |
|
|
|
|
|
Jun. 30, 2023
EUR (€)
€ / shares
|
Jun. 30, 2022
EUR (€)
€ / shares
|
Jun. 30, 2023
EUR (€)
€ / shares
|
Jun. 30, 2022
EUR (€)
€ / shares
|
Jun. 30, 2023
$ / shares
|
Mar. 31, 2023
€ / shares
|
Mar. 31, 2023
$ / shares
|
Jun. 30, 2022
$ / shares
|
Mar. 31, 2022
€ / shares
|
Mar. 31, 2022
$ / shares
|
| Statements [Line Items] |
|
|
|
|
|
|
|
|
|
|
| Change in fair value of warrant liabilities |
€ (13,105)
|
€ (2,786)
|
€ (5,708)
|
€ 13,743
|
|
|
|
|
|
|
| Top of range [member] |
|
|
|
|
|
|
|
|
|
|
| Statements [Line Items] |
|
|
|
|
|
|
|
|
|
|
| Exercise Price of Warrants or Rights | (per share) |
€ 3.15
|
€ 1.96
|
€ 3.15
|
€ 1.96
|
$ 3.42
|
|
|
$ 2.04
|
|
|
| Bottom of range [member] |
|
|
|
|
|
|
|
|
|
|
| Statements [Line Items] |
|
|
|
|
|
|
|
|
|
|
| Exercise Price of Warrants or Rights | (per share) |
|
|
|
|
|
€ 1.32
|
$ 1.44
|
|
€ 1.58
|
$ 1.75
|
| X |
- DefinitionChange in fair value of warrant liabilities.
| Name: |
imtx_ChangeInFairValueOfWarrantLiabilities |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionClass Of Warrant Or Right Exercise Price Of Warrants Or Rights1
| Name: |
imtx_ClassOfWarrantOrRightExercisePriceOfWarrantsOrRights1 |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
imtx_StatementsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_RangeAxis=ifrs-full_TopOfRangeMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_RangeAxis=ifrs-full_BottomOfRangeMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Income Tax - Additional Information (Detail) - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
|
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
Dec. 31, 2022 |
| Statements [Line Items] |
|
|
|
|
|
| Statutory tax rate |
28.50%
|
28.50%
|
28.50%
|
28.50%
|
|
| Corporate income tax rate |
21.00%
|
21.00%
|
21.00%
|
21.00%
|
|
| Current tax liabilities |
€ 4,298
|
|
€ 4,298
|
|
€ 4,298
|
| GERMANY | Immatics Biotechnologies GmbH [Member] |
|
|
|
|
|
| Statements [Line Items] |
|
|
|
|
|
| Percentage Of Income That Can Be Used To Set Off Tax Losses Carry Forward |
|
60.00%
|
|
|
|
| Percentage Of Pretax Income Subject To Income Tax |
|
40.00%
|
|
|
|
| Current tax liabilities |
|
€ 1,200
|
|
€ 1,200
|
|
| X |
- DefinitionThe applicable income tax rate. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 12
-IssueDate 2022-03-24
-Paragraph 81
-Subparagraph c
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=12&code=ifrs-tx-2022-en-r&anchor=para_81_c_ii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ApplicableTaxRate |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of current tax for current and prior periods to the extent unpaid. Current tax is the amount of income taxes payable (recoverable) in respect of the taxable profit (tax loss) for a period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph n
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_n&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentTaxLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionPercentage of income that can be used to set off tax losses carry forward.
| Name: |
imtx_PercentageOfIncomeThatCanBeUsedToSetOffTaxLossesCarryForward |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
imtx_StatementsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
srt_StatementGeographicalAxis=country_DE |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_SignificantInvestmentsInSubsidiariesAxis=imtx_ImmaticsBiotechnologiesGmbhMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe expected volatility of the share price used to calculate the fair value of the share options granted. Expected volatility is a measure of the amount by which a price is expected to fluctuate during a period. The measure of volatility used in option pricing models is the annualised standard deviation of the continuously compounded rates of return on the share over a period of time. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfExpectedVolatilityShareOptionsGranted |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe option life of share options granted. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfOptionLifeShareOptionsGranted |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe implied yield currently available on zero-coupon government issues of the country in whose currency the exercise price for share options granted is expressed, with a remaining term equal to the expected term of the option being valued (based on the option's remaining contractual life and taking into account the effects of expected early exercise). [Refer: Government [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfRiskFreeInterestRateShareOptionsGranted |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfTermsAndConditionsOfSharebasedPaymentArrangementLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exercise price of share options granted. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ExercisePriceShareOptionsGranted2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe percentage of an expected dividend used to calculate the fair value of share options granted. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ExpectedDividendAsPercentageShareOptionsGranted |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:percentItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average share price used as input to the option pricing model to calculate the fair value of share options granted. [Refer: Option pricing model [member]; Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 47
-Subparagraph a
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_47_a_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageSharePriceShareOptionsGranted2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_AdditionalGrantsUnderTwoThousandAndTwentyPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Share-based payments - Summary of Employee Share Options (Detail)
|
6 Months Ended |
|
Jun. 30, 2023
Warrants
shares
$ / shares
|
|---|
| Performance Based Options [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| Weighted average exercise price, Outstanding at January 1, |
$ 10.08
|
| SARs forfeited |
10
|
| Weighted average exercise price, Outstanding at June 30, |
10.08
|
| SARs exercisable |
$ 0
|
| Number of share options, outstanding at January 1, | shares |
3,666,000
|
| SARs forfeited | shares |
12,000
|
| Number of share options, outstanding at June 30, | shares |
3,654,000
|
| SARs exercisable | shares |
0
|
| Weighted average remaining contract life (years) |
7 years 18 days
|
| Performance Based Options [Member] | Granted On September 2021 [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| SARs granted |
$ 0
|
| SARs granted | shares |
0
|
| Additional Grants Under 2020 and 2022 Plan [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| Weighted average exercise price, Outstanding at January 1, |
$ 10.07
|
| SARs forfeited |
9.8
|
| SARs exercised |
9.51
|
| SARs expired |
10.75
|
| Weighted average exercise price, Outstanding at June 30, |
10.02
|
| SARs exercisable |
$ 10.07
|
| Number of share options, outstanding at January 1, | Warrants |
6,129,160
|
| SARs forfeited | shares |
145,494
|
| SARs exercised | Warrants |
2,707
|
| SARs expired | shares |
17,825
|
| Number of share options, outstanding at June 30, | shares |
6,469,297
|
| SARs exercisable | shares |
2,135,617
|
| Weighted average remaining contract life (years) |
8 years 6 months 25 days
|
| Additional Grants Under 2020 and 2022 Plan [Member] | Granted On March 2021 [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| SARs granted |
$ 9.48
|
| SARs granted | Warrants |
506,163
|
| Converted Options [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| Weighted average exercise price, Outstanding at January 1, |
$ 2.74
|
| SARs exercised |
1.28
|
| SARs expired |
1.17
|
| Weighted average exercise price, Outstanding at June 30, |
2.78
|
| SARs exercisable |
$ 2.78
|
| Number of share options, outstanding at January 1, | Warrants |
525,181
|
| SARs exercised | shares |
11,341
|
| SARs expired | shares |
8
|
| Number of share options, outstanding at June 30, | shares |
512,923
|
| SARs exercisable | shares |
469,161
|
| Weighted average remaining contract life (years) |
4 years 6 months 3 days
|
| Converted Options [Member] | Granted On June 2020 [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| SARs forfeited |
$ 1.14
|
| SARs forfeited | shares |
909
|
| Matching Stock Options [Member] |
|
| Disclosure of terms and conditions of share-based payment arrangement [line items] |
|
| Weighted average exercise price, Outstanding at January 1, |
$ 10
|
| SARs forfeited |
0
|
| SARs exercised |
10
|
| SARs expired |
10
|
| Weighted average exercise price, Outstanding at June 30, |
10
|
| SARs exercisable |
$ 10
|
| Number of share options, outstanding at January 1, | Warrants |
1,348,004
|
| SARs forfeited | shares |
0
|
| SARs exercised | Warrants |
720
|
| SARs expired | shares |
4,280
|
| Number of share options, outstanding at June 30, | shares |
1,343,004
|
| SARs exercisable | shares |
1,343,004
|
| Weighted average remaining contract life (years) |
7 years 3 days
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfTermsAndConditionsOfSharebasedPaymentArrangementLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of share options outstanding in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_d&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_i&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause vi
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_vi&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfOutstandingShareOptions |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe number of share options exercisable in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause vii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_vii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfShareOptionsExercisableInSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe number of share options exercised in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause iv
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_iv&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfShareOptionsExercisedInSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of share options expired in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause v
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_v&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfShareOptionsExpiredInSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of share options forfeited in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_iii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfShareOptionsForfeitedInSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of share options granted in a share-based payment arrangement. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_ii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfShareOptionsGrantedInSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:decimalItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options exercisable in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause vii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_vii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsExercisableInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe weighted average exercise price of share options exercised in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause iv
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_iv&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsExercisedInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options expired in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause v
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_v&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsExpiredInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options forfeited in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_iii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsForfeitedInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options granted in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_ii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsGrantedInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options outstanding in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_i&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause vi
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_vi&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsOutstandingInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe weighted average remaining contractual life of outstanding share options. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageRemainingContractualLifeOfOutstandingShareOptions2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:durationItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_DateOfGrantAxis=imtx_GrantedOnSeptemberTwoThousandAndTwentyOneMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_AdditionalGrantsUnderTwoThousandAndTwentyPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_DateOfGrantAxis=imtx_GrantedOnMarchTwoThousandAndTwentyOneMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_ConvertedOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_DateOfGrantAxis=imtx_GrantedOnJuneTwoThousandAndTwentyMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_MatchingStockOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe description of the maximum term of options granted for a type of share-based payment arrangement that existed at any time during the period. An entity with substantially similar types of share-based payment arrangements may aggregate this information. [Refer: Share-based payment arrangements [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfMaximumTermOfOptionsGrantedForSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe description of the vesting requirements for a type of share-based payment arrangement that existed at any time during the period. An entity with substantially similar types of share-based payment arrangements may aggregate this information. [Refer: Share-based payment arrangements [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DescriptionOfVestingRequirementsForSharebasedPaymentArrangement |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfTermsAndConditionsOfSharebasedPaymentArrangementLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe weighted average exercise price of share options granted in a share-based payment arrangement. [Refer: Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 2
-IssueDate 2022-03-24
-Paragraph 45
-Subparagraph b
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=2&code=ifrs-tx-2022-en-r&anchor=para_45_b_ii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageExercisePriceOfShareOptionsGrantedInSharebasedPaymentArrangement2019 |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionDate of vesting of stock options.
| Name: |
imtx_DateOfVestingOfStockOptions |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionMinimum market capitalization.
| Name: |
imtx_MinimumMarketCapitalization |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionShare based compensation by share based payment arrangement weighted average fair value per share.
| Name: |
imtx_ShareBasedCompensationByShareBasedPaymentArrangementWeightedAverageFairValuePerShare |
| Namespace Prefix: |
imtx_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_TrancheAxis=imtx_TrancheOneMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_TrancheAxis=imtx_TrancheTwoMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_TrancheAxis=imtx_TrancheThreeMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_AdditionalGrantsUnderTwoThousandAndTwentyPlanMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_DateOfGrantAxis=imtx_GrantedInJuneTwoThousandAndTwentyTwoMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_TypesOfSharebasedPaymentArrangementsAxis=imtx_ConvertedOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_DateOfGrantAxis=imtx_GrantedOnJuneTwoThousandAndTwentyMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_ShareReinvestmentSchemeAxis=imtx_MatchingStockOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Share-based payments - Summary of Employee Related Share Based Compensation Expense (Detail) - EUR (€)
€ in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Disclosure Of Detailed Information About Share Based Compensation Expense [Line Items] |
|
|
|
|
| Share-based compensation expense |
€ (5,513)
|
€ (5,560)
|
€ (11,615)
|
€ (11,262)
|
| Research And Development Expense [Member] |
|
|
|
|
| Disclosure Of Detailed Information About Share Based Compensation Expense [Line Items] |
|
|
|
|
| Share-based compensation expense |
(3,282)
|
(3,107)
|
(6,815)
|
(6,375)
|
| General And Administrative Expense [Member] |
|
|
|
|
| Disclosure Of Detailed Information About Share Based Compensation Expense [Line Items] |
|
|
|
|
| Share-based compensation expense |
€ (2,231)
|
€ (2,453)
|
€ (4,800)
|
€ (4,887)
|
| X |
- DefinitionThe amount of expense from share-based payment transactions with employees. [Refer: Expense from share-based payment transactions] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ExpenseFromSharebasedPaymentTransactionsWithEmployees |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_IncomeStatementLocationAxis=imtx_ResearchAndDevelopmentExpenseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_IncomeStatementLocationAxis=imtx_GeneralAndAdministrativeExpenseMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Other current and non-current assets -Summary of Other current assets (Detail) - EUR (€)
€ in Thousands |
Jun. 30, 2023 |
Dec. 31, 2022 |
| Trade and other receivables [abstract] |
|
|
| Prepaid expenses |
€ 9,546
|
€ 10,450
|
| Value added tax receivables |
1,605
|
1,031
|
| Other assets |
5,517
|
2,357
|
| Total |
€ 16,668
|
€ 13,838
|
| X |
- DefinitionThe amount recognised as a current asset for expenditures made prior to the period when the economic benefit will be realised. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentPrepaidExpenses |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current value added tax receivables. [Refer: Value added tax receivables] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentValueAddedTaxReceivables |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of assets that the entity does not separately disclose in the same statement or note. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current assets that the entity does not separately disclose in the same statement or note. [Refer: Current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherCurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_TradeAndOtherReceivablesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfSignificantInvestmentsInSubsidiariesLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of non-current prepayments. [Refer: Prepayments] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentPrepayments |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionCurrent incremental cost for collaboration agreement.
| Name: |
imtx_CurrentIncrementalCostForCollaborationAgreement |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionCurrent prepaid insurance.
| Name: |
imtx_CurrentPrepaidInsurance |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionCurrent prepaid maintenance expenses.
| Name: |
imtx_CurrentPrepaidMaintenanceExpenses |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionNon current prepaid maintenance expenses.
| Name: |
imtx_NonCurrentPrepaidMaintenanceExpenses |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionNoncurrent prepayments of licensing agreement.
| Name: |
imtx_NoncurrentPrepaymentsOfLicensingAgreement |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionPrepaid expenses of licenses and software.
| Name: |
imtx_PrepaidExpensesOfLicensesAndSoftware |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
imtx_CollaborationAgreementAxis=imtx_CelgeneSwitzerlandLlcAndGenmabAsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe amount of non-current prepayments. [Refer: Prepayments] Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NoncurrentPrepayments |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_NoncurrentReceivablesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of non-current assets that the entity does not separately disclose in the same statement or note. [Refer: Non-current assets] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherNoncurrentAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
v3.23.2
| X |
- DefinitionThe amount of additions to property, plant and equipment other than those acquired through business combinations. [Refer: Business combinations [member]; Property, plant and equipment] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 16
-IssueDate 2022-03-24
-Paragraph 73
-Subparagraph e
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=16&code=ifrs-tx-2022-en-r&anchor=para_73_e_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdditionsOtherThanThroughBusinessCombinationsPropertyPlantAndEquipment |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of additions to right-of-use assets. [Refer: Right-of-use assets] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 16
-IssueDate 2022-03-24
-Paragraph 53
-Subparagraph h
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=16&code=ifrs-tx-2022-en-r&anchor=para_53_h&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdditionsToRightofuseAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
imtx_StatementsLineItems |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfPropertyPlantAndEquipmentAxis=ifrs-full_LeaseholdImprovementsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfAssetsAxis=imtx_RentalLandMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionThe amount of liabilities of uncertain timing or amount, including provisions for employee benefits. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph l
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_l&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_Provisions |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- References
+ Details
| Name: |
ifrs-full_ProvisionsAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of provisions for employee benefits. [Refer: Employee benefits expense; Provisions] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProvisionsForEmployeeBenefits |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.23.2
| X |
- References
+ Details
| Name: |
ifrs-full_TradeAndOtherCurrentPayablesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe current amount of payment due to suppliers for goods and services used in entity's business. [Refer: Current liabilities; Trade payables] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 70
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_70&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_TradeAndOtherCurrentPayablesToTradeSuppliers |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of payment due to suppliers for goods and services used in the entity's business. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_TradeAndOtherPayablesToTradeSuppliers |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.23.2
| X |
- DefinitionThe amount of accruals classified as current. [Refer: Accruals] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AccrualsClassifiedAsCurrent |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current provisions for employee benefits. [Refer: Provisions for employee benefits] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 78
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_78_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentProvisionsForEmployeeBenefits |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current tax for current and prior periods to the extent unpaid. Current tax is the amount of income taxes payable (recoverable) in respect of the taxable profit (tax loss) for a period. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph n
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_n&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentTaxLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe current amount of current tax liabilities. [Refer: Current tax liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 54
-Subparagraph n
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_54_n&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_CurrentTaxLiabilitiesCurrent |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of current liabilities that the entity does not separately disclose in the same statement or note. [Refer: Current liabilities] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 55
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_55&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_OtherCurrentLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
v3.23.2
| X |
- DefinitionThe maturity of borrowings. [Refer: Borrowings] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 7
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_7&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_BorrowingsMaturity |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_BorrowingsByNameAxis=imtx_NonInterestBearingLiabilityMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Shareholders' equity - Additional Information (Detail) - EUR (€)
€ / shares in Units, € in Thousands |
6 Months Ended |
|
Jun. 30, 2023 |
Jun. 30, 2022 |
Dec. 31, 2022 |
| Disclosure of classes of share capital [line items] |
|
|
|
| Gross proceeds from issuance of shares |
€ 38,608
|
€ 17,112
|
|
| SVB Securities LLC [Member] | At The Market Agreement [Member] |
|
|
|
| Disclosure of classes of share capital [line items] |
|
|
|
| Increase decrease in shares outstanding |
3,700,000
|
|
|
| Gross proceeds from issuance of shares |
€ 38,600
|
|
|
| Share issuance costs |
1,200
|
|
|
| Increase in share capital |
37
|
|
|
| Increase in share premium |
€ 37,400
|
|
|
| Reorganization [Member] |
|
|
|
| Disclosure of classes of share capital [line items] |
|
|
|
| Number of shares outstanding |
80,397,851
|
|
76,670,699
|
| Par value per share |
€ 0.01
|
|
€ 0.01
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfClassesOfShareCapitalLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe increase (decrease) in the number of ordinary shares issued. [Refer: Ordinary shares [member]] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 112
-Subparagraph c
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_112_c&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_IncreaseDecreaseInNumberOfOrdinarySharesIssued |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares that have been authorised and issued, reduced by treasury shares held. [Refer: Treasury shares] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 79
-Subparagraph a
-Clause iv
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_79_a_iv&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfSharesOutstanding |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe nominal value per share. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 79
-Subparagraph a
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_79_a_iii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ParValuePerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash inflow from issuing shares. Reference 1: http://www.xbrl.org/2003/role/exampleRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 17
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_17_a&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProceedsFromIssuingShares |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease in share capital.
| Name: |
imtx_IncreaseInShareCapital |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionIncrease in share premium.
| Name: |
imtx_IncreaseInSharePremium |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
imtx_InvesteeAxis=imtx_SvbSecuritiesLlcMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_AtTheMarketAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_EventAxis=imtx_ReorganizationMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- Details
| Name: |
imtx_TypeOfVestingAxis=imtx_ServiceOptionsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
Financial Instruments - Summary of Carrying Amounts and Fair values of Group's Financial Instruments (Detail) - EUR (€)
€ in Thousands |
Jun. 30, 2023 |
Dec. 31, 2022 |
| Carrying amount [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
|
€ 58,756
|
| At fair value [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
|
58,300
|
| Cash and cash equivalents [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
€ 130,405
|
148,519
|
| Short-Term Deposits [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
217,222
|
154,930
|
| Bonds [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
|
58,756
|
| Bonds [member] | Carrying amount [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
|
58,756
|
| Bonds [member] | At fair value [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
|
58,300
|
| Accounts receivables [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
330
|
1,111
|
| Other current/non-current assets [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
5,158
|
2,402
|
| Accounts payable [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
19,212
|
11,735
|
| Other current liabilities [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
58
|
54
|
| Liabilities for warrants [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
22,622
|
16,914
|
| Lease liabilities [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets and Financial liabilities |
16,822
|
14,563
|
| Financial liabilities at fair value through profit or loss, category [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
22,622
|
16,914
|
| Financial liabilities at fair value through profit or loss, category [member] | Liabilities for warrants [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
22,622
|
16,914
|
| Financial liabilities at amortised cost, category [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
36,092
|
26,352
|
| Financial liabilities at amortised cost, category [member] | Accounts payable [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
19,212
|
11,735
|
| Financial liabilities at amortised cost, category [member] | Other current liabilities [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
58
|
54
|
| Financial liabilities at amortised cost, category [member] | Lease liabilities [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial liabilities, Carrying amount |
16,822
|
14,563
|
| Financial assets at amortised cost, category [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
353,115
|
365,718
|
| Financial assets at amortised cost, category [member] | Cash and cash equivalents [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
130,405
|
148,519
|
| Financial assets at amortised cost, category [member] | Short-Term Deposits [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
217,222
|
154,930
|
| Financial assets at amortised cost, category [member] | Bonds [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
|
58,756
|
| Financial assets at amortised cost, category [member] | Accounts receivables [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
330
|
1,111
|
| Financial assets at amortised cost, category [member] | Other current/non-current assets [member] |
|
|
| Disclosure of detailed information about financial instruments [line items] |
|
|
| Financial assets, Carrying amount |
€ 5,158
|
€ 2,402
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfFinancialInstrumentsLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of assets that are: (a) cash; (b) an equity instrument of another entity; (c) a contractual right: (i) to receive cash or another financial asset from another entity; or (ii) to exchange financial assets or financial liabilities with another entity under conditions that are potentially favourable to the entity; or (d) a contract that will, or may be, settled in the entity’s own equity instruments and is: (i) a non-derivative for which the entity is, or may be, obliged to receive a variable number of the entity’s own equity instruments; or (ii) a derivative that will, or may be, settled other than by the exchange of a fixed amount of cash or another financial asset for a fixed number of the entity’s own equity instruments. For this purpose the entity’s own equity instruments do not include puttable financial instruments classified as equity instruments in accordance with paragraphs 16A-16B of IAS 32, instruments that impose on the entity an obligation to deliver to another party a pro rata share of the net assets of the entity only on liquidation and are classified as equity instruments in accordance with paragraphs 16C-16D of IAS 32, or instruments that are contracts for the future receipt or delivery of the entity’s own equity instruments. [Refer: Financial instruments, class [member]; Financial liabilities] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 35H
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_35H&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 35M
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_35M&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 35N
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_35N&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 35I
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_35I&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 25
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_25&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinancialAssets |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- DefinitionThe amount of liabilities that are: (a) a contractual obligation: (i) to deliver cash or another financial asset to another entity; or (ii) to exchange financial assets or financial liabilities with another entity under conditions that are potentially unfavourable to the entity; or (b) a contract that will, or may be, settled in the entity’s own equity instruments and is: (i) a non-derivative for which the entity is, or may be, obliged to deliver a variable number of the entity’s own equity instruments; or (ii) a derivative that will, or may be, settled other than by the exchange of a fixed amount of cash or another financial asset for a fixed number of the entity’s own equity instruments. For this purpose, rights, options or warrants to acquire a fixed number of the entity’s own equity instruments for a fixed amount of any currency are equity instruments if the entity offers the rights, options or warrants pro rata to all of its existing owners of the same class of its own non-derivative equity instruments. Also, for those purposes the entity’s own equity instruments do not include puttable financial instruments that are classified as equity instruments in accordance with paragraphs 16A-16B of IAS 32, instruments that impose on the entity an obligation to deliver to another party a pro rata share of the net assets of the entity only on liquidation and are classified as equity instruments in accordance with paragraphs 16C-16D of IAS 32, or instruments that are contracts for the future receipt or delivery of the entity’s own equity instruments. As an exception, an instrument that meets the definition of a financial liability is classified as an equity instrument if it has all the features and meets the conditions in paragraphs 16A-16B or paragraphs 16C-16D of IAS 32. [Refer: Financial instruments, class [member]; Financial assets; Derivatives [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 7
-IssueDate 2022-03-24
-Paragraph 25
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=7&code=ifrs-tx-2022-en-r&anchor=para_25&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_FinancialLiabilities |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
instant |
|
| X |
- DefinitionFinancial assets and Financial liabilities.
| Name: |
imtx_FinancialAssetsAndFinancialLiabilities |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
instant |
|
| X |
- Details
| Name: |
ifrs-full_MeasurementAxis=ifrs-full_CarryingAmountMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_MeasurementAxis=ifrs-full_AtFairValueMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_CashAndCashEquivalentsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_ShorttermDepositsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_BondsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=ifrs-full_TradeReceivablesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_OtherCurrentNonCurrentAssetsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_AccountsPayableMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_OtherCurrentLiabilitiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=imtx_LiabilitiesForWarrantsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_ClassesOfFinancialAssetsAxis=ifrs-full_LeaseLiabilitiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_CategoriesOfFinancialLiabilitiesAxis=ifrs-full_FinancialLiabilitiesAtFairValueThroughProfitOrLossCategoryMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_CategoriesOfFinancialLiabilitiesAxis=ifrs-full_FinancialLiabilitiesAtAmortisedCostCategoryMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
ifrs-full_CategoriesOfFinancialAssetsAxis=ifrs-full_FinancialAssetsAtAmortisedCostCategoryMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
v3.23.2
| X |
- DefinitionDilutive effect of share options on number of warrants.
| Name: |
imtx_DilutiveEffectOfShareOptionsOnNumberOfWarrants |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
imtx_TextBlockAbstract |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Earnings and Loss per Share - Summary of Earnings and Loss per Share (Detail) - EUR (€)
€ / shares in Units, € in Thousands |
3 Months Ended |
6 Months Ended |
Jun. 30, 2023 |
Jun. 30, 2022 |
Jun. 30, 2023 |
Jun. 30, 2022 |
| Earnings per share [abstract] |
|
|
|
|
| Net profit/(loss): |
€ (24,610)
|
€ (13,980)
|
€ (44,357)
|
€ 71,682
|
| Basic |
€ (0.32)
|
€ (0.22)
|
€ (0.58)
|
€ 1.12
|
| Diluted |
€ (0.32)
|
€ (0.22)
|
€ (0.58)
|
€ 1.11
|
| Weighted average shares outstanding: |
|
|
|
|
| Basic |
77,311,053
|
64,915,600
|
76,994,713
|
63,932,449
|
| Diluted |
77,311,053
|
64,915,600
|
76,994,713
|
64,477,256
|
| X |
- DefinitionThe weighted average number of ordinary shares outstanding plus the weighted average number of ordinary shares that would be issued on the conversion of all the dilutive potential ordinary shares into ordinary shares. [Refer: Ordinary shares [member]; Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 70
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_70_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_AdjustedWeightedAverageShares |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator) divided by the weighted average number of ordinary shares outstanding during the period (the denominator). Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 67
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_67&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_BasicEarningsLossPerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe amount of profit (loss) attributable to ordinary equity holders of the parent entity (the numerator), divided by the weighted average number of ordinary shares outstanding during the period (the denominator), both adjusted for the effects of all dilutive potential ordinary shares. [Refer: Ordinary shares [member]; Weighted average [member]] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 66
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_66&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 67
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_67&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_DilutedEarningsLossPerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_EarningsPerShareAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe total of income less expenses from continuing and discontinued operations, excluding the components of other comprehensive income. [Refer: Other comprehensive income] Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 18
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_18_b&doctype=Standard
-URIDate 2022-03-24
Reference 2: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 32
-Subparagraph a
-Clause ii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_32_a_ii&doctype=Standard
-URIDate 2022-03-24
Reference 3: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 1
-IssueDate 2022-03-24
-Paragraph 24
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=1&code=ifrs-tx-2022-en-r&anchor=para_24_b&doctype=Standard
-URIDate 2022-03-24
Reference 4: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 28
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_28_b&doctype=Standard
-URIDate 2022-03-24
Reference 5: http://www.xbrl.org/2003/role/exampleRef
-Note Effective on first application of IFRS 9
-Name IFRS
-Number 4
-IssueDate 2022-03-24
-Paragraph 39L
-Subparagraph e
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=4&code=ifrs-tx-2022-en-b&anchor=para_39L_e&doctype=Standard
-URIDate 2022-03-24
Reference 6: http://www.xbrl.org/2003/role/disclosureRef
-Name IFRS
-Number 8
-IssueDate 2022-03-24
-Paragraph 23
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=8&code=ifrs-tx-2022-en-r&anchor=para_23&doctype=Standard
-URIDate 2022-03-24
Reference 7: http://www.xbrl.org/2003/role/exampleRef
-Name IFRS
-Number 12
-IssueDate 2022-03-24
-Paragraph B10
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=12&code=ifrs-tx-2022-en-r&anchor=para_B10_b&doctype=Appendix&subtype=B
-URIDate 2022-03-24
Reference 8: http://www.xbrl.org/2003/role/exampleRef
-Note Effective 2023-01-01
-Name IFRS
-Number 17
-IssueDate 2022-03-24
-Paragraph 113
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IFRS&num=17&code=ifrs-tx-2022-en-r&anchor=para_113_b&doctype=Standard
-URIDate 2022-03-24
Reference 9: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 81A
-Subparagraph a
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_81A_a&doctype=Standard
-URIDate 2022-03-24
Reference 10: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-Clause i
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d_i&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProfitLoss |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of ordinary shares outstanding at the beginning of the period, adjusted by the number of ordinary shares bought back or issued during the period multiplied by a time-weighting factor. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 33
-IssueDate 2022-03-24
-Paragraph 70
-Subparagraph b
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=33&code=ifrs-tx-2022-en-r&anchor=para_70_b&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_WeightedAverageShares |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- References
+ Details
| Name: |
ifrs-full_WeightedAverageSharesAndAdjustedWeightedAverageSharesAbstract |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
v3.23.2
Events occurring after the reporting period - Additional Information (Detail) - Issue Of Securities [Member]
$ / shares in Units, € in Millions, $ in Millions |
Aug. 17, 2023
USD ($)
shares
|
Aug. 17, 2023
EUR (€)
shares
|
Jul. 19, 2023
USD ($)
$ / shares
shares
|
| Disclosure of non-adjusting events after reporting period [line items] |
|
|
|
| Number of shares issued |
|
|
2,419,818
|
| Share Price | $ / shares |
|
|
$ 14.46
|
| Proceeds from issue of ordinary shares | $ |
|
|
$ 35.0
|
| SVB Securities LLC [Member] | At The Market Agreement [Member] |
|
|
|
| Disclosure of non-adjusting events after reporting period [line items] |
|
|
|
| Number of shares issued |
1,800,000
|
1,800,000
|
|
| Gross proceeds from issue of securities |
$ 22.2
|
€ 20.4
|
|
| X |
- DefinitionLine items represent concepts included in a table. These concepts are used to disclose reportable information associated with members defined in one or many axes of the table.
| Name: |
ifrs-full_DisclosureOfNonadjustingEventsAfterReportingPeriodLineItems |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:stringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe number of shares issued by the entity. Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 106
-Subparagraph d
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_106_d&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_NumberOfSharesIssued |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:sharesItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe nominal value per share. Reference 1: http://www.xbrl.org/2003/role/disclosureRef
-Name IAS
-Number 1
-IssueDate 2022-03-24
-Paragraph 79
-Subparagraph a
-Clause iii
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=1&code=ifrs-tx-2022-en-r&anchor=para_79_a_iii&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ParValuePerShare |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
dtr-types:perShareItemType |
| Balance Type: |
na |
| Period Type: |
instant |
|
| X |
- DefinitionThe cash inflow from the issuing of ordinary shares. [Refer: Ordinary shares [member]] Reference 1: http://www.xbrl.org/2009/role/commonPracticeRef
-Name IAS
-Number 7
-IssueDate 2022-03-24
-Paragraph 17
-URI https://taxonomy.ifrs.org/xifrs-link?type=IAS&num=7&code=ifrs-tx-2022-en-r&anchor=para_17&doctype=Standard
-URIDate 2022-03-24
| Name: |
ifrs-full_ProceedsFromIssueOfOrdinaryShares |
| Namespace Prefix: |
ifrs-full_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
debit |
| Period Type: |
duration |
|
| X |
- DefinitionGross proceeds from issue of securities.
| Name: |
imtx_GrossProceedsFromIssueOfSecurities |
| Namespace Prefix: |
imtx_ |
| Data Type: |
xbrli:monetaryItemType |
| Balance Type: |
credit |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
ifrs-full_NonadjustingEventsAfterReportingPeriodAxis=imtx_IssueOfSecuritiesMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_InvesteeAxis=imtx_SvbSecuritiesLlcMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
imtx_AgreementAxis=imtx_AtTheMarketAgreementMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|

















































































