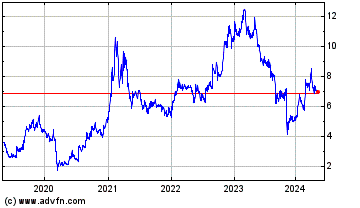
EDAP TMS (NASDAQ:EDAP)
Historical Stock Chart
From Jul 2019 to Jul 2024